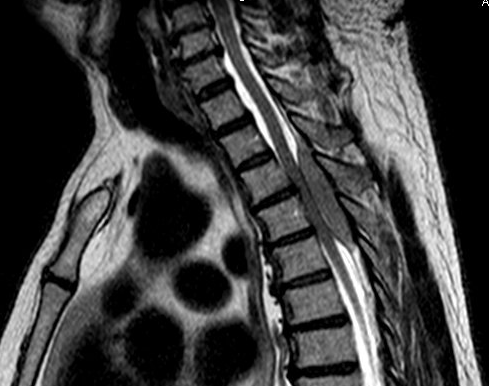
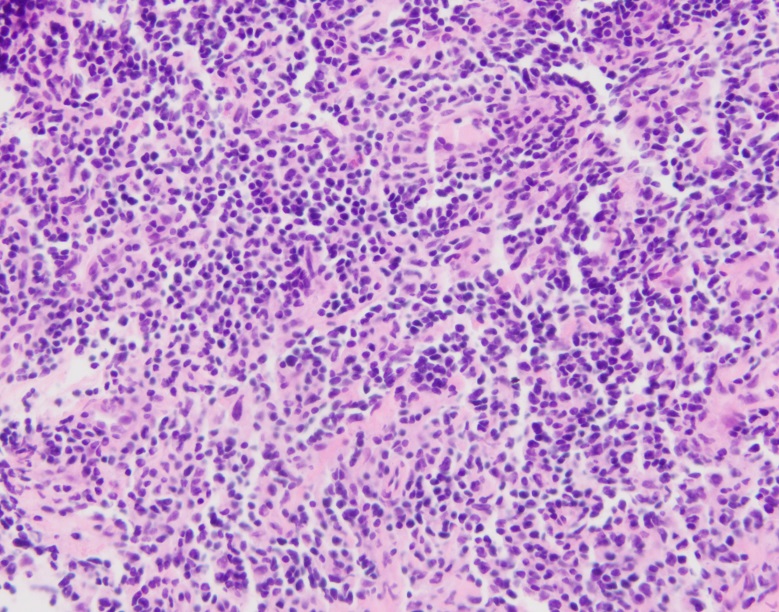
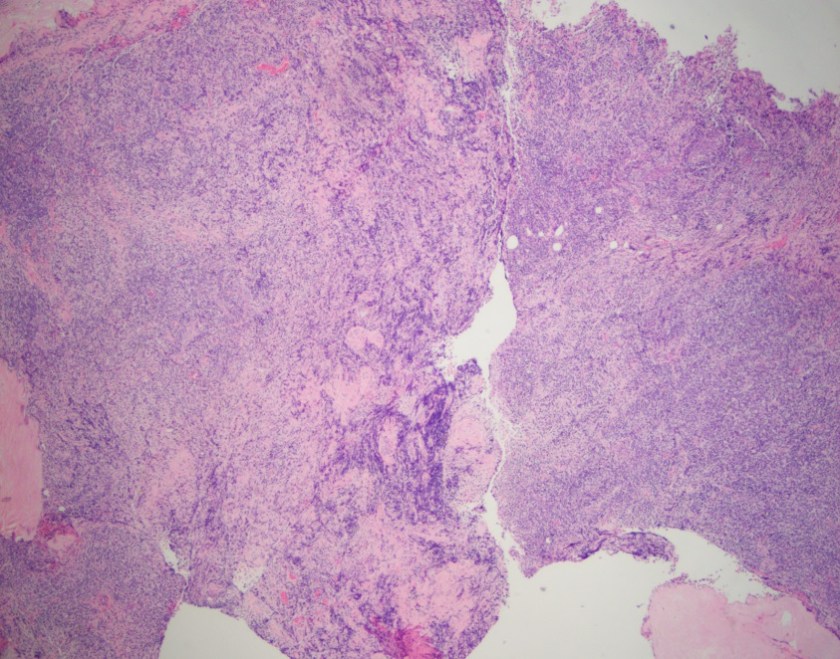
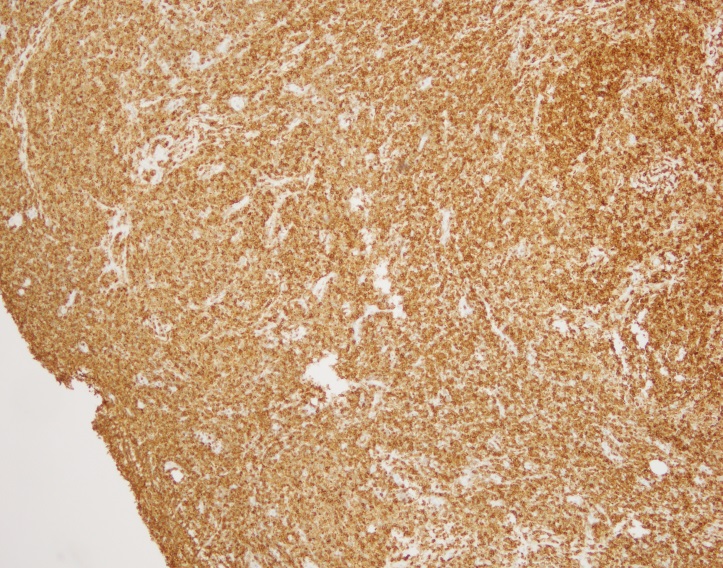
History plays a significant part in the development of any person; we are changed and altered by big historical events that take place during our life time. Understanding history is therefore an essential aspect of understanding people, communities, cultures, and generations.
