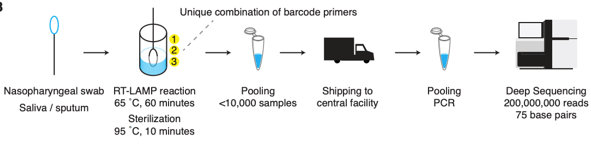
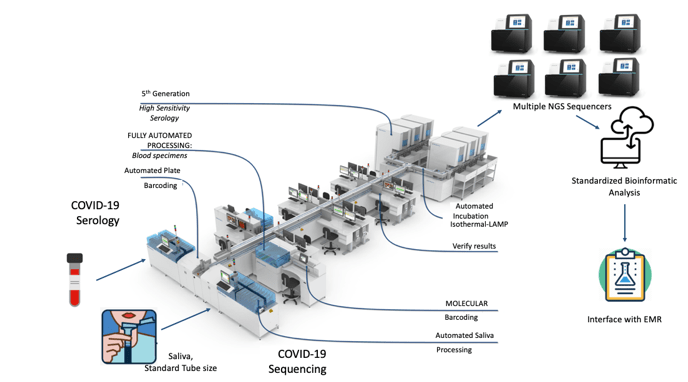
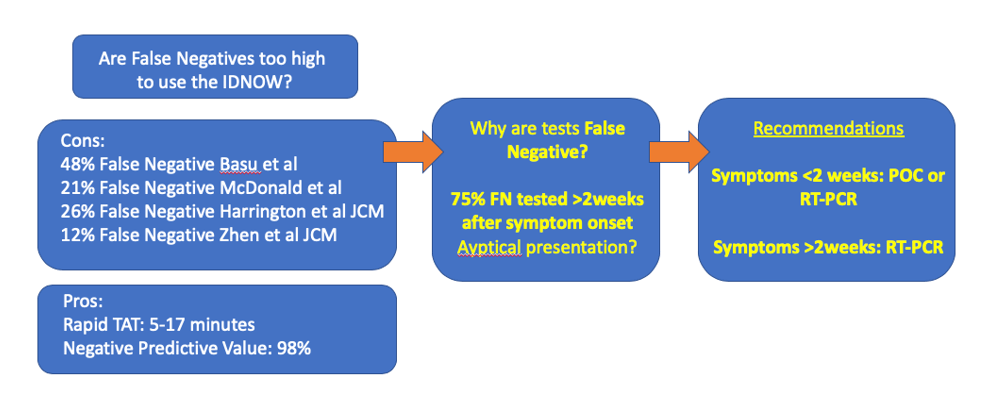
Welcome to October 2020 and a flu season unlike any other. What can we expect? Well, it’s complicated. And if we aren’t sure what to expect, can we still be prepared? Yes (at least for some things)!
From the beginning of the COVID-19 pandemic and throughout the summer of 2020 clinicians and laboratorians have been anxiously wondering what effect global presence of respiratory virus SARS-CoV-2 would have on the 2020-2021 flu season. “Flu season,” the annual, relatively predictable period of increased cases and deaths due to Influenza A and B, occurs during colder, winter months. In the northern hemisphere this is September through March. We have extensive experience tracking the onset and genetic variability of the predominant influenza viruses. We manufacturer flu vaccines based on data of potentially likely influenza strains. Other viruses that cause respiratory symptoms follow similar seasonal patterns. These include common (non-SARS-CoV-2) human coronaviruses, and Respiratory Syncytial Virus (RSV). In short: this is a known, annual occurrence that we can usually prepare to some extent.
So what will that look like this year? During the historic 1918 pandemic influenza, deaths seen during the first winter of the outbreak paled in comparison to those seen the following winter. Even if that kind of terrible scenario doesn’t occur during this pandemic year, it is possible we are facing “perfect storm” of COVID-19 plus influenza resulting in overwhelmed hospitals and depleted testing supplies. [https://www.cidrap.umn.edu/news-perspective/2020/09/fears-perfect-storm-flu-season-nears]
We know that COVID-19 spreads well in enclosed spaces with prolonged person-to-person contact, regardless of climate and temperature, via respiratory secretions. Because of this, there has been widespread adoption of mask wearing, social distancing, and limitations on in-person gathering. Promisingly, these interventions to prevent the spread of COVID-19 seem to be contributing to historically low influenza rates in the Southern Hemisphere! [https://www.cdc.gov/mmwr/volumes/69/wr/mm6937a6.htm] But adoption of these mitigation strategies are not being universally or rigorously followed in all regions and communities. As temperatures drop, we could see more people conducting activity indoors – will this change transmission patterns? Will regions with ongoing COVID-19 outbreaks be more prone to influenza as well? If hospital capacity becomes strained, will criteria for ordering tests change?
During COVID-19 laboratories have responded heroically and rapidly to test kit shortages, supply chain issues, and staffing challenges. At this stage (October of 2020) many high-level decisions about SARS-CoV-2 testing, like test platform purchasing and validation or manufacturer test kit allocations, might already be set in stone. So is there anything that can be done to help labs and laboratory workers successfully make it through flu season?
Here are 3 suggestions:
1) Establish testing algorithms and clear sample workflows.
