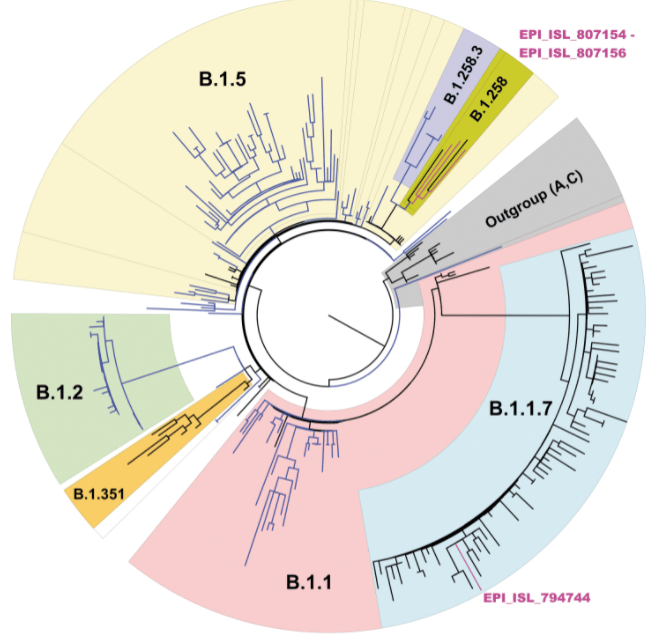
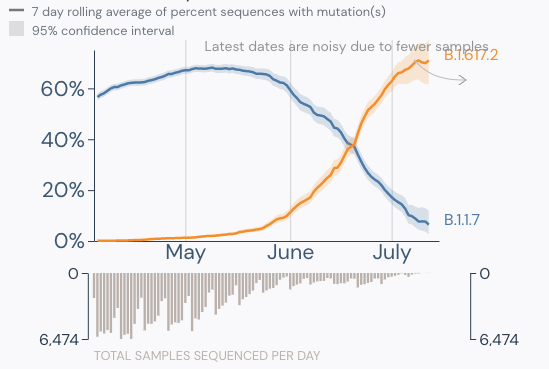
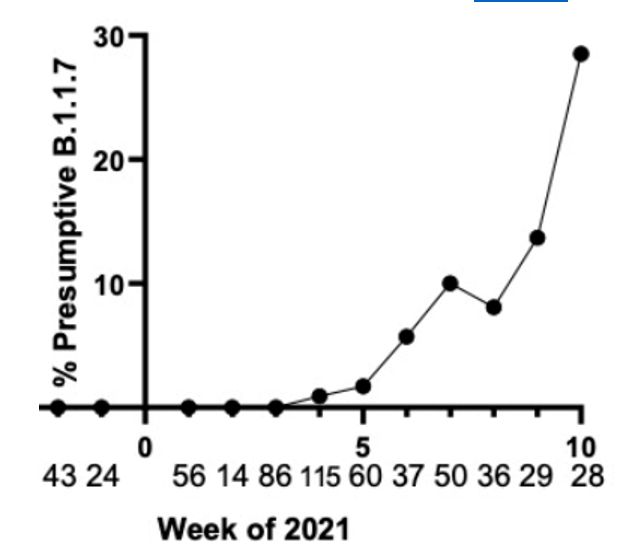
Omicron is now the dominant variant in the United States and gained that title faster than any variant before it. I have been tracking variants in the North Texas region since February of this year and detected the first Alpha variant (B.1.1.7). During this time, there were multiple substrains circulating. Some like Epsilon (origin California) rose in prominence then declined to extinction. Rise in Alpha (origin U.K.) and Delta variants (B.1.617.2, origin India) were tracked over the course of weeks, but Omicron has been tracked on a daily basis, since it is rising so quickly.
Many places are using S-Gene Target Failure (SGTF) as a surrogate for Omicron variant (Yale, University of Washington below).
