For patients with gender dysphoria, the Endocrine Society has endorsed the use of hormone therapy to promote secondary sexual characteristics of the desired gender. These guidelines were first established in 2007 and revised last year, and gave the first evidence guided recommendations for clinicians treating transgender patients.
For transgender males, testosterone by itself is prescribed as an injectable oil-based solution. These doses are given as intramuscular injections- usually into the thigh. If that’s too painful, subcutaneous injections have been shown to have similar efficacy. The doses given to transgender males is much higher (50-100mg/ injection) than that given to men with testosterone deficiency (30-50 mg/ injection). Primarily because the men have more testosterone to start with. Also, whereas topical testosterone gel may be sufficient for men with “low T,” it doesn’t seem to provide enough testosterone to transgender males and is quite expensive, so it is generally not used.
Upon starting testosterone injections, the frequency of injections is every one to two weeks. However, the onset of physical secondary sexual characteristics takes 3-6 months to begin. After about 3 years, most of the changes to occur will have manifested. These physical changes are outlined in the table below. You’ll notice how certain traits like cessation of menses and fat redistribution start within the first 6 months whereas muscle growth and voice change take effect after 6 months. Also, the time certain effects take maximal effect varies; the voice doesn’t deepen further after 2 years, but hair growth continues to increase through 5 years.
| Physical Effect | Begins | Maximal Effect |
| Facial/body hair growth | 6-12 mo | 4-5y |
| Skin oiliness/acne | 1-6mo | 1-2y |
| Scalp hair loss | 6-12 mo | – |
| Increased muscle mass | 6-12 mo | 2-5y |
| Fat redistribution | 1-6mo | 2-5y |
| Cessation of menses | 1-6mo | – |
| Deepening of voice | 6-12 mo | 1-2y |
Table 1. Timeframe of physical traits that manifest in transgender males while taking testosterone hormone therapy. Based on Hembree et al. 2017 (1).
Just as hormone therapy induces physical manifestations of secondary sexual characteristics for transgender men, we would suspect that internal aspects of physiology are affected too. Values measured by the laboratory provide meaningful insight into how our body and its different organ systems are functioning. Accordingly, the Endocrine Society also recommended laboratory monitoring of transgender patients starting hormone therapy.
- Measure Testosterone and hemogoblin/ hematocrit every 3 months for the 1st year, then 1-2x/ year afterwards.
- Monitor Lipids at regular intervals
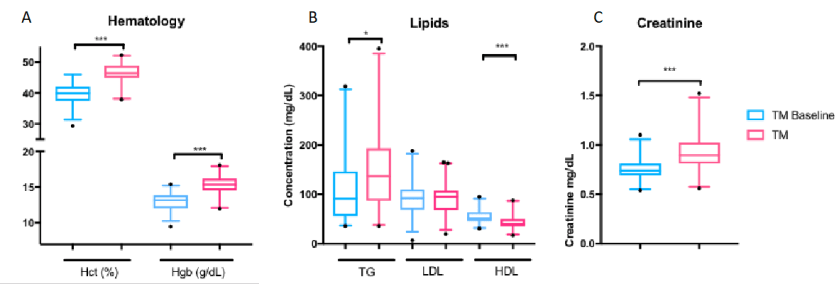
Previous studies have monitoring these lab values found consistent increases in hemoglobin and hematocrit (2,3). This is due to the stimulation of erythropoiesis by testosterone (4). While excessive testosterone could lead to polycythemia (excessive RBCs in the blood), it is not a commonly described complication in transgender patients. Some summary results from our study for hemoglobin and hematocrit are shown in Figure 1A, which shows a clear shift in levels.
However, reports on lipids have been varied LDL and triglyceride changes (2,3). The only consistent finding was that HDL decreased in transgender males taking testosterone (2,3). In our study, we found triglycerides were increased with decreased HDL (Figure 1B). The take-away is that because cardiovascular cut-offs are based on risk and not a reference range, patients and clinicians will have to be aware of these possible metabolic changes.
Creatinine, when it was checked, increases for transgender males (5). We found creatinine was strongly increased in our study to become similar to baseline creatinine in transgender women before taking hormone therapy (Figure 1C). This topic as it relates to glomerular filtration rate is very complex and will be discussed further in a future post.
