In 2006, an international group of human rights experts assembled in Yogyakarta, Indonesia to address patterns of discrimination and abuse of individuals related to sexual orientation and gender identity. This document, The Yogyakarta Principles: Principles on the application of
international human rights law in relation to sexual orientation and gender identity, has been widely accepted as setting the standards for fundamental human rights for all, with specific attention to sexual orientation and gender identify. It is worth studying, as it articulates rights in many domains of everyday life. Of direct relevance to our Profession are Principle #12, The Right to Work; Principle number #17, The Right to the Highest Attainable Standard of Health; and Principle #18, Protection from Medical Abuses.1 It is the goal to “Adopt the policies, and programmes of education and training, necessary to enable persons working in the healthcare sector to deliver the highest attainable standard of healthcare to all persons, with full respect for each person’s sexual orientation and gender identity.”
Because the medical laboratory provides critical data for patient management, laboratory professionals and pathologists must be able to evaluate laboratory and biopsy results wisely and appropriately. Yet we often lack fundamental and essential information necessary to support optimal and personalized care for patients on cross-sex hormones.
The number of individuals who self-identify as transgender has risen significantly in the past decade. Transgender people face discrimination, harassment, abuse, and denial of legal rights. They often feel unsafe, and a high proportion face bullying at school or at work. The Centers for Disease Control (CDC) estimates that about 2% of high school students in the U.S. identify as transgender; among them, 35% attempt suicide. Transgender adults are twice as likely of being homeless, four times more likely to live in poverty, four times as likely as being HIV-infected, and twice as likely to be unemployed compared to the general population.2 Individuals without access to appropriate care may purchase hormones from unreliable sources, so that the dose, drug contents, and potential side-effects are poorly controlled or even toxic.
