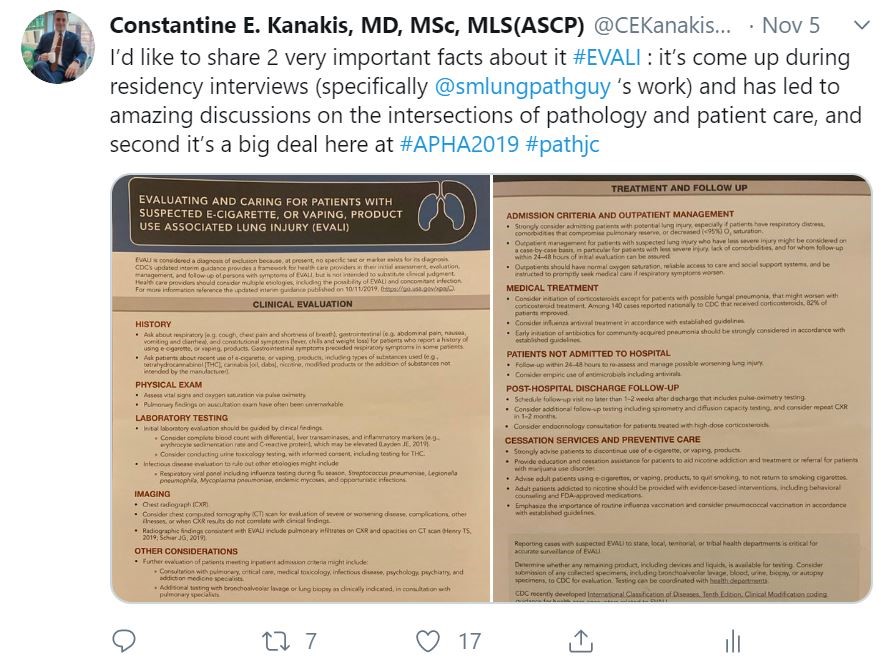
We are seeing an unprecedented amount of new targeted therapies for cancer treatment that are tied to diagnostic tests. Drug companies are heavily invested in ensuring the right patients get the right therapy. This is because it actually benefits pharma companies and patients. Patients get a very specific therapy that will likely improve their survival rate and improve their quality of life. By being selective and targeting only patient populations that are likely to respond based on the biology of their tumor, pharma companies show improvements over existing therapies which supports their request for FDA-approval.
With every pharma company tying their drug to specific rare biomarkers, broad molecular profiling such as NGS becomes more important than ever. We will never find the needle in the haystack if we don’t examine the entire stack. However, most cancer patient care occurs in the community where NGS testing is not usually offered locally. There are specific barriers to biomarker identification in the community setting. I will take the next few months to discuss specific barriers and how a lab might overcome these obstacles in order to increase patient access to precision medicine. Just as no barrier is identical between institutions, no solution will be one-size fits all. Feel free to cherry pick and modify solutions that you feel would address your local issues. Remember don’t let perfect be the enemy of the good. Small incremental improvements are impactful and generally require fewer resources than trying to revamp your entire process.
Here are the top 10 barriers that I’ve seen to biomarker testing in the community:
- High cost of testing.
- Long turnaround time for results.
- Limited tissue quantity.
- Preanalytical issues with tissue.
- Low biomarker testing rates.
- Lack of standardization in biomarker testing.
- Siloed disciplines.
- Low reimbursement.
- Lengthy complex reports.
- Lack of education on guidelines.
This month I will address the first two barriers that I commonly see with respect to biomarker testing. Molecular testing is expensive and turnaround time is often long. This was especially true for technology such as NGS. There are a few solutions to the high cost and long turnaround time for molecular testing that I’ve seen work well.
Solutions to costly molecular testing such as NGS:
- Insource NGS testing.
- Continue to send-out but renegotiate your contracts with reference laboratories to ensure pricing is as low as possible.
Let’s dig into the decision to insource NGS versus continuing to outsource testing. It’s easy for me to say insource the test and describe the benefits of doing so, but if your volume is low and you don’t have the facility or expertise, this solution is not likely to work for you. There is a new platform coming to market that claims to make it easier to insource NGS without extensive molecular expertise, however the company will need to provide data to support that claim. If they do show they can provide NGS testing with less expertise, then this could be a game changer for community labs looking to insource NGS testing.
The benefits of insourcing testing include decrease cost of providing biomarker testing, decreased turnaround time on testing, and local provider input into the test menu. Some of the things that we considered when deciding to insource NGS was the cost to perform NGS testing versus sending it out, volume of specimens to be tested, expertise required, facility requirements, ease of workflow, did available panels meet our clinician and guideline needs, and if there was a comprehensive pipeline available from the vendor. We found a solution that fit our needs in all of these buckets.
After determining that insourcing NGS was the right thing to do for our health system we had to secure funding for the project. We prepared a business case using reference laboratory cost avoidance. This is an example business case for a NGS project:
- Imagine that you currently send out 200 NGS tests per year for the same panel.
- This reference lab NGS panel costs $3500 per sample.
- You calculate that by insourcing the testing you can perform the test for $600 per sample (fully loaded with tech time, repeat rate, control cost, validation cost, QA cost, overhead).
- This would save the health system $580,000 per year [($3500-$600)X(200 tests)].
- Pretend the instrumentation required to perform the test in house cost $300,000.
Even the first year, the project could save the health system $280,000 ($580,000-$300,000). Subsequent years would be even more favorable. Showing a favorable return on investment (usually within a 5 year time period) would make it easy for the C-suite to approve insourcing this project.
Obviously money is not the only deciding factor when insourcing testing. I have to be able to perform a test cheaper, faster, and at least as well as the reference laboratory if not better or I will not insource a test.
There are a variety of reasons that you may not want to insource NGS testing. You may not have the expertise, facility, or volume for it to make sense to insource the testing. Are you stuck paying whatever your reference lab is charging you because you can’t in source the test? No.
If you have not negotiated the pricing and billing structure of your molecular pathology reference lab recently, it may be time to take a look around. Here are a few things to consider getting better pricing on send out testing:
- Renegotiate. You can try to renegotiate with your current reference lab to decrease your contracted price.
- Shop around. The molecular pathology lab market is growing. With competition comes better pricing.
- Increase volume. You could try to standardize which lab your physicians are using to increase the volume to your reference lab. Most reference lab contracts are negotiated based on volume. So if you can increase the volume, it is likely that you can decrease the price you’re paying.
- Direct billing. It is worth addressing who is billing the patient (and who has the highest risk of being stuck with the bill if the testing is not covered). Many molecular pathology labs now directly bill the patient (as long as the patient was not an inpatient within the last 14 days). You may want to explore this option when negotiating contracts.
- Insurance coverage. You should also consider whether the test offered by the lab is approved for coverage by your most common payers.
- Out of pocket costs. Many labs now have maximum out of pocket costs to patients that are reasonable. This ensures your patients are stuck with large bills.
Whether you decide to insource or continue to outsource NGS testing, there are options that could decrease the cost and turnaround time for biomarker testing.

-Tabetha Sundin, PhD, HCLD (ABB), MB (ASCP)CM, has over 10 years of laboratory experience in clinical molecular diagnostics including oncology, genetics, and infectious diseases. She is the Scientific Director of Molecular Diagnostics and Serology at Sentara Healthcare. Dr. Sundin holds appointments as Adjunct Associate Professor at Old Dominion University and Assistant Professor at Eastern Virginia Medical School and is involved with numerous efforts to support the molecular diagnostics field.