Hi everyone! Back with another piece about the life between the lab and medical school. This time, I’d like to take a minute to talk about some new and exciting developments in laboratory diagnostics happening right now: immunoassays for critical troponins are undergoing an evolution. Fourth generation testing is slowly developing into its fifth-generation upgrade. Labs across the country are starting to discuss the relatively new FDA approved fifth-gen cardiac troponin T assay which has been shown to be a high-sensitivity test. But what does this mean for labs? Specifically, what does it mean between the bench and the bedside? The hospital I’m currently on service at is rolling out the first beta-test of this assay in New York City right now, and as it turns out—it’s going to change a lot. Not only will the new understanding of cardiac enzyme reference ranges need a complete overhaul but tailoring appropriate clinical responses to those values will need to be looked at as well. I’m not a sales rep and this isn’t going to be an adventure in comparative statistical analytics, but I think it’s a great time to have a conversation early on about what these new generation assays could mean for us in the lab.
A Whole New World
When I was in graduate school, doing my MLS training we were taught the same cardiac enzyme assay history that was developed over the last 50 or so years. Early acute markers of inflammation relating to acute myocardial infarctions (AMIs) with respect to acute phase reactants AST, LDH, CRP, etc. As more technology advanced, specific biomarker analyses of individual detection of things like CK or myoglobin became useful. The WHO criteria for AMI then established (and re-established since the 1970s) the laboratory requirements for CK-MB and detectable levels of troponin to correlate with clinical findings. Further sensitivity and specificity developments, and clinical research like the GUSTO and APACE trials, showed us just how sensitive newer (then troponin T and I) cardiac assays could be. Not to mention, instead of rule-in/rule-out criteria, we had the development of risk stratification. And as instrumentation developed so did our testing—CK and LDH replaced with CK-MB and its isoforms, AST went the way of Myoglobin, and LD ratios became reliable troponins!

Ask most clinicians today about cardiac enzyme, cardiac injury studies, or other related markers and you’ll hear about CPK, CK-MB 1, 2, 3 and other isoenzymes, LDH electrophoresis, and of course troponins T and I. What’s more is that the reference ranges for most of these tests haven’t really changed much either. If I called you and said your patient in 706-W has a Troponin of <0.03 you might be relieved for now. If I said that same patient’s troponin was 0.560—we might have a different story unfold. But what if I told you your patient’s troponin was 13, and was trending down from 15? What do you do with that? What if I only called to report a troponin-DELTA which was 0.0? Was there an in-service you missed? Possibly. Sounds like your institution might have 5th-gen on board.
What was wrong with the old troponin?
In a word: nothing, really. This really isn’t about buy-in for a “better” test or a better detection method. This is about creating a dialogue about improving risk stratification for our patients with coronary disease. Let’s go back to Chicago, specifically the last hospital I worked in before starting medical school: Swedish Covenant Hospital. Having been through a few hospitals in my time, I can say you’d be hard pressed to find a more streamlined, albeit small community hospital, laboratory service. Running a full gamut of SIEMENS instrumentation and critical middleware-software, the management there ran a tight ship—which included critical troponins. We ran the TnI-Ultra assay on the ADVIA Centaur/XP platform. It was your standard three-part immunoassay sandwich test with a biotin-streptavidin antigen-antibody detection. It was fast, used little reagents, was relatively stable, had a great system of QC, and was calculably-flexible between heparinized and ETDA plasma samples. Two-point calibration kept it tight between (and this is from memory, loosely) 0.006 and 0.50 ng/mL, and I believe we called our critical values at 0.40 ng/mL. This was a good test, and it