Case History
An 80 year old male was seen by his cardiologist for approximately one month of dyspnea, fatigue, and weight loss. Past medical history was significant for aortic stenosis requiring placement of a bioprosthetic valve and multivessel coronary artery disease 13 years prior. He underwent cardiac catheterization and echocardiography that revealed severe bioprosthetic valve stenosis. The patient was in the process of evaluation for a prosthetic valve replacement when he presented to the emergency room for rapid decline of the previously noted symptoms. Exam upon hospital admission was notable for cardiac murmur, lower extremity edema, mild leukocytosis, and anemia. He had normal dentition and no skin lesions. A pre-operative TEE confirmed severe aortic prosthetic valve stenosis, restricted leaflet motion, thrombus on all three leaflets, and thickening of the periannular aortic root and ascending aorta. Subsequent cardiac CT was concerning for either pseudoaneurysm or paravalvular leak suggestive of an infectious or inflammatory process.
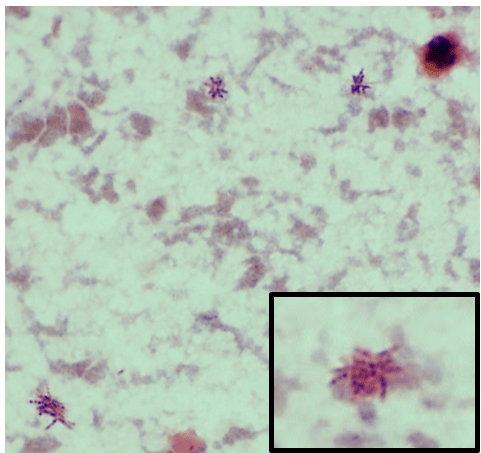
Due to the persistent, mild leukocytosis, blood cultures were obtained on the second day of admission. On hospital day 3, one set of blood cultures flagged positive with Gram-variable rods in the aerobic bottle (Image 1). The patient was empirically started on vancomycin and piperacillin/tazobactam. Repeat blood cultures were obtained on hospital days 4 and 7, both again positive for Gram-variable rods within 2 days of collection. The infectious diseases consult team suspected subacute bacterial endocarditis and changed therapy to ceftriaxone.On hospital day 9, the patient underwent a redo sternotomy for aortic valve replacement and aortic root repair. Intraoperative findings included a large amount of phlegmon on the aortic leaflets, near circumferential aortic annulus tissue destruction and abscess cavity. Culture of the intraoperative specimens was negative for bacterial growth. The anatomic pathology findings revealed fibrinoid vegetations and acute inflammation and reparative changes. The patient was subsequently discharged home in stable condition 20 days after his admission. Interval outpatient clinic visits demonstrate that he is recovering well, including a return to baseline levels of endurance and function.
Laboratory Identification
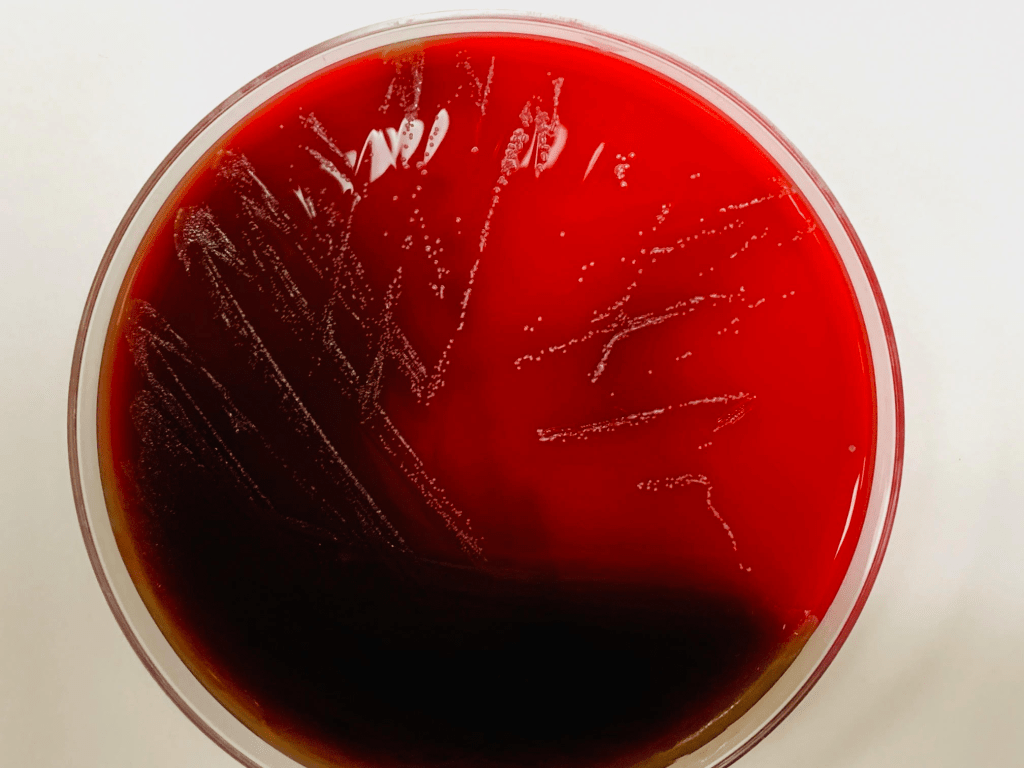
Gram stain of the positive blood cultures revealed pleomorphic gram variable rods which were arranged in clusters, pairs, short chains, and characteristic rosette patterns (Image 1 and inset). Pinpoint, opaque colonies were visible on blood and chocolate agars after 48-72 hours of incubation at 35°C in CO2 (Image 2). No growth was observed on MacConkey agar. The colonies were catalase-negative, and oxidase- and indole-positive. The recovered organism was definitively identified by MALDI-TOF MS as Cardiobacterium hominis.
