We are all aware that there is substantial waste in testing. The mantra of utilization management is “the right test for the right patient at the right time.” This month, I want to focus on the right time. It turns out that many test results are never seen because they arrive after the patient has been discharged. This occurs for both routine and send-out testing. I will examine both.
Turnaround times for send-out testing are generally longer than those for tests performed in house. This means that results for tests ordered toward the end of a hospital stay are likely to be received after the patient has been discharged. Sendout tests are often expensive and, unlike tests performed in house, reducing sendout testing saves the hospital the full charge of the test. The savings can be substantial.
How do you prevent this? A recent article by Fang et al. shows one approach.[1] In this study, conducted at Stanford University, researcher displayed the cost and turnaround time of sendout tests in the computerized provider order entry (CPOE) system and achieved a 26% reduction in orders. I am aware of another hospital that restricts orders of sendout tests when the expected turnaround time is close to the expected remaining length of stay. Consider the graph in Figure 1. The upper panel shows the expected length of stay for a particular patient. The lower panel shows the expected turnaround time for a sendout test. In this case, there is a 62% chance that the test result will arrive after the patient has left the hospital. Expected discharge dates are routinely kept and it is relatively easy to maintain a database of turnaround times. A hospital could combine these data and set a threshold for orders based on the probability that the result will arrive in time.
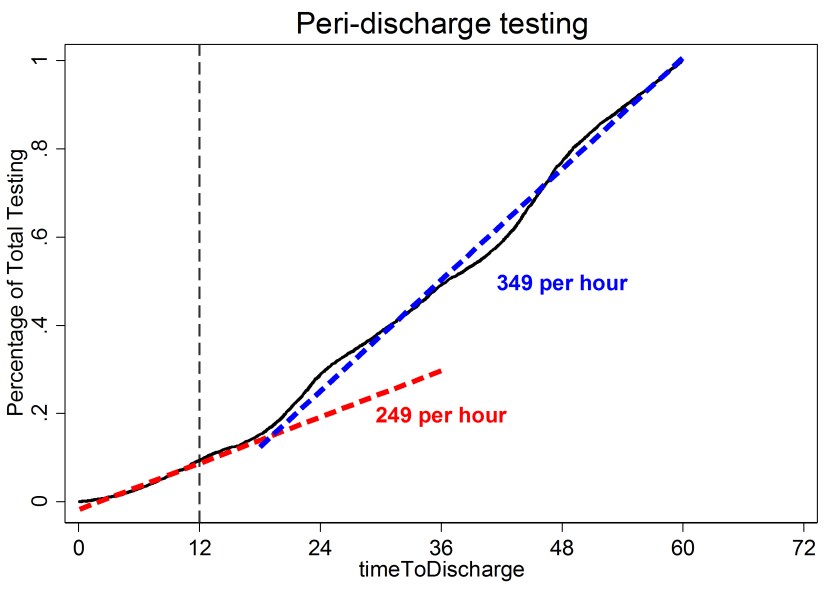
Standing orders are another source of waste. I recently performed an analysis of the test rate as a function of the time until discharge (Figure 2). The test rate was 249 tests per hour for patients who were within 12 hours of discharge and 349 tests per hour for all other patients. It seems odd to me the testing rate in the final 12 hours is 70% of the “normal” testing rate. Further, the distribution of tests in both groups (those about to be discharged vs. all other patients) is very similar (Table 1). The main tests are basic metabolic panels and complete blood counts. I suspect the majority of the testing within 12 hours of discharge is due to standing orders and the results were not needed for patient care. The best intervention is less clear in this case because some peri-discharge testing is appropriate and it is difficult to distinguish the appropriate testing from the inappropriate testing. Education is one option. Perhaps the CPOE could raise a flag on orders for patients who are about to be discharged; however, this could be cumbersome and clinicians object to flags and popups that interfere with their workflow. I would be interested in readers’ thoughts on methods to reduce inappropriate peri-discharge testing.
In summary, some results do not reach clinicians in time to affect patient care. This is a source of waste. It is relatively easy to create an intervention to reduce inappropriate sendout testing but more difficult to reduce unnecessary peri-discharge testing.
Reference
- Fang DZ, Sran G, Gessner D, Loftus PD, Folkins A, Christopher JY, III, Shieh L: Cost and turn-around time display decreases inpatient ordering of reference laboratory tests: A time series. BMJ Quality and Safety 2014, 23(12):994-1000.



-Robert Schmidt, MD, PhD, MBA, MS is a clinical pathologist who specializes in the economic evaluation of medical tests. He is currently an Associate Professor at the University of Utah where he is Medical Director of the clinical laboratory at the Huntsman Cancer Institute and Director of the Center for Effective Medical Testing at ARUP Laboratories.