Each facility and laboratory will have their own platforms for testing COVID-19 and other respiratory pathogens. Depending on the service ordering the test, there can be both immediate and downstream consequences for when a test comes back positive, negative, or even when that test result is slower than expected!
An algorithm helps set institutional expectations for what tests are ordered under different scenarios. For example symptomatic patients presenting to a hospital with influenza-like illness (ILI), especially when they will be admitted, should likely have both SARS-CoV-2 and influenza tests ordered simultaneously. But asymptomatic patients being admitted for procedures may only require a SARS-CoV-2 test.
Let’s say your lab has both a SARS-CoV-2 PCR test and SARS-CoV-2 rapid antigen test. But due to risk a false negative, lab and clinical leaders are uncomfortable using only a rapid antigen test to conclusively rule out COVID-19 in patients being admitted to the hospital. Your algorithm could use specify the use of SARS-CoV-2 antigen testing in symptomatic patients to quickly “rule in” potential positives, where antigen-negative patients will also have a PCR test. Algorithm specifics come down to what your institutions stake holders (clinical AND laboratory) need and capacity are. The details of an algorithm will be dependent on your lab test platforms, your available test orders, and may need to be modified to accommodate restricted test allocations.
Along with clinical algorithms, clear workflow for specimens and test types can help laboratory workers get tests where they need to go within the lab. Not all SARS-CoV-2 tests have approval in the instructions for use for, say, nasal swabs. If nasal swab comes to the lab with orders for both influenza and SARS-CoV-2 tests, what is the procedure for informing the floor for an appropriate collection? Or say that your test platforms for different tests live in different areas of the lab. Your workflow may be to set up one test and do a pour off into an aliquot tube so tests can be run at the same time. Or you may have sufficient test collection materials to request a separate sample for each test.
Probably the most important part of developing or reviewing your existing algorithms and laboratory workflow is doing it in connection with others. The purpose is to streamline the entire process from clinical decision making to test performing and reporting and help everyone be on the same page.
2) Communicate to clinical staff frequently about your tests.
Because of the intense interest surrounding COVID-19 laboratory testing, it’s entirely possible that more people have had to learn about previously niche laboratory concepts like “sensitivity vs. specificity” and “PCR vs. antibody vs. antigen tests” than at any previously time in human history! However, it is also likely that many clinicians or administrators in your own institution may know more about a test platform they read about in the news than the COVID-19 test platform that their laboratory performs.
Even at this stage in the pandemic with perhaps more exposure (pun not intended!) then the laboratory has ever had, miscommunication and unclear expectations abound surrounding test performance or turnaround times.
Whenever possible, lab leaders who interact with clinicians and administrators should look for ways to educate on test platforms, testing capacity, and expected test performance (i.e. time to result, comparative sensitivity etc.). This could include asking for time to provide formal updates during monthly meetings, monitoring test statistics (e.g. a test “dashboard”), or just informal reminders about what tests the lab performs during phone calls.
3) Keep the lab staff off the phone.
A critical part of the job of the lab is to provide information and updates on when test results are available. But when the hospital floors or clinics are busiest with patients, often the lab is busiest performing those patients’ tests. A phone call about the status of a respiratory virus test can be undeniably helpful to that patient’s clinical care team! But a dozen such phone calls over the course of a lab worker’s shift, especially under normal lab conditions (e.g. no staff shortages or instrument issues) is a failure of communication and can be detrimental to both lab performance and lab worker wellbeing.
In addition to the need for regular education about testing mentioned above, to help protect your lab staff’s bench time here are some possible ways keep from being overwhelmed with phone calls:
- In some institutions, passive reminders (for example about hand hygiene or upcoming events) cycle through computer screen savers or on television screens in clinical areas. You could see if a message like “Reminder from the lab: COVID-19 tests are completed in [length of time].” could be put on a rotation.
- If there is no client service or switchboard for your lab, but people call the lab directly for updates, you could institute a message stop. This is where phone calls routed to the laboratory must listen to a reminder that (for example), “If you are calling for an update of a COVID-19 test, these tests cannot be completed faster than [length of time] after arriving in the lab.”
While these messages can be undeniably annoying and disruptive for people calling the lab for other reasons (and become less effective over time) if phone calls get out of hand, this option could be considered. - A lab instrument going down can result in test backlogs and numerous phone calls to the lab. Some institutions centralize their information in the form of a duty officer (for example in the emergency department). This will be a person who can be informed of actionable information, like test delays due to instrument issues, and who will post and distribute that information to those affected.
There is a lot we don’t know about what’s to come in the COVID-19 pandemic. While we can’t predict the ways the lab may be challenged with the next unforeseen disruption, or even what our flu season testing needs may look like, hopefully we can prepare now to continue to support our patients by helping and supporting our labs.

-Dr. Richard Davis, PhD, D(ABMM), MLS(ASCP)CM is a clinical microbiologist and regional director of microbiology for Providence Health Care in Eastern Washington. A certified medical laboratory scientist, he received his PhD studying the tropical parasite Leishmania. He transitioned back to laboratory medicine (though he still loves parasites!), and completed a clinical microbiology fellowship at the University of Utah/ARUP Laboratories in Utah before accepting his current position. He is a 2020 ASCP 40 Under Forty Honoree.