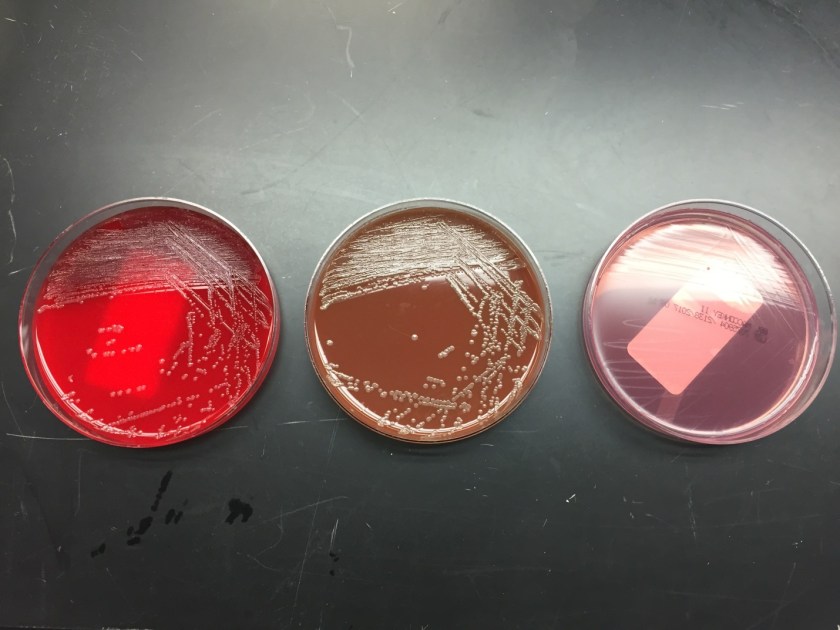
Everyone understands that listening is an essential skill for effective leadership. But many people do not realize that there are multiple ways to listen to someone. No, I am not talking about active and passive listening, where you either do or don’t reflect back what was just said. Honestly, reflective listening can become quite awkward during a long conversation… been there, done that (and not recommended).
The five different listening approaches that this course refers to are either feeling oriented, meaning that people listen to the emotions behind what someone is saying, or information oriented, meaning that they listen for data, arguments, and structure. The two feeling oriented listening approaches are appreciative and empathic which listen for a speaker’s enthusiasm and how to provide support respectively. The information oriented approaches are discerning, comprehensive, and evaluative. Discerning listeners want to gather complete and accurate information, comprehensive listeners organize the information, and evaluative listeners critique the information and make decisions.
The job of the leader (and speaker) is to provide the information and emotion that their listeners respond to well. The key is, therefore, to provide something for each of the five types of listeners when you are giving your next presentation.

-Lotte Mulder earned her Master’s of Education from the Harvard Graduate School of Education in 2013, where she focused on Leadership and Group Development. She’s currently working toward a PhD in Organizational Leadership. At ASCP, Lotte designs and facilitates the ASCP Leadership Institute, an online leadership certificate program. She has also built ASCP’s first patient ambassador program, called Patient Champions, which leverages patient stories as they relate to the value of the lab.
Considering myself to be a “good listener,” I was particularly interested in the “Listening with Purpose” course during the leadership institute program. I was keen to see how the “scientific approach” of the leadership institute would be applied to this particular skill. Before the course, I answered a series of questions; my answers were analysed and I received a personalized listening profile. This profile included statements about my listening tendencies in specific situations. Some were uncannily accurate: “You may mentally argue with speakers as they talk,” for example, is a true description of my frequent mental disposition at meetings and conferences!
In addition to identifying my natural listening approach (the “information-oriented evaluative” style), my profile also indicated how likely I am to use the other listening approaches. Not surprising to me was the observation that I tend not to adopt the more feeling-oriented approaches of appreciative and empathic listening. My attitude has typically been to “cut through the red tape and stick with the facts.”
Beyond the characterization of my natural listening style, my personal profile also provided guidance on how to work with my listening style and identified areas where I could grow as a listener. It was clear that with regards to listening approaches, one size does not fit all. My “good listening” skills need a lot improvement. Miscommunication, with a loss of intended meaning, will occur if I consistently use only my natural listening style without regard for the differing circumstances and situations. This certainly explains some of my struggles with people who try to appeal to my emotions instead of (or in spite of) facts to make decisions.
The course also covered communication, defined as the actual exchange of information. The course highlights the various reasons why people try to communicate: to persuade, to inform, to self-express, or to entertain. I was surprised to learn that over fifty percent of face-to-face communication may be dependent on body language and less than ten percent on the words spoken! One then has to be careful to actually process what is being said rather than just the speaker’s body language, tone, and energy so as not to lose the message.
The most important message I took from the course was the importance of letting people know you are listening to them. This requires situational awareness and understanding the intention of the speaker. I found especially useful the behavioural indicators that let a speaker know the listener is engaged. I have actually found practical utility and deployed a couple of these: “Demonstrating an open, laidback posture” and “remaining relatively silent, not offering solutions immediately.” The course has shown me a practical way to improve my listening skills.

-Dr. Modupe Kuti, MB, MS(Ib), FWACP (Lab Med), has served as an attending pathologist for the past thirteen years for a tertiary hospital in Southwest Nigeria, West Africa.