Molecular diagnostics tests come in many forms, but one of the simplest assays is a fragment based assay. The principle of such an assay is to perform polymerase chain reaction (PCR) on a segment of DNA. If there is a mutation, the PCR fragments will be different in size. Notably, this method is good for detecting mutations that cause the insertion or deletion of multiple nucleotides. This type of assay is not suitable for single base pair changes or small insertion/ deletions.
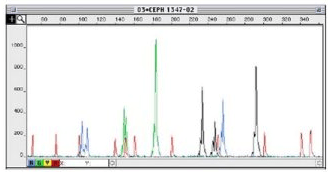
The fragment size is analyzed by labeling the PCR products with a fluorescent dye and then running them through a Sanger capillary sequencer. The fragments will be separated based on size and ideally give clean peaks with low background (Figure 1).
One common application of this assay type is to detect FLT3 internal tandem duplications (ITD). FLT3, Fms Related Tyrosine Kinase 3, is a tyrosine kinase growth factor receptor for FTL3-ligand, and regulates hematopoiesis. Mutations in FLT3 are found in 1/3 of Acute Myeloid Leukemia cases and confer a worse prognosis. FLT3 mutations lead to ligand-independent activation by either disrupting the auto-ihibitory loop of the juxtamembraneous domain through an ITD mutation or by an activating point mutation in the tyrosine kinase domain (TKD) (Figure 2).
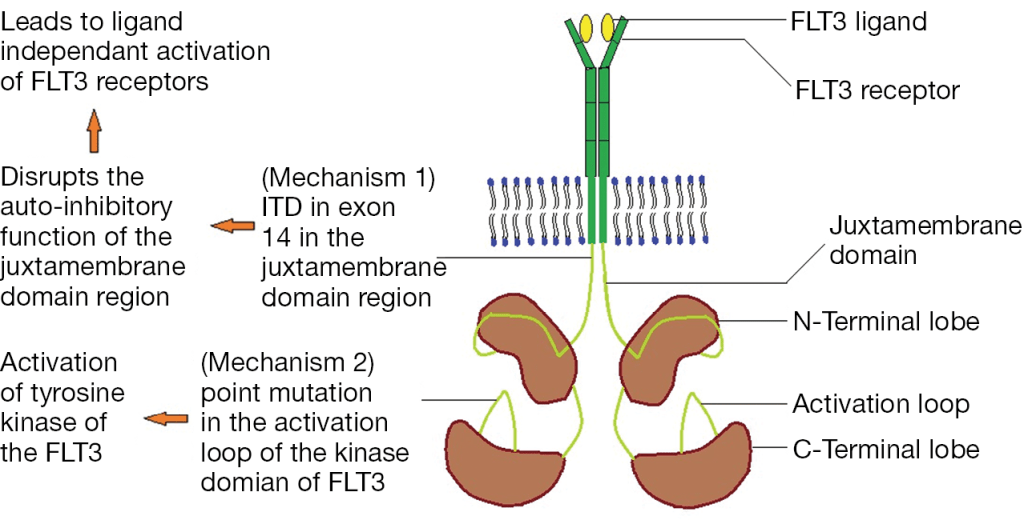
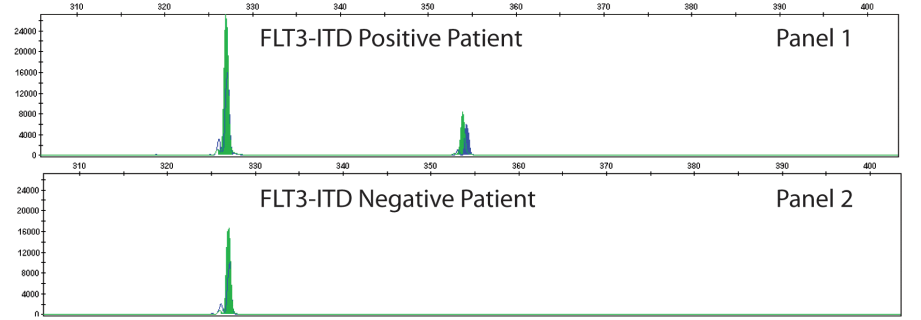
The type of FLT3 mutation is also important as there are tyrosine kinase inhibitors (TKI’s) that are being investigated for use in FTL3+ cases. Type I inhibitors bind FLT3 in the active conformation either in the ATP binding pocket or at the activation loop; these inhibitors are useful for both ITD and TKD mutations. However, Type II inhibitors bind inactive FLT3 near the ATP binding domain, so they affect ITD but not TKD mutations.As the site of ITDs is consistently in exons 14 and 15 of FLT3, primers flanking this region were designed to detect any mutations in this area (Figure 3). As some artifacts can arise from the PCR process and create false positive peaks, a green primer labels PCR products from one direction and a blue primer labels PCR fragments from the other direction, therefore enhancing specificity (Figure 4). A wild type (WT) sequence will thus be 327bp in either direction.