Discussion
In 1962, four cases of infective endocarditis (IE) due to a Pasteurella-like organism belonging to CDC Group-IID were reported. Two years later, this group of organisms was reclassified as Cardiobacterium in recognition of their ability to cause endocarditis. Two species, Cardiobacterium hominis and Cardiobacterium valvarum, have been reported to cause IE, with the former being the etiological agent in a vast majority of cases.1 There is a strong association between C. hominis bacteremia and IE, as the organism is rarely recovered from blood cultures outside of this setting. Most cases of C. hominis endocarditis involve the aortic valve, particularly in the presence of pre-existing abnormalities or when a prosthetic valve is in place.2 C. hominis is a member of the normal flora of the nose and throat of ~70% of individuals (1), and endocarditis can be caused by periodontitis or dental procedures without prophylaxis.3
C. hominis is a member of the HACEK group of organisms which also include Haemophilus spp., Aggregatibacter spp., Eikenella corrodens, and Kingella kingae. HACEK organisms exhibit similar manifestations of disease, prognosis, and epidemiology. While over 80% of cases of IE are caused by Gram-positive bacteria (notably staphylococci and oral streptococci), Gram-negative IE is far less frequent, with a majority of cases caused by HACEK organisms (1-3% of all IE cases).4 In general, IE caused by HACEK organisms has an excellent prognosis, but delays in diagnosis and associated complications can lead to poorer outcomes.2 Susceptibility testing of C. hominis is difficult to perform due to its nutritional requirements. Most strains are susceptible to fluoroquinolones, rifampin, tetracycline, and beta-lactams. As beta-lactamase producing isolates have been reported, the current American Heart Association Guidelines recommend the use of a 4-6 week course of ceftriaxone for treatment of HACEK IE; fluoroquinolones may be used in cases where patients cannot tolerate cephalosporin therapy.5
Historically, prolonged blood culture incubation for the recovery of HACEK group organisms has been recommended due to their fastidious nature and slow growth rate. However, modern automated blood culture systems utilize enriched media which readily support their growth and facilitate recovery within a standard 5-day incubation period (average of 3.4 days incubation).6 Additional studies have demonstrated that prolonged incubation times do not significantly enhance the recovery of HACEK organisms and are of little clinical value.7 This case demonstrates many hallmarks of a characteristic description of a HACEK bacterial endocarditis: 1) the patient had a prosthetic valve as a pre-existing risk factor, 2) the subacute presentation caused a delay in recognition of an infectious etiology as contributing to his clinical decline, 3) C. hominis grew in less than 5 days in our automated blood culture system without prolonged incubation, 4) blood culture Gram stain findings were consistent with the MALDI identification of a HACEK group member, and 5) the patient was treated with ceftriaxone and with surgical intervention and has recovered successfully.
References
- Malani AN, Aronoff DM, Bradley SF, Kauffman CA.2006. Cardiobacterium hominis endocarditis: two cases and a review of the literature. European Journal of Clinical Microbiology and Infectious Diseases 25:587-595.
- Sharara SL, Tayyar R, Kanafani ZA, Kanj SS.2016. HACEK endocarditis: a review. Expert Review of Anti-infective Therapy 14:539-545.
- Steinberg JP, Burd EM. 2015. 238 – Other Gram-Negative and Gram-Variable Bacilli, p 2667-2683.e4. In Bennett JE, Dolin R, Blaser MJ (ed), Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases (Eighth Edition) doi:https://doi.org/10.1016/B978-1-4557-4801-3.00238-1. Elsevier, Philadelphia, PA.
- Revest M, Egmann G, Cattoir V, Tattevin P.2016. HACEK endocarditis: state-of-the-art. Expert Review of Anti-infective Therapy 14:523-530.
- Baddour Larry M, Wilson Walter R, Bayer Arnold S, Fowler Vance G, Tleyjeh Imad M, Rybak Michael J, Barsic B, Lockhart Peter B, Gewitz Michael H, Levison Matthew E, Bolger Ann F, Steckelberg James M, Baltimore Robert S, Fink Anne M, O’Gara P, Taubert Kathryn A.2015. Infective Endocarditis in Adults: Diagnosis, Antimicrobial Therapy, and Management of Complications. Circulation 132:1435-1486.
- Petti CA, Bhally HS, Weinstein MP, Joho K, Wakefield T, Reller LB, Carroll KC.2006. Utility of extended blood culture incubation for isolation of Haemophilus, Actinobacillus, Cardiobacterium, Eikenella, and Kingella organisms: a retrospective multicenter evaluation. Journal of clinical microbiology 44:257-259.
- Weinstein MP.2005. Emerging Data Indicating that Extended Incubation of Blood Cultures Has Little Clinical Value. Clinical Infectious Diseases 41:1681-1682.
-Francesca Lee, MD, is an associate professor in the Departments of Pathology and Internal Medicine (Infectious Diseases) at UT Southwestern Medical Center. She serves as Medical Director of the microbiology laboratory and pre-analytical services at Clements University Hospital.
-Julia Sweetnam, MLS(ASCP)CM has worked for six years as medical technologist in the microbiology laboratory at Clements University Hospital. She is interested in antimicrobial susceptibility testing and diagnostic bacteriology.

-Andrew Clark, PhD, D(ABMM) is an Assistant Professor at UT Southwestern Medical Center in the Department of Pathology, and Associate Director of the Clements University Hospital microbiology laboratory. He completed a CPEP-accredited postdoctoral fellowship in Medical and Public Health Microbiology at National Institutes of Health, and is interested in antimicrobial susceptibility and anaerobe pathophysiology.
