Hello again everyone! Welcome back.
This month I think it’s important to take a step back from clinical pearls, developing our interpersonal skills, and interdisciplinary dynamics and go back to what I started writing about here on Lablogatory: public health and shaping policy. (Sorry, no Transformers, Simpsons, or Star Trek this time.)
Now, you may or may not have heard in recent news that the United States Preventive Services Task Force (USPSTF) updated their long-standing guidelines for screening women for cervical cancer. Normally I wouldn’t file this away under “exciting must-read,” but I was piqued when I also read that ASCP along with the College of American Pathology (CAP), American Society of Cytopathology (ASC), American Society for Colposcopy and Cervical Pathology (ASCCP), the Society of Gynecologic Oncology, the American College of Physicians (ACP), the American Society for Cytotechnology (ASCT), the American Cancer Society (ACS) the Papanicolaou Society of Cytopathology, as well as the American College of Gynecologists (ACOG) and other professional institutions and individuals voiced concerns over the changes to the USPSTF standard.
This is a topic that can be discussed for days, but I’ll do my best to give you the readers’ digest and present the main contentions regarding this standard of patient care and laboratory methodology.
Woah. What’s going on?
Basically, because of some new research and recommendations, the USPSTF—a body which publishes the standard of care for nearly every conceivable aspect of preventive care in the US—rolled back on the algorithms for screening women for HPV and cervical cancer. It all comes down to the utilization of co-testing (doing both Pap smear cytology and HPV testing for certain age demographics) as a point of contention. Under a banner of addressing keywords like “cost” and “harm,” these new recommendations have left clinicians both in and out of the lab in stirrups—sorry, couldn’t resist that one. But don’t worry, I wouldn’t be able to track these changes or even understand them without some sort of visualization. When it comes to recommendations, standards, and guidelines I’m about as proficient as a broken manual diff counter…
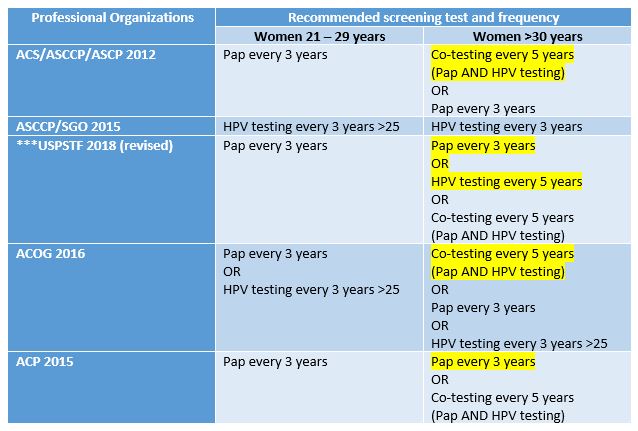
Slow down. Explain co-testing and primary testing? What exactly do the old and new recommendations mean?
Okay. When women undergo routine cervical cancer screening they receive Pap smears (cytologic examinations) every three years. This testing has been the standard for a number of years and is adequately sensitive for women up to the age of 30. Often times, these younger women may have slight intraepithelial changes (LGIL) which are considered low grade and remiss on their own. After that age it has become standard practice to add the additional test (while collecting the Pap specimen) of HPV DNA testing. This adds an increased level of sensitivity/specificity and is called co-testing. The new recommendations depart from this co-testing model, citing that there are increased harms (in the form of false positives) which ultimately lead to waste and unnecessary testing for women after the age of 30. Primary testing would mean only screening with HPV DNA assays after 30. According to the National Cancer Institute, all available literature on the subject of HPV and cervical cancer testing adequately demonstrates that co-testing is the best option. A number of studies were compiled to address the harm vs. benefit of Pap and HPV testing. Together, however, these tests decrease the incidence of cervical cancer. New guidelines were made based off mathematical projections and cost-benefit analyses which try and minimize losses for screening. Dr. J. Kim, a public health researcher at Harvard, was integral in contributing models which projected the cost/benefit of changing HPV guidelines. Essentially, the study projected that, when considering “harm” (i.e. colposcopy/false negative) abandoning co-testing changed the mortality rate from 0.3-0.76 per 1000 women with co-testing, to 0.23-0.29 per 1000 women with primary HPV testing. An impressive and significant statistical advantage. However, the total number of unscreened women with mortality rates was between 1-2%. This study was a microsimulation done from historical data within rates of cytologic detection and retrospective testing data on women, projected for a future hypothetical 5 year interval. Fascinated by this study, I tried to reach out to Dr. Kim to discuss limitations in using models and simulations and public health evidence to change practices, but I’m sure she is busy and could not respond in time.