To illustrate lab value changes in transgender men, I’ll direct you to data that I found in a large study of over 300 transgender patients including about 80 transgender men. The completed manuscript is not currently available but will be printed soon:
However, this does not mean Cisgender male reference intervals are adequate for transgender men. This topic needs further exploration and ideally a prospective trial to be performed in a controlled manner. A double-blind study would not be possible as it would be unethical to perform.
References
- Hembree WC, Cohen-Kettenis PT, Gooren L, Hannema SE, Meyer WJ, Murad MH, et al. Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society* Clinical Practice Guideline. J Clin Endocrinol Metab. 2017
- Wierkx K, et al. Cross-Sex Hormone Therapy in Trans Persons is Safe and Effective at Short-Time Follow-Up: Results from the European Network for the Investigation of Gender Incongruence. J Sex Med, 2014. 11(8):1999-2011.
- Mueller A, Kiesswetter F, Binder H, Beckmann MW, Dittrich R. Longer-term administration of testosterone undecanoate every 3 months for testosterone supplementation in female-to-male transsexuals. J Clin Endocrinol Metab. 2007
- Paller CJ, Shiels MS, Rohrmann S, Menke A, Rifai N, Nelson WG, et al. Association Between Sex Steroid Hormones and Hematocrit in a Nationally Representative Sample of Men. J Androl. 2012 33(6): 1332-1341.
- Fernandez JD, Tannock LR. Metabolic Effects of Hormone Therapy in Transgender Patients. Endocr Pract. 2016;22:383–8.

-Jeff SoRelle, MD is a Molecular Genetic Pathology fellow at the University of Texas Southwestern Medical Center in Dallas, TX. His clinical research interests include understanding how the lab intersects with transgender healthcare and advancing quality in molecular diagnostics.


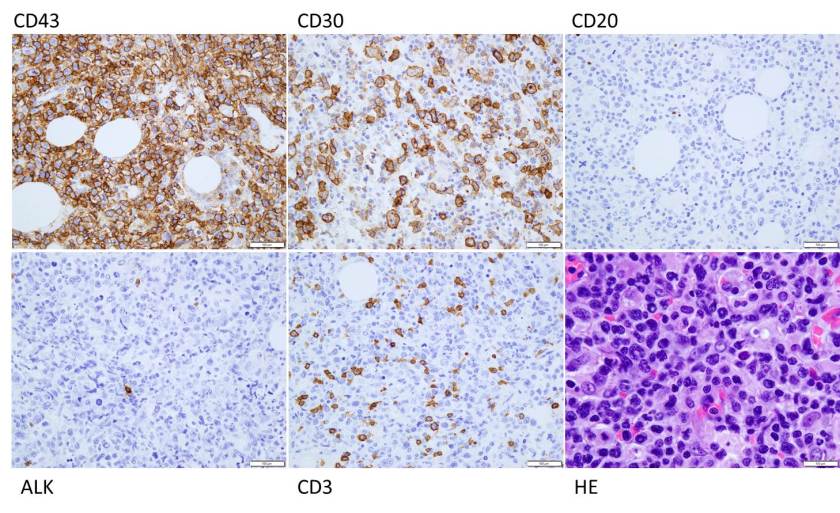


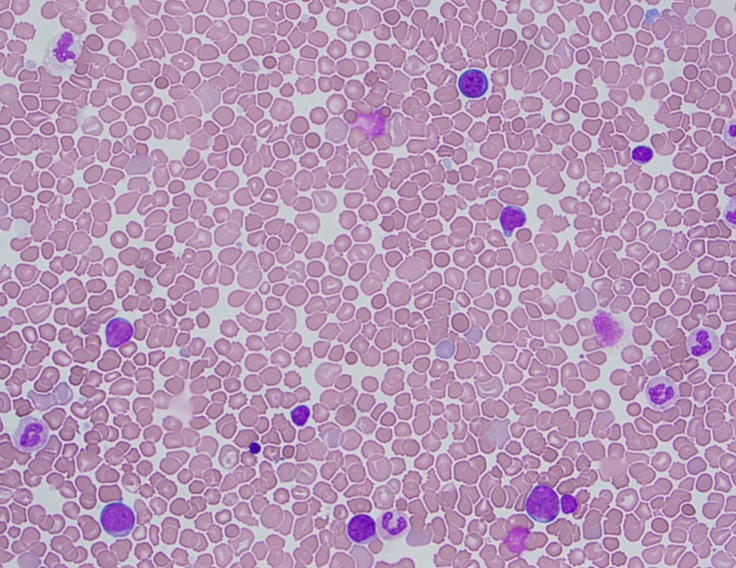
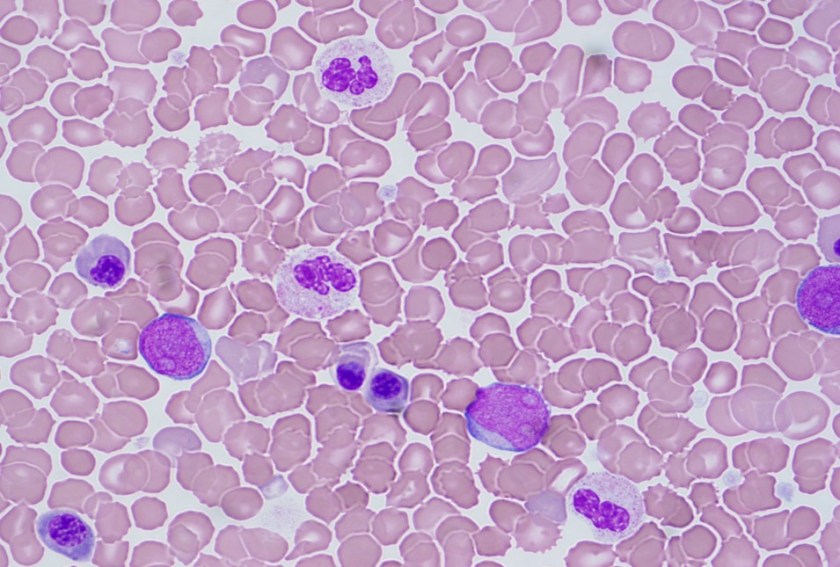
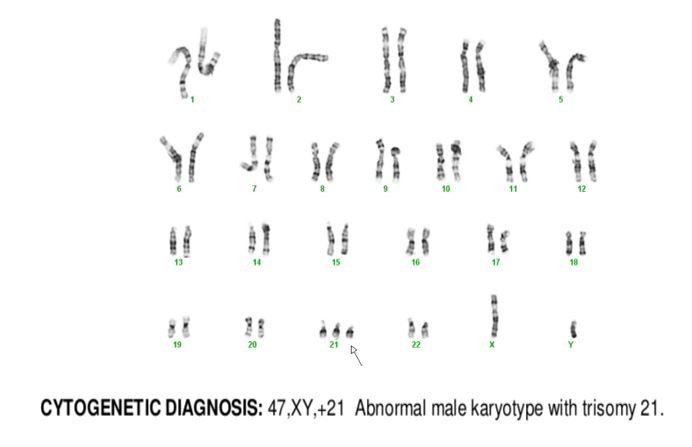

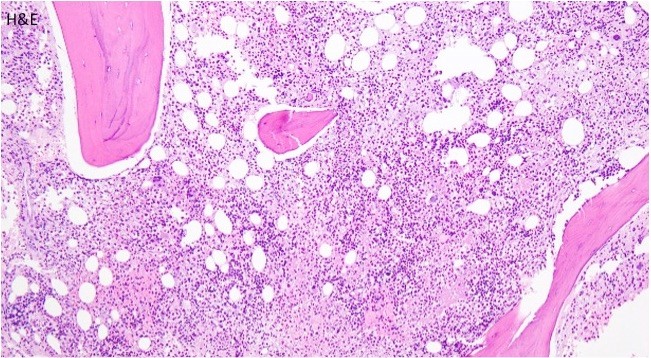
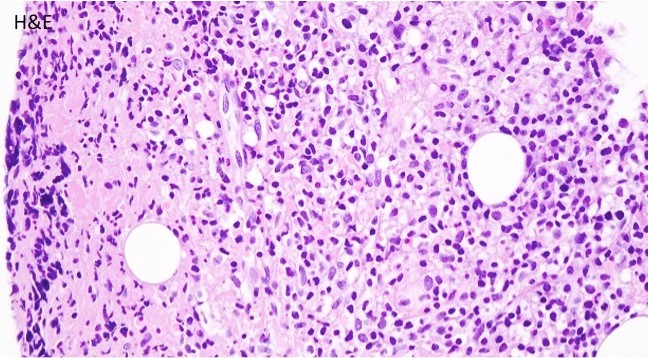
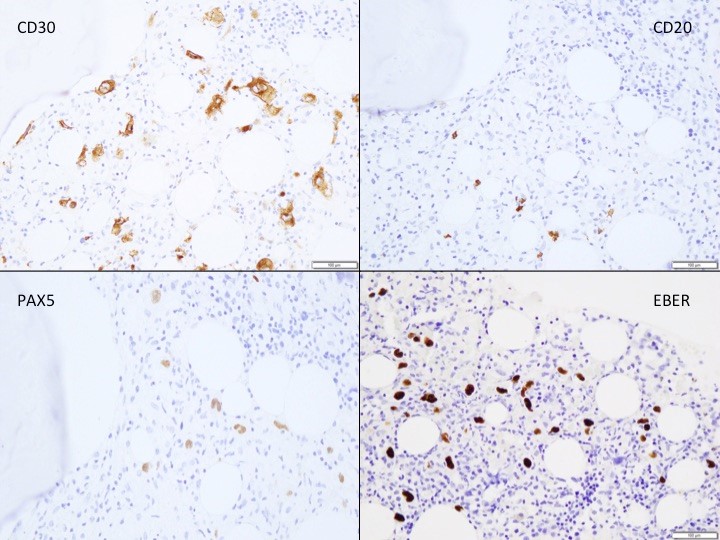
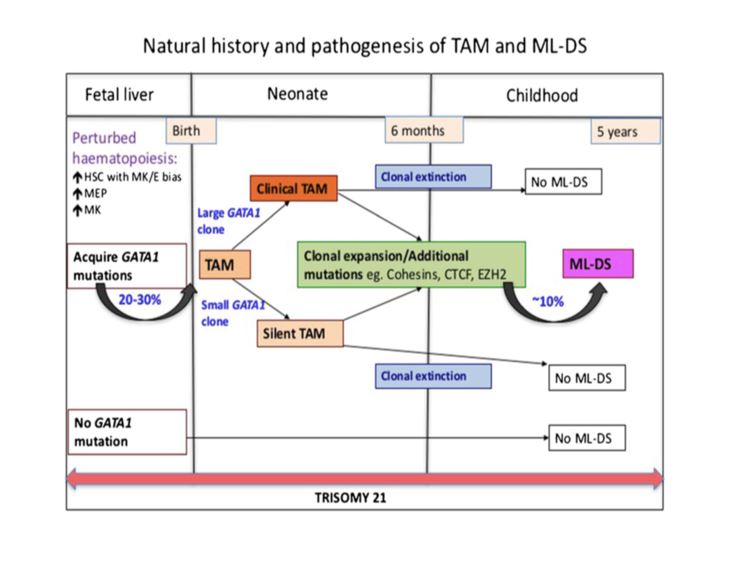 TAM has a high rate of spontaneous remission and typically resolves spontaneously in 90% of patients over several weeks to 6 months. This coincides with extinction of the GATA1 clone. However, in around 10% of these patients, myeloid leukemia of Down syndrome (ML-DS) develops within 5 years of the initial presentation. Additional mutations in cohesion component genes and epigenetic regulators occur in these patients that result in clonal expansion and non-transient leukemia. 2 Children who develop ML-DS generally have a good response to chemotherapy and a have a better prognosis than children without Down syndrome who develop AML.
TAM has a high rate of spontaneous remission and typically resolves spontaneously in 90% of patients over several weeks to 6 months. This coincides with extinction of the GATA1 clone. However, in around 10% of these patients, myeloid leukemia of Down syndrome (ML-DS) develops within 5 years of the initial presentation. Additional mutations in cohesion component genes and epigenetic regulators occur in these patients that result in clonal expansion and non-transient leukemia. 2 Children who develop ML-DS generally have a good response to chemotherapy and a have a better prognosis than children without Down syndrome who develop AML.