During the 2019-2020 residency interview season, I “courted” – no better way to describe those interactions over lunch–several potential co-residents, who were eager to know why I came to University of Chicago (NorthShore) for my residency. My answers and those of my fellow residents would help the candidates determine how high they should rank our program, so I enthusiastically recalled things I liked when I interviewed at NorthShore about a year earlier. I had also recently completed my first microbiology rotation in residency and I had enjoyed seeing all of those factors work synergistically to improve patient health outcomes through improved testing. So passionately, I shared how I fell in love with the physical structure of the department which has almost all the labs and offices one floor, the automation of the labs-especially the core and microbiology labs, the capability and regular expansion of its molecular laboratory, the people and of course, “the feel” about NorthShore.
With these experiences, I looked forward to my second microbiology in March 2020, where I would learn more about the diagnostics of various microorganisms–E. coli: Gram negative short stubby/broad shouldered rods vs. Pseudomonas aeruginosa, Gram negative long slender rods, etc. (Un)fortunately, March came, but the novel coronavirus (SARS-CoV-2) had other plans for my learning. Cases of Coronavirus disease 19 (COVID-19), caused SARS-CoV-2[1] were increasing rapidly in the US, so laboratories, including ours had rapidly implement testing. Rather than have morning rounds and other educational activities where the differential diagnoses of several clinically relevant microorganisms were discussed, we had virtual and in-person meetings discussing what to do about one virus. These continued and by the middle of March, we had become the only non-government lab in Illinois and second in the Midwest that had developed a clinical PCR test for SARS-CoV-2. I was excited to be part of that success, but more so, about learning how we achieved that as a team.
Our approach could be summarized using the Double diamond or 4D model of design process which consists of four phases: Discover, Define, Develop and Deliver (Figure 1).
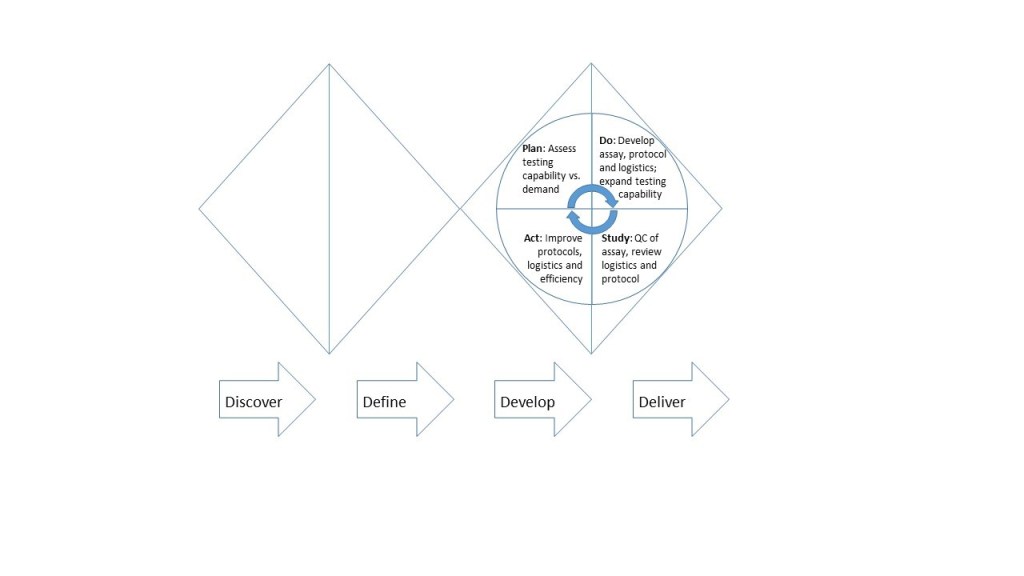
- In the discover phase, a phase of divergent thought [2] and exploration, we identified from events in China and other parts of the world as well as some other states in the US that the community we care for could potentially be affected by the COVID-19 outbreak.