So, to co-test or not to co-test, that’s the question. Right?
In its simplest sense, yes. The major medical professional societies also publish their most current recommendations for practice standards—and the issue is that the USPSTF took a departure from what most of the professional societies recommend regarding co-testing. Late last year, the CAP, ASC, ASCT, ASCP, and the PSC issued a statement under their independent collaboration called the Cytopathology Education and Technology Consortium (or CETC). In this response to the USPSTF guideline changes, they discussed their concerns. Specifically, their objections center around the fact that without co-testing for screening, cancer prevention might be impacted negatively. The CETC claimed that sensitivity is already maximized with previously recommended co-testing guidelines. They also cite that there is only one FDA approved HPV primary screening test available—and not all labs have it! More so, CETC discussed the need to keep morphological testing continuous for women who have histories of Pap smears, the potential to overwhelm colposcopy services for screening all positive HPV patients, and the honest reality that not all clinicians would be compliant with the way the USPSTF recommends testing. The bottom line from this consortium:
- Cytology and high-risk HPV co-testing should be kept as the standard screening for women aged 30-65
- Primary HPV screening should only be done with validated, FDA approved testing methodologies
- HPV screening methods should continue their current schedule until longitudinal data can offer new evidence for changes
So, what’s the current technological climate for how we test for HPV?
Currently, most clinicians do co-testing. The standard for Pap smears utilizes a physical tool to collect epithelial cells from the cervix at vaginal, ectocervical, and endocervical sites. The swabs are prepped on 1-2 slides, fixed with alcohol or other spray cell-preservatives and sent to labs for cytologic examination. The basic Papanicolaou staining procedure uses hematoxylin for nuclear staining, and two cytoplasmic counterstains. This is essentially a modified H&E stain to clearly visualize morphology. Staining is rarely done manually and some instruments offer stain/prep combination capability. I couldn’t find too much information on this, but I remember there not being too many official FDA approved prep machines for Pap specimens. Cytotechs and pathologists read the slides and issue sign outs on morphology according to the Bethesda system—very heavy read, don’t bother; essentially it has three main categories of normal, benign changes, and abnormal. According to ASC “for squamous lesions, TBS terminology includes atypical squamous cells of undetermined significance (ASCUS), low grade intraepithelial lesion (LGSIL or LSIL), high grade intraepithelial lesion (HGSIL or HSIL) and squamous cell carcinoma. Some laboratories also incorporate other terminologies of dysplasia and/or cervical intraepithelial neoplasia (CIN) into their reports. For glandular lesions, TBS terminology includes atypical glandular cells of undetermined significance (AGUS) and adenocarcinoma.”
As of now FDA approval for HPV primary testing for high-risk strains is limited to the Roche Cobas hrHPV test. I could link you to their website, but you’ll be sold right away. They tout the future of HPV screening is HPV primary testing and to do away with the Pap! Their graphs and figures are impressive (just like their price tag!) and there’s no doubt that sensitivity is something that real-time PCR provides more than cytologic examination. But, as always, more assays will be approved, and advancements will tweak the sensitivity and specificity higher and higher.
Got it. So, technology and lab tests are always advancing, why can’t we make this change?
It’s not so easy to change the method or assay we use to screen or diagnose patients in the lab. If you recall, I talked about how the hospital I’m currently rotating in is leading the region in advancing the new high-sensitivity troponin assays. It’s still a hard sell to many even though the data and projections seem to all point to a green light. But that’s a paradigm shift that involves side-stepping from one immunochemical assay to a more sensitive immunochemical assay. It’s the same stuff, just sharper and with more clinical data to interpret with regards to acute coronary symptoms and clinical risk stratification. Swapping an old car for a new car. This conversation is a bit more complex. The recommendations for cervical cancer screening suggest that we should move away from mostly morphologically-driven, human-based cytology interpretation and move toward PCR-based assays for detection. Literally apples to oranges. We might think we know which one is better right now, but longitudinal studies are the only way to really tease out if this change in practice to improve patient outcomes in the long run.
Where do we go from here?