The oldest generation living today is the GI Generation. This generation was born between circa 1901-1926 and have gone through significant changes in life and work environments during their lifetimes. The term GI Generation stems from the fact that a lot of soldiers from both WWI and WWII came from this generation. This generation came of age during the First World War and the Great Depression and most grew up without electricity, refrigerators, and credit cards.
The Traditionalist Generation was born around 1927-1945, so during the Great Depression and at the end of WWII. This is the era of pre-feminism, so women generally stayed at home to raise children. If women had jobs, it was typically until they were married and in professions such as secretary, nurse, and teacher.
This started to change during the next generation, the Baby Boomers, who were born between 1946 and 1964. The timeframe for this generation is so large that there are essentially two main groups: the revolutionaries from the ‘60s and ‘70s and the yuppies of the ‘70s and ‘80s. Women began working outside the home in record numbers, which created double-income households. Divorce also became more accepted and people starting buying things on credit.
The following generation is Generation X, who are born circa 1965-1980. Because most of their parents both worked, this generation is known as the “latch-key kids”, because they would walk home after school themselves as both their parents were working or divorced. This generation experienced the transition to digital knowledge, but remembers a time without computers.
The Millennial Generation, also known as Generation Y, was born around 1981-2000. This generation grew up in a world of technology and they have experiences some significant technological advances, which typically are very natural to them. They also grew up with enormous academic pressure and also the notion that you might not be save at school due to school shootings.
The newest generation is Generation Z who are born after 2001. People born during this time have never known a world without cell phones or computer and they are very technological savvy. Growing up during the great recession of the late 2000s, Z’ers feel unsettled and a level of professional insecurity.
The events mentioned above are all focused on events that took place in the United States of America, with some worldwide events included. To understand generations from other countries, it is important to learn about important historical events that occurred, while there are also some events that overlap. For instance, internet and cell phone are more widely available worldwide and there might be some similarities across nations in terms of the effect on generational understanding.

-Lotte Mulder earned her Master’s of Education from the Harvard Graduate School of Education in 2013, where she focused on Leadership and Group Development. She’s currently working toward a PhD in Organizational Leadership. At ASCP, Lotte designs and facilitates the ASCP Leadership Institute, an online leadership certificate program. She has also built ASCP’s first patient ambassador program, called Patient Champions, which leverages patient stories as they relate to the value of the lab.
The GI generation experienced events that impacted their assertive characteristics. If you know someone in this generation, they probably worked until they couldn’t work anymore instead of retiring. This work ethic comes from growing up during the deprivation of the Great Depression and are often referred to as the “Greatest Generation.” This term was coined by the NBC Nightly News anchor, Tom Brokaw in his book by the same name.
The Traditionalist generation are, well, traditional. The value old-time morals, safety, security and may try your patience, especially in the work place. They are still working and act as the historians of the organization and/or the family because they have been there for a long time. You still might see them serve on Board of Directors and are Presidents because of their organizational knowledge and expertise. They are also known as the Silent Generation for an interesting reason. It was this generation that coined the phrase, “Children are to be seen, and not heard!”
Did you know there are two groups of Baby Boomers? The first group was born between 1946 and 1964. They are often called the “Leading-edge Boomers.” Those born between 1955 and 1964 are often called the “Shadow Boomers or Generation Jones.” The Baby Boomers are the largest generation in the US today, but they are slowly overpowered by the Millennial Generation. The have a team-oriented attitude and take their self-worth from their job. They are driven and optimistic and are often willing to learn how to use technology, but it takes a process as it doesn’t come as natural to them as to younger generations.
The Generation X are often referred to as the “middle child.” This generation is street smart because most grew up in homes where both parents worked or were divorced. They started school without computers, but are experienced with them. They change careers often and are independent, flexible, and can easily adapt to new circumstances. They have an entrepreneurial spirit.
The Millennial Generation is our fastest growing generation in the U.S. workforce. They are the most diverse and are also known as the “Echo Boomers, Millenials, or Generation Y. Millenials understand the world of technology and it comes natural to them. They are resilient, optimistic, and creative because they experienced enormous academic pressure. They are very focused on professional development and to learn and improve what they do.
Generation Z is just starting to enter the workforce and they are independent, open-minded, and determined. They also have an entrepreneurial spirit, like Generation X, and they are loyal and compassionate. This emerging generation will be our new teachers because their minds work in so many directions because of their technology skills and aptitude.
It is easy to see how working with multiple generations in one department offers a full range of experiences, work styles, ideas, as well as, challenges. How can you improve the generational diversity of your personal or professional life?

-Catherine Stakenas, MA, is the Senior Director of Organizational Leadership and Development and Performance Management at ASCP. She is certified in the use and interpretation of 28 self-assessment instruments and has designed and taught masters and doctoral level students.