A 74 year old male patient with an extensive cardiac history initially presented to the ER with black stool, warranting a CT scan, upper endoscopy, and colonoscopy, identifying a large, obstructive mass in the colon, smaller, yet unresectable polyps, and subcentimeter liver lesions and lung nodules. The colonic mass was biopsied, consistent with adenocarcinoma; however, the liver lesions were too small to characterize. One month after the onset of symptoms, a right hemicolectomy was performed, and the pathology was signed out as moderately differentiated adenocarcinoma, microsatellite stable, with evidence of lymphovascular and perineural invasion, placing the patient’s stage at IIA (pT3, pN0, cM0). Through shared decision-making, the medical oncologist and patient elected for surveillance due to multiple comorbidities. Forgoing adjuvant therapy, the patient was discharged to physical therapy/rehabilitation. The patient returned for imaging 4 months after his hemicolectomy, demonstrating an enlargement in one of the liver lesions, but then, the patient was lost to follow-up for 20 months.
The patient reestablished care and surveillance imaging, which demonstrated a hypodense liver lesion (in a background of poorly visualized subcentimeter liver lesions), a nonocclusive thrombus in the right portal vein, a heterogenous enhancement of the left portal vein (suggestive of an underlying tumor thrombus), and an 8 cm heterogenous right adrenal mass. Based on the most recent CT scan, the differential diagnoses of the adrenal mass include metastatic disease or a primary adrenal lesion including adrenal cortical carcinoma or pheochromocytoma (for which biochemical analysis should be performed before attempting a biopsy). Extensive peritoneal lymphadenopathy was visualized as well. The area of the right hemicolectomy, however, did not show evidence of recurrence. After biochemical evaluation for metanephrines ruled out a pheochromocytoma, the patient underwent a CT scan-guided adrenal FNA and core biopsy.
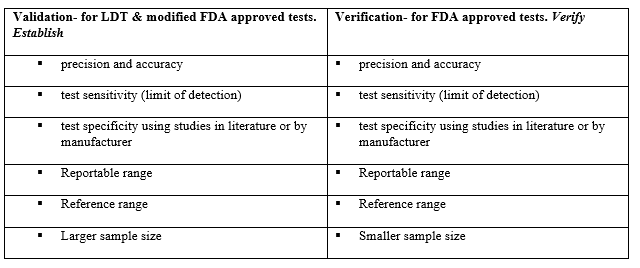
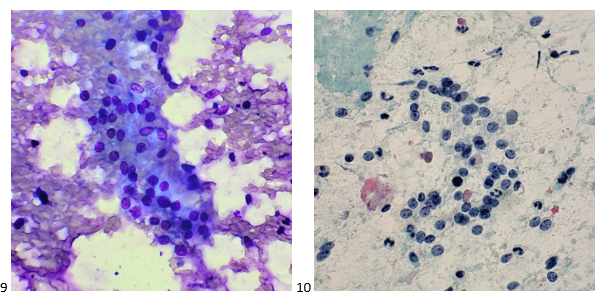
The Diff-Quik smear assessed at the time of biopsy revealed a highly cellular specimen, some cells with bare nuclei, enlarged nuclei, and some pseudoglandular structures.

Telepathology confirmed an adequate sample of tumor cells present, and core biopsies were obtained.
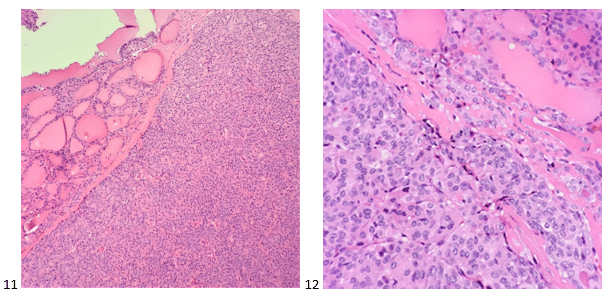
The following morning, the pap-stained smears and H&E cell block sections were screened. The cells appeared polygonal with a high N/C ratio and prominent macronucleoli. Cell arrangements formed thickened trabeculae. However, the cytoplasm is more granular than the lipid-rich cytoplasm seen in an adrenal cortical carcinoma. The H&E cell block sections depicted a beautiful trabecular pattern with endothelial cells wrapping the periphery.

The preliminary morphology was interpreted as carcinoma, and both cytotechnologist (or cytologist, as we now prefer to be called) and pathologist suggesting features of adrenal cortical carcinoma; however, the IHC markers proved otherwise!

Other differential diagnoses considered renal cell carcinoma and pheochromocytoma (to be safe). The IHC profile ruled out adrenal cortical carcinoma as the cells of interest were negative for inhibin, calretinin, and Melan A. Negative PAX-8, EMA, AE1/AE3, and vimentin staining ruled out renal cell carcinoma, and negative chromogranin, synaptophysin, GATA-3, vimentin, and S100 staining enabled us to safely say that a pheochromocytoma was out of the equation as well. Positive staining for HepPar1, arginase, pCEA (canalicular pattern), and CAM5.2 supported the unlikely diagnosis of metastatic hepatocellular carcinoma (HCC).
This diagnosis placed the patient at Stage IV HCC. It came to light that the patient has a remote history of hepatitis and a high-risk history of drinking, contributing to a poor prognosis. Due to the patient’s condition, they held off on HCV antiviral therapy and decided to observing viral load through regular blood work. The patient and clinician discussed the risks and benefits along with alternatives of systemic therapy, as his multiple comorbidities still pose a significant risk. Immunotherapy was determined to be the best option to delay the progression of his cancer and maintain quality of life.

-Taryn Waraksa, MS, SCT(ASCP)CM, CT(IAC), has worked as a cytotechnologist at Fox Chase Cancer Center, in Philadelphia, Pennsylvania, since earning her master’s degree from Thomas Jefferson University in 2014. She is an ASCP board-certified Specialist in Cytotechnology with an additional certification by the International Academy of Cytology (IAC). She is also a 2020 ASCP 40 Under Forty Honoree.