Ultimately, I think a few things need to happen for this recommendation to become standard practice. First, professional societies in every discipline from gynecology to cytology need to come to an agreement. It remains to be seen whether certain agencies will adopt and recommend the USPSTF guidelines, and statements from groups like CETC reveal a vote of no confidence in this current climate. Ultimately, because of numerous objections (including the ones from ASCP and the CETC) the USPSTF does say that co-testing is still optional between patient and provider, so we’re not really in crisis mode. But what happens when the advancements and the recommendations catch up to our ability to abandon the cytologic contributions of a future useless Dr. Papanicolou? We could probably deal with that when it comes to fruition, but until then we have a real discussion about what “harm” really is. Is colposcopy flat out harm? Or are the false positives that reflex to further testing? Is the current practice a safety-net for populations across socio-economic tiers with varying access to screening in the United States? When compared to other countries, HPV prevention, vaccination, and screening is much more easily facilitated. Is this a contributing factor for our messy guidelines? Will there be more options for FDA approved methodology in the near future? There remain a number of good questions which require examining cross-sections of data and patient outcomes. And, I believe, we may see change soon—but not quite yet.
What are your thoughts? What have you experienced in your lab or clinic? Leave your comments below!
Thank you and see you next month!
References
- ASCP One Lab. 2018. ASCP Declares Victory for Patients with Revised USPSTF Cervical Cancer Recommendation. Aug 21, 2018. Accessed Sep 2018: http://labculture.ascp.org/community/news/2018/08/21/ascp-declares-victory-for-patients-with-revised-uspstf-cervical-cancer-recommendation
- 2018. Vaccines and Preventable Diseases. HPV Vaccine Recommendations. Centers for Disease Control and Prevention. Atlanta, GA. Accessed Sep 2018. https://www.cdc.gov/vaccines/vpd/hpv/hcp/recommendations.html
- 2017. Response to New USPSTF Draft Guidelines for Cervical Cancer Screening. Cytopathology Education and Technology Consortium. Accessed Sep 2018: https://s3.amazonaws.com/ascpcdn/static/ONELab/pdf/2017/CETC+-USPSTF+Letter+10-2-17.PDF
- Basu, S. 2013. Complexity in Mathematical Models of Public health: A Guide for Consumers of Models. PLOS, Medicine. Oct 29, 2013. https://doi.org/10.1371/journal.pmed.1001540
- Felscher, K. 2018. The science behind new screening guidelines for cervical cancer. An Interview with Dr. J. Kim. Harvard T.H. Chan, School of Public Health. Accessed Sep 2018: https://www.hsph.harvard.edu/news/features/science-behind-new-screening-guidelines-cervical-cancer/
- Kim, J. 2017. Screening for Cervical Cancer in Primary Care. Journal of the American Medical Association (JAMA). 2018;320(7):706-714. Doi:10.1001/jama.2017.19872
- Lerman, L. 2018. Screening for Cervical Cancer – New Tools and Opportunities. Journal of the American Medical Association (JAMA) – Opinion, Editorial. Vol. 320(7):647-649
- Nayar, R. 2017. Primary HPV Cervical Cancer Screening in the United States: Are We Ready? Journal of the American Society of Cytopathology (2017) 7, 50e55
- Nelson, R. 2018. USPSTF Updated Recommendations for Cervical Cancer Screening. Medscape. Accessed Sep 2018: https://www.medscape.com/viewarticle/900985#vp_3
- 2018. Cervical Cancer Screening (PDQ) Health Professional Version. National Cancer Institute. Accessed Sep 2018: https://www.cancer.gov/types/cervical/hp/cervical-screening-pdq#link/_133_toc
- Sawaya, G. 2018. Cervical Cancer Screening—Moving from the Value of Evidence to the Evidence of Value. Journal of the American Medical Association (JAMA), Internal Medicine. doi:10.1001/jamainternmed.2018.4282
- Up To Date. 2018. Cervical cancer screening recommendations from United States professional organizations. Accessed Sep 2018: https://www.uptodate.com/contents/image?topicKey=7575&search=&source=outline_link&imageKey=PC%2F82951
- 2018. Cervical Cancer: Screening. Recommendation Summary. August 2018. Accessed Sep 2018: https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/cervical-cancer-screening2
- USPSTF. 2018. Screening for Cervical Cancer, US Preventive Task Force Recommendation Statement. US Preventive Task Force. Journal of the American Medical Association (JAMA) 2018;320(7):674-686. Doi:10.1001/jama/2018.10897

–Constantine E. Kanakis MSc, MLS (ASCP)CM graduated from Loyola University Chicago with a BS in Molecular Biology and Bioethics and then Rush University with an MS in Medical Laboratory Science. He is currently a medical student actively involved in public health and laboratory medicine, conducting clinicals at Bronx-Care Hospital Center in New York City.