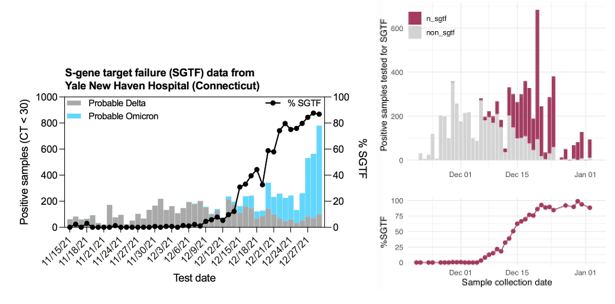
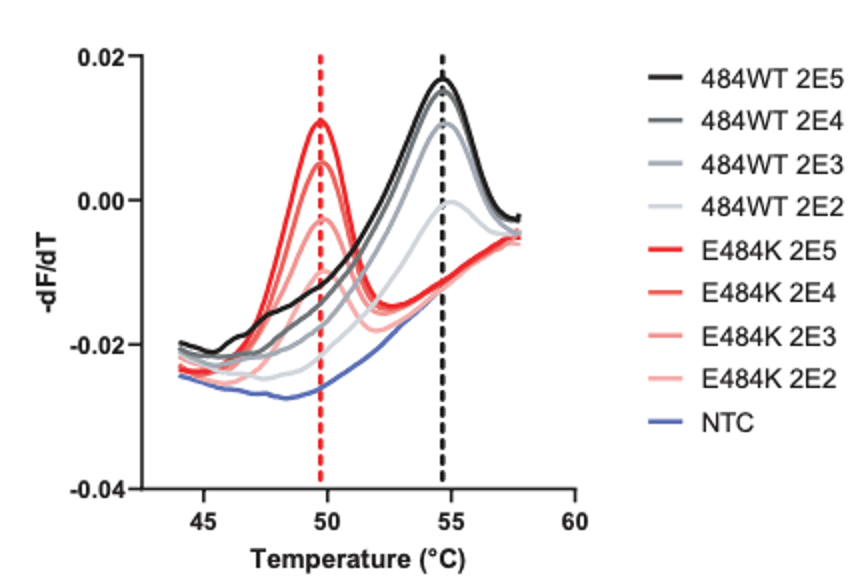
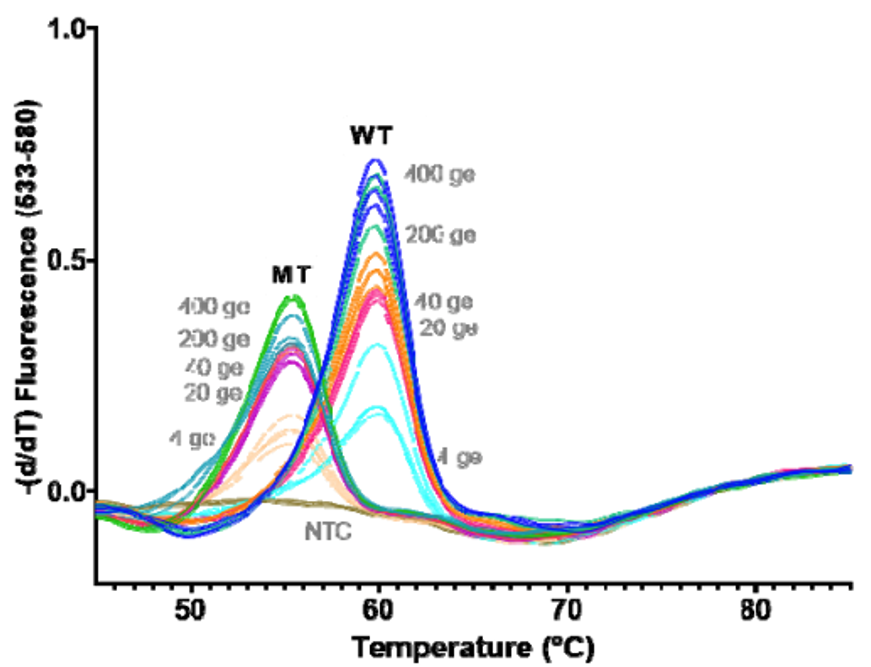
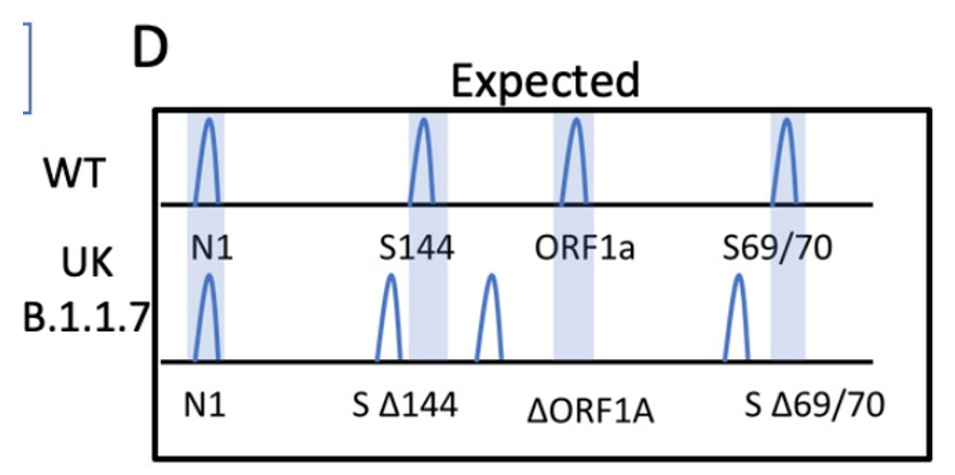
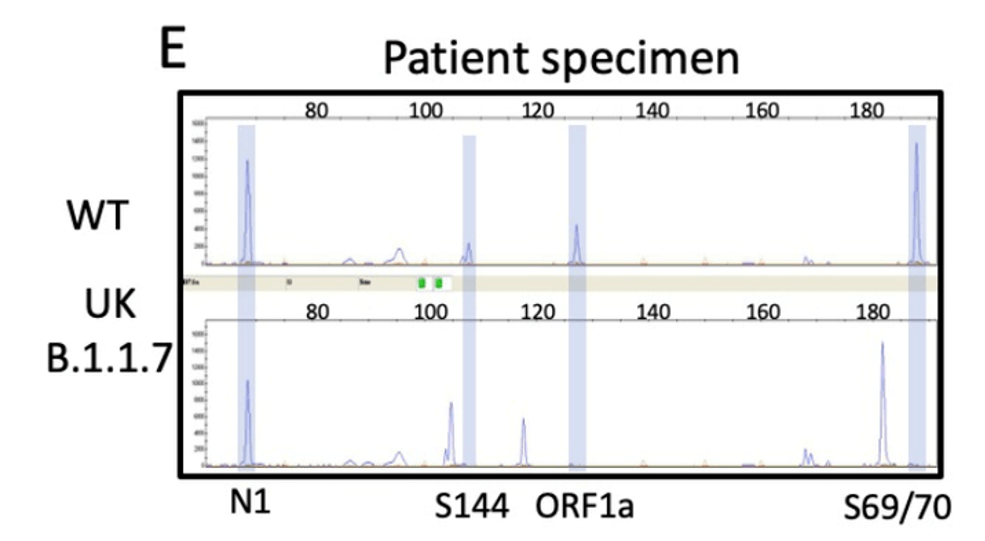
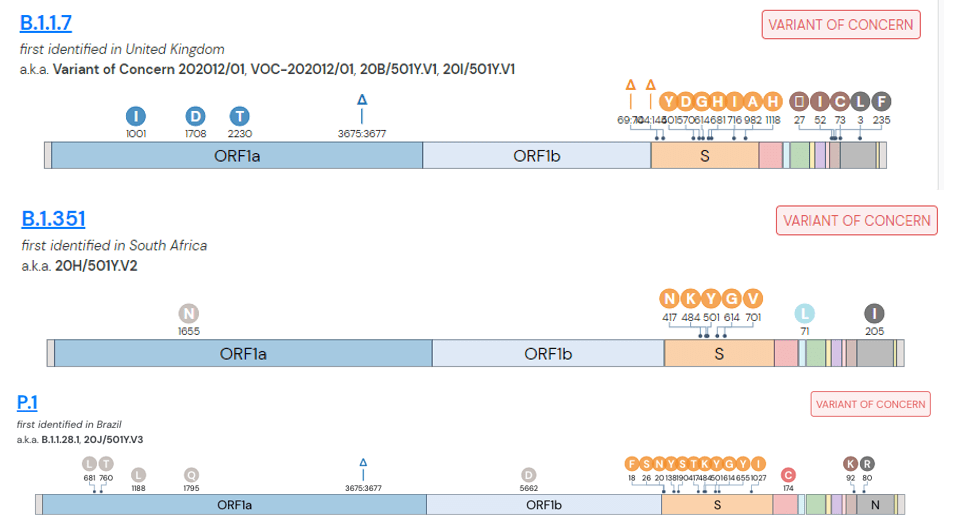
SGTF occurs when the TaqPath COVID-19 multiplex test has 2/3 targets successfully amplify when the S-gene target does not or “drops out.” This phenomenon was first observed in the Alpha variant, because the probe for this target overlapped a characteristic mutation: S:Del69_70 (deletion of the 69th and 70th amino acids in the spike protein from a 6 base pair deletion). This mutation is absent in Delta, but present in Omicron, so has been used as an early tracker of Omicron prevalence.