- The next phase- define- is a convergent phase where the problem to be solved, as well as the resources available and resources needed to solve it are delineated [2]. As we transitioned from the discovery to define phases-and recalling the 2009 H1N1 influenza outbreak about 10 years ago- it became evident that an epidemic of a relatively fatal respiratory virus which we knew very little about was heading our way. As clinical laboratory professionals, our objective was to help identify members of the community who had been infected through testing so appropriate steps could be taken to sequester and care for them. Among our available resources was our molecular laboratory, but like most laboratories outside the Centers for Disease Control and Prevention, CDC we lacked the reagents, primers and authorization to run the test.
- Develop is the next phase in the process and this is a divergent phase where the team explores and refines potential solution to the issues and selects one[2]. This is often followed by the convergent deliver phase where one of the solutions from the develop phase is implemented. Feedbacks which are used for projects are also received during this phase[2]. But, the outbreak continued to evolve rapidly [3] with briskly increasing positivity rates[4] and some of the solutions we considered would require some time to be implemented and/or have long turnaround times. For instance, since we had a roust molecular laboratory, one option was to develop our assays and test in-house, while another was to send the samples to outside labs where they could be run. Running the tests in-house would have a shorter turnaround time and would be more efficient, which is extremely important considering the severity of COVID-19.
- Deliver is the last phase of the process. We decided to develop a SARS-CoV-2 RT-PCR test at our institution, but we also knew we needed to put logistics and protocols in-place to deliver our solution. For example, COVID-19 presents with flu-like symptoms but flu is common between December and March[5-7] so it would be impractical to expect to test all patients with flu-like symptoms – at least with the limited resources we had. In any case, it was clear that we would not have an ideal amount of time or information to develop and implement the perfect solution. As such, the revolving and fluid nature of the develop and deliver phases of our response is best depicted using the Plan Do Study Act (PDSA), an iterative model of quality improvement. As shown in Fig. 1, we developed and validated our assay, as well as developed an initial protocol for screening patients and logistics for patient-centered delivery in the “Do” step. Importantly, we also reviewed the effectiveness of these operations, and made necessary changes corresponding in the “Study” and “Act” steps respectively.
The prompt decision to implement in-house COVID-19 testing at NorthShore has proven to be the right one. To date we have tested 75,000 specimens and nearly 20,000 tests have been positive. Success which was possible because of the factors which made me come to NorthShore, amongst others. The LEAN, bright and capacious design of the department limits the innate barriers of hierarchical organizational structure; encouraging seamless horizontal and vertical intradepartmental consultation and collaboration as COVID-19 led us into uncharted territory. Also, having a molecular lab that regularly expands its capability made the decision to test in-house relatively easy. In addition, having an automated microbiology lab made it easier for staff to be flexible and deal with the various demands of testing for a new bug in a pandemic. And of course, the people at NorthShore who are ready to volunteer, take up new roles or change shifts to accommodate the demands of a rapidly evolving pandemic, stay in constant communications and provide feedback, and who make everything else at NorthShore work!
References
- https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200130-sitrep-10-ncov.pdf?sfvrsn=d0b2e480_2
- Council, Design. “Eleven lessons: Managing design in eleven global companies-desk research report.” Design Council (2007).
- https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/summary.html
- https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200324-sitrep-64-covid-19.pdf?sfvrsn=723b221e_2
- https://www.who.int/news-room/q-a-detail/q-a-coronaviruses#:~:text=symptoms
- https://www.cdc.gov/flu/symptoms/symptoms.htm
- https://www.cdc.gov/flu/about/season/flu-season.htm
- Christoff, Patricia. “Running PDSA cycles.” Current problems in pediatric and adolescent health care 48.8 (2018): 198-201.

–Adesola Akinyemi, M.D., MPH, is a first year anatomic and clinical pathology resident at University of Chicago (NorthShore). He is interested in most areas of pathology including surgical pathology, cytopathology and neuropathology -and is enjoying it all. He is also passionate about health outcomes improvement through systems thinking and design, and other aspects of healthcare management. Twitter: @AkinyemiDesola

-Erin McElvania, PhD, D(ABMM), is the Director of Clinical Microbiology NorthShore University Health System in Evanston, Illinois. Follow Dr. McElvania on twitter @E-McElvania.