Healthcare systems often fail the transgender community. First, given the high rate of poverty, unemployment, and homelessness, access to basic health services is not available for many individuals. Second, even in those healthcare institutions that serve the uninsured, appropriate services are often lacking, including the absence of knowledgeable providers and the lack of cultural competency in the institution. The few academic hospitals with services and clinics oriented to serving transgender patients struggle to provide optimal care, because there are important gaps in knowledge regarding how best to care for transgender patients. Many health care clinics and professionals lack training in asking all patients “What pronouns do you prefer to use in referring to yourself?” This is a straightforward way to acknowledge gender diversity and sets the first stage of a potentially trusting relationship.
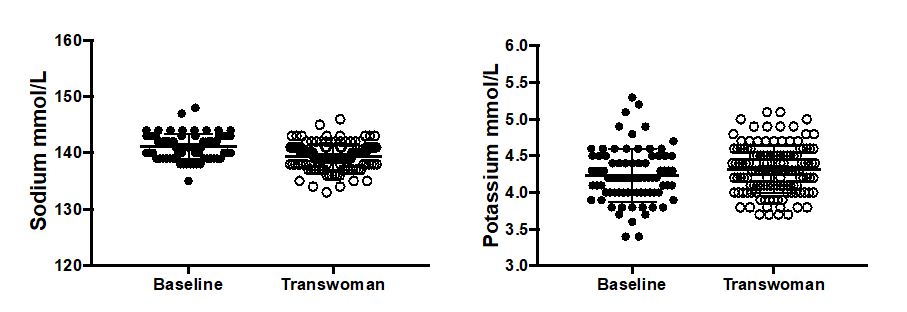
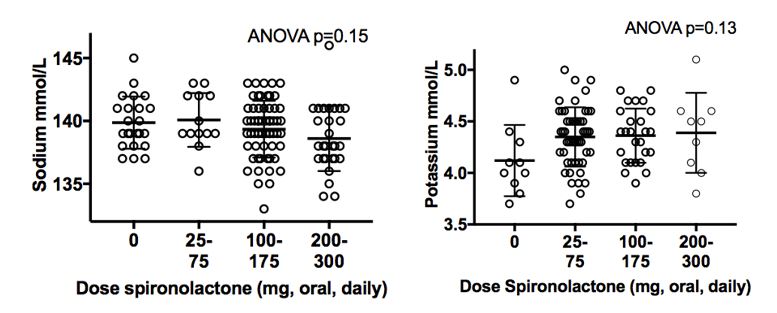
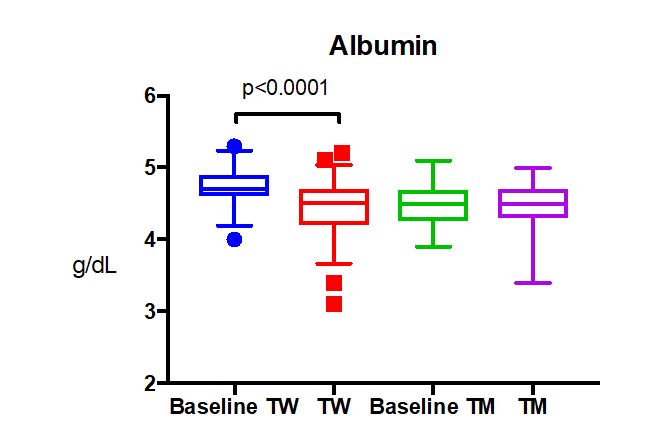
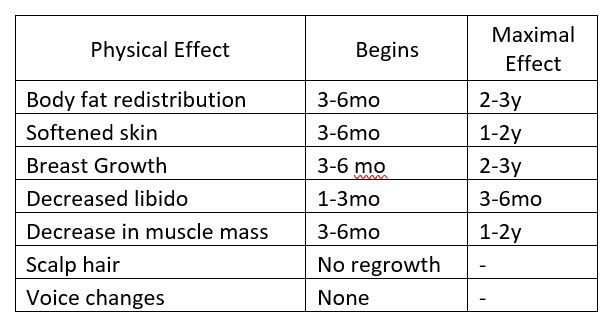
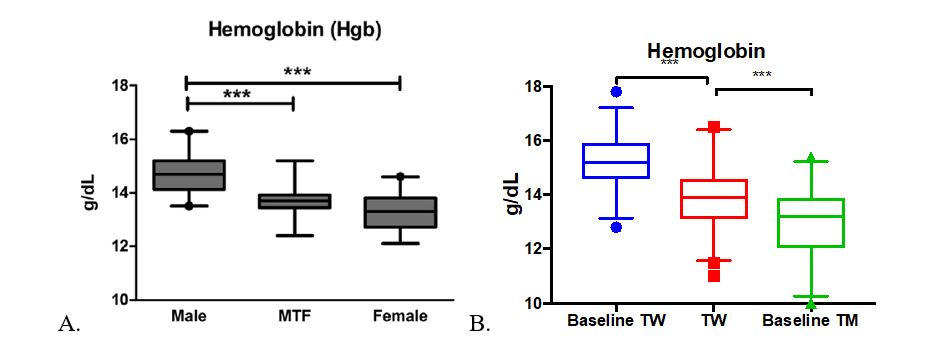
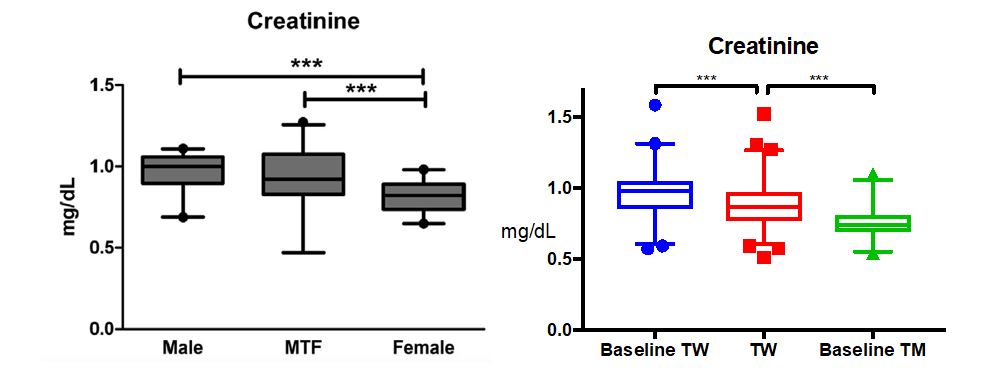
Examples abound regarding information gaps in managing patients on cross-sex hormones. For instance, there are only a handful of papers in the literature addressing care of elderly patients, and little is known about the risks or health benefits of long-term cross-sex hormone use. For children who elect to start puberty blockers so that their development in adolescence is more appropriate to their self-identified gender, long-term effects on bone health are poorly understood. A number of laboratory tests have different reference ranges for “men” and “women,” such as n-telopeptide as a marker of bone turnover. Most labs have not established appropriate reference ranges for patients on cross-sex hormones, nor are there good long-term studies to help guide management of bone health in this setting. And there may be times when the managing health care professionals do not realize that a patient is taking cross-sex hormones.