Most of this discussion is speculative and we won’t ever really know, but given the rate of transmission of this variant, it seems unlikely that it would have acquired so many mutations and not been detected before now. The most recent common ancestor is from over a year ago suggesting it was incubating for a long time.
We’ve seen a case of a person severely immunocompromised with no antibody response to vaccination + booster who still has an unmutated wild type strain in their system. With no immune pressure, the virus has not evolved.
However, in HIV+ patients with variable/ low immunity, there could be enough pressure to drive the immune evasion properties seen in Omicron. Southern Africa has over 30% of their HIV+ patients not on therapy who would be likely candidates for this type of host.
Did we see this coming?
Yes. Other immune evasive variants have arisen in areas with high prevalence of previous infection (Brazil/ S. Africa). Organisms evolve just enough to overcome the challenges in their environment. Thus the level of immunity provided by various immune exposures are approximately:
Previous infection < 2x Vaccine < 2x Vaccine+ previous infection ~ x3 Vaccine
Scientists theorized that either Delta would evolve more immune evasive mutations or a totally new variant would arise. However, I didn’t think it would spread this quickly.
What is the impact?
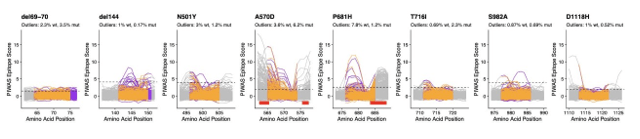
Therapies. Most antibody therapies are directed as the business end of the spike protein—the receptor binding domain (RBD). The rest of the protein is covered in glycosylation modifications that block much recognition. Thus with many mutations in Omicron compared to the wild type strain (white), most therapeutic antibodies no longer bind/ inactivate viral replication.