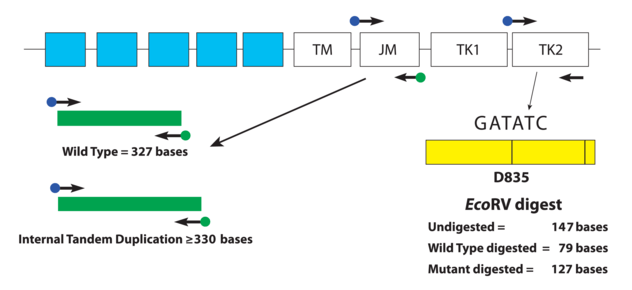
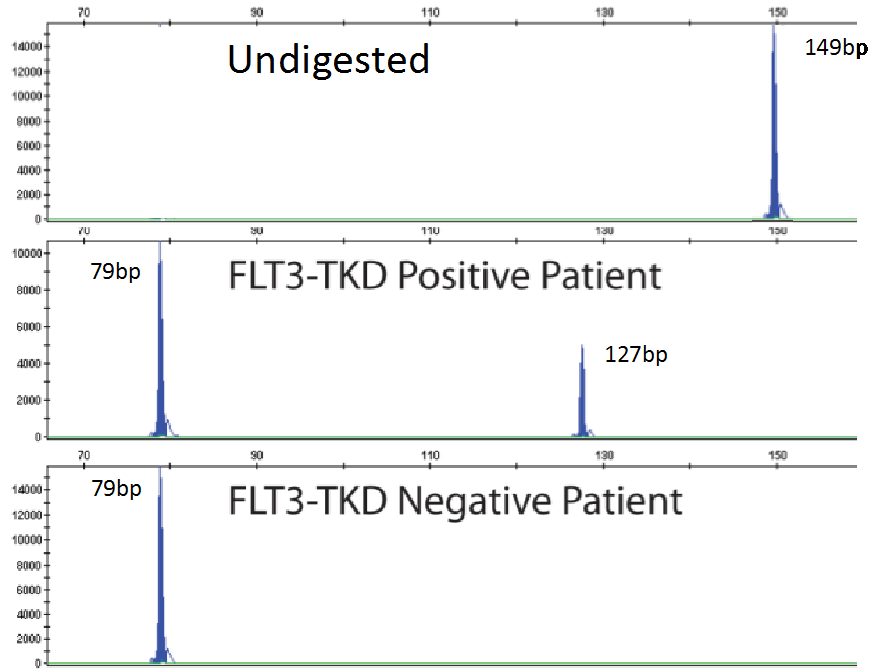
As mentioned previously, fragment analysis is not suited to detecting point mutations as would be found for TKDs. However, the FLT3 assay has overcome this issue. Investigators determined that the TKD point mutation at codon D835 disrupts the endonuclease recognition site of the enzyme ecoRV (Figure 3). Customized primers again produce a unique PCR fragment (149bp long), which when digested with ecoRV will produce a 79bp fragment in wild type FLT3. If a FLT3-TKD mutation is present the ecoRV will not cleave the fragment at this location, but another ecoRV cleavage site (right side of yellow box) will create a 127bp fragment (Figure 5). Without this second cleavage site, an enzyme failure could be interpreted as a mutation. Thus, the enzyme, ecoRV, must be active and only functional at a single site to produce a TKD mutation.
References
- Daver Naval, Schlenk RF, Russell NH, and Levis MJ. Targeting FLT3 mutations in AML: review of current knowledge and evidence. Leukemia 2019; 33:299-312.
- https://invivoscribe.com/products/companion-diagnostics-cdx/. Last accessed December 8th, 2019.
- Pawar R, Bali OPS, Malhotra BK, Lamba G. Recent advances and novel agents for FLT3 mutated acute myeloid leukemia. Stem Cell Invest. 2014; 1(3). doi: 10.3978/j.issn.2306-9759.2014.03.03

-Jeff SoRelle, MD is a Chief Resident of Pathology at the University of Texas Southwestern Medical Center in Dallas, TX. His clinical research interests include understanding how the lab intersects with transgender healthcare and improving genetic variant interpretation.