Patient identifiers are often incomplete. Many patients on cross-sex hormones have not had surgery to remove their gonads. Therefore, a trans-man can present to the Emergency Room with severe abdominal pain, but those managing his care may not suspect ovarian torsion, ectopic pregnancy, or other conditions of the fallopian tubes, ovaries, uterus, and cervix. Similarly, trans-women may have testes and prostates. Most patient registration systems lack the ability to record sex chromosomes and gender identity separately. Also, many individuals identify as non-binary; some are not taking cross-sex hormones at all. These factors affecting presentation are currently captured poorly. If at all, in the medical record but may have profound implications for care. Otherwise, implicit biases can adversely affect patient care.
Finally, we all have work to do to ensure that our patients and colleagues feel welcomed and respected in our labs, training programs, and hospitals. One important step is for each of us to gain self-awareness of our attitudes and biases, and to educate ourselves. A good starting place is Gupta’s article in Lab Medicine;3 another is the book Trans Bodies, Trans Selves by psychologist Laura Erickson-Schroth.4 Second, we must continue to foster inclusive workplaces, to stand up when we witness abuses or so-called “microagressions.” Third, we must work directly with patients to hear their concerns, and to provide the information needed regarding our lab results and pathology reports. We must respond to the gaps identified by our patients, do the research necessary to get better answers, and partner with other health care professionals to address the needs of our patients.
References
- The Yogyakarta Principles. 2017. https://yogyakartaprinciples.org/
- Meerwijk EL, Sevelius J. Transgender population size in the United States- a Meta-Regression of Population-Based Probability Samples. Am J Public Health 2017; 107(2):e1-e8. PMID 28075632
- Gupta S, Imborek KL, Kraswoski MD. Challenges in transgender healthcare: the pathology perspective. Lab Medicine 2016;47:3;180–188.
- Erickson-Schroth L. Trans Bodies, Trans Selves: A Resource for the Transgender Community. 2014, Oxford University Press.

-Dr. Upton is board certified in Anatomic Pathology and Cytology and directed an autopsy service and forensic pathology fellowship program at Beth Israel Deaconess Medical Center in Boston, Massachusetts. She has also practiced cytopathology and general surgical pathology, and has focused on genitourinary pathology, head and neck pathology, and gastroenterology (GI) and liver pathology. From 1982-85 and 1987-2002, Dr. Upton lived in Boston and taught at Tufts, Boston, and Harvard Universities. Since 2002, she has been at the University of Washington in Seattle, where she formerly directed the GI and Hepatic Pathology Service the Pathology Residency Program and the UW GI and Hepatic Pathology Fellowship. Currently Emeritus Professor of Pathology, she continues to practice Surgical Pathology, Autopsy Pathology, and Cytopathology, and she is one of the specialists at UW in the areas of GI, liver, and pancreatic pathology.