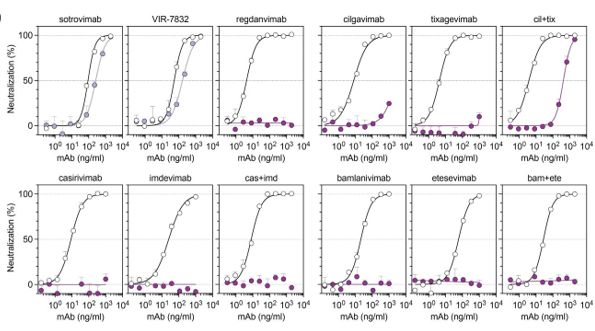
Only one monoclonal antibody—Sotrovimab from GSK—is effective, because it binds a pan-coronovirus epitope outside of the RBD. However, this antibody is in short supply.
- Thus, knowing which variant someone has can direct therapy. Several hospitals in our area are out of Sotrovimab, and only people with the Delta variant can access other options. Thus, knowing the variant in a short time frame has clinical implications.
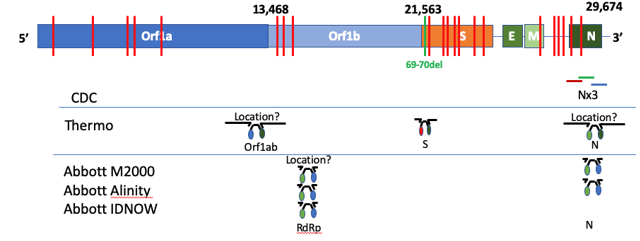
- Whole genome sequencing takes too long, so the FDA has agreed to review PCR genotyping approaches for clinical use. I have described some previous approaches, but many of these methods are useful as a screening method and would not have sufficient specificity to determine whether an omicron variant is present. Next time, I will discuss variant genotyping, why it is important, how it can be done, and what clinical actions can be taken with the knowledge.
Severity. There are signs that it is less severe. Is this due to increase in immune tolerance? We now have been prepared by either previous infection or vaccination to be protected from hospitalization or severe disease.
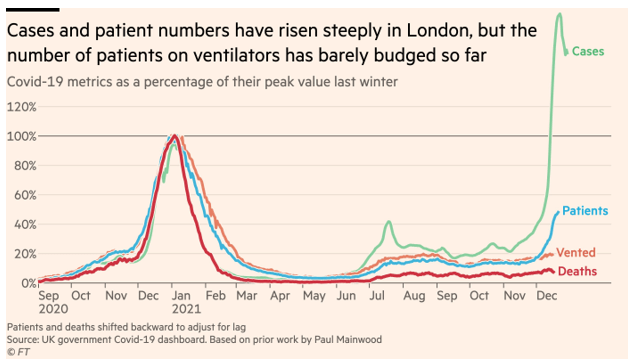
Or is the decline in severity due to lower pathogenicity? A recent non-peer reviewed study indicates the virus replicates x70 faster than Delta in the upper airways (left), but infiltrates cells 10% as well as the original strain.
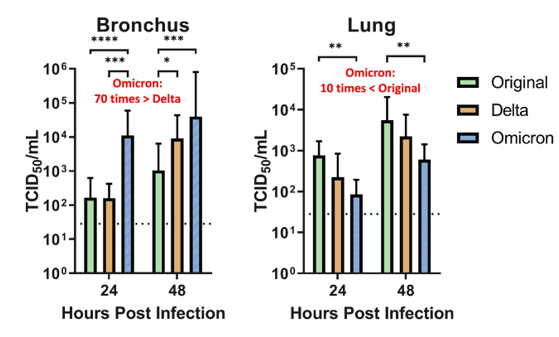
We all hope this will continue to be better news about the severity of Omicron, but from the lab side, I’ve heard of positivity rates >50% at some places. So this can still have a broad impact.

-Jeff SoRelle, MD is Assistant Professor of Pathology at the University of Texas Southwestern Medical Center in Dallas, TX working in the Next Generation Sequencing lab. His research interests include the genetics of allergy, COVID-19 variant sequencing, and lab medicine of transgender healthcare. Follow him on Twitter @Jeff_SoRelle.