Population COVID-19 testing
Population-wide testing to identify symptomatic and asymptomatic infections could be a powerful tool to control Coronvirus Disease 2019 (COVID-19) spread, but current global testing capacity does not permit widespread testing of asymptomatic individuals. These tests are still limited to individuals who are symptomatic with limited availability to those with recent exposure to an infected person.
Because of the high prevalence of asymptomatic COVID-19 infections, proposals from the Rockefeller Foundation for disease mitigation include widespread and frequent testing of the US population. In the United States, diagnostic testing for SARS-CoV-2, the causative virus of COVID-19 is currently >2 million per week. Estimates for US testing needs for population wide surveillance range from 30 to 300 million per week. In order to scale testing by an order of magnitude, novel technologies and rethinking current testing paradigms are needed. The NIH has initiated a rapid funding program to develop SARS-CoV-2 testing, and these new technologies may play a part. However, we can broadly conceptualize key problems to address in population-wide testing in the US. The first is high-sensitivity testing which identifies active infection and can be performed with massive throughput. The second is the logistics of gathering hundreds of thousands of samples to each testing laboratory each day.
Next Generation Solutions to COVID testing
Emerging technologies using targeted next-generation sequencing have been suggested as a potential solution to population-wide testing. The key features include 1) extraction free amplification 2) an easily collected specimen such as saliva, 3) nucleotide barcodes to enable sample pooling, and 4) a limited number of targets (to allow deeper sequencing, i.e. higher sensitivity). Illumina is selling a whole genome test for SARS-CoV-2, but this limits sequencing to 3,000 tests/ run. Another recent approval for a private testing lab uses only one target, and may allow it to increase to 100,000 tests/ day. And a recent protocol for LAMP-Seq in pre-print outlines how this could work in a scheme below. An attractive aspect of this approach is decentralized specimen processing.
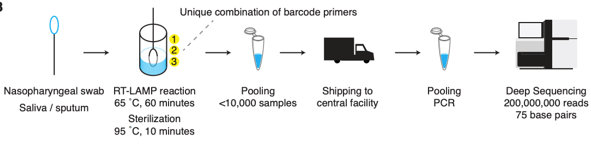
Whereas Bill Gates has supported a portfolio approach to vaccines placing multiple bets on different processes in parallel, a similar approach should be applied to multiplexed sequencing methods. Two sequencing runs can be performed on a single instrument in a single day, which can process several thousand samples. However, sequencing is not the only step in sequencing; library preparation and specimen handling take significant amounts of time too.
Laboratory Logistics
This technology would represent an exponential expansion in analytic testing capacity, but clinical labs will require a similar escalation in logistic capacity. The largest clinical laboratories in the world process less than 100,000 samples per day. Clinical laboratories have a long history of automation with the first robotic specimen track systems developed in the 1980s. Engineering and clinical lab expertise should thus partner to innovate on methods to handle high volumes. This level of investment for an issue that is likely to fade in 2 years, is not attractive to most private health systems, so public investment from multiple states in regional reference labs is needed.
It is still hard to conceive the necessary scale up in sample processing can be achieve within the time frame needed, so I would also propose a de-centralized sample processing approach. This would include self-collection of saliva (a safe, effective sample type with similar sensitivity as nasopharyngeal swabs), drop-off sites, and processing at places like Pharmacies (>90% of Americans live within 5 miles of a pharmacy and they could be authorized to administer tests- just as they administer vaccines). This would introduce pre-analytic problems, but if the goal is frequent and high rates of testing, then we will have to accept certain losses in sensitivity (which currently is arguably better than it needs to be). Interestingly, pre-analytic concerns with saliva have not led to sample instability or degradation of RNA causing false negatives, as described in my last post. However, other factors could affect saliva quality: smoking, age, and genetic factors of water: protein ratio affecting viscosity.
Testing solutions should be considered in the context of the planned testing network. The specimen type should be easy for the patient to provide, processed with existing laboratory equipment and resulted electronically. For example, current COVID-19 testing is based on sample collections requiring a healthcare worker encased in personal protective equipment (PPE) utilizing a swab device. Testing needs to progress to a simpler solution such as saliva which can be collected by the patient in the absence of a swab or PPE. Preliminary studies have demonstrated that saliva is sample type comparable to nasopharyngeal swab. The ideal saliva sample would be collected into an existing collection tube type (e.g. red-top tubes) which are already compatible with existing laboratory automation. In aggregate, a person could spit into a tube at-home, have the tube sent to a laboratory, and in the laboratory the tube would be directly placed onto an automated robotic track system.
Laboratory professionals need to provide a comprehensive plan for regional and national laboratory networks which can scale to provide overwhelming force to COVID-19 testing. No other profession or governmental organization understands testing as much as we do. Our understanding of managing samples from collection to result should be applied to the pandemic at hand. Until now most laboratorians in the US have focused on the immediate needs of providing testing for symptomatic patients and healthcare workers.
Vision for automated COVID-19 testing
One could envision an automated line of testing that moves samples through processing to allow multiplexing and combinations of samples to allow large numbers of patients to be tested at once (see below). This is feasible in some specialized centers, but would require investments in automation, bioinformatics, and interfaces for a seamless process (figure below). If testing mostly asymptomatic patients, it may also be possible to do this on pooled samples. The number of samples to pool would depend on the likelihood to having a positive result (this would require sequencing all individuals in a pool).
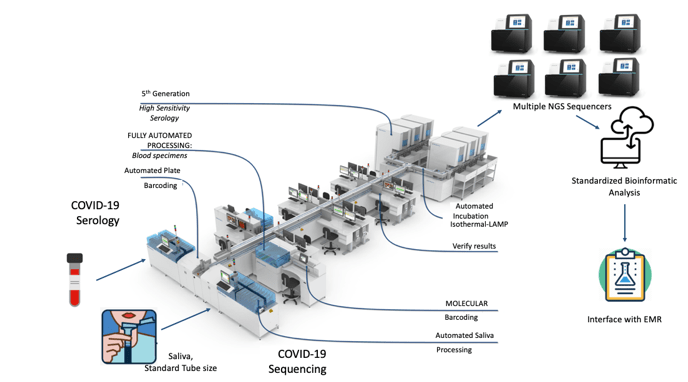
This represents a synthesis of ideas in decentralized specimen collection, laboratory automation and massive testing throughput with Next-Generation Sequencing, but unfortunately this is not yet a reality.
References
- Jonathan L. Schmid-Burgk et al. LAMP-Seq: Population-Sclae COVID-19 Diagnostics Using Combinatorial Barcoding. bioRxiv 2020.04.06.025635.
- The Rockefeller Foundation. National Covid-19 Testing Action Plan Pragmatic steps to reopen our workplaces and our communities. 2020.
- Cahill TJ, Cravatt B, Goldman LR, Iwasaki A, Kemp RS, Lin MZ et al. Scientists to Stop COVID-19. OR Rob Copeland, Wall Street Journal (2020) The Secret Group of Scientists and Billionaires Pushing a Manhattan Project for Covid-19. April 27
- https://www.illumina.com/products/by-type/ivd-products/covidseq.html

-Jeff SoRelle, MD is a Chief Resident of Pathology at the University of Texas Southwestern Medical Center in Dallas, TX. His clinical research interests include understanding how the lab intersects with transgender healthcare and improving genetic variant interpretation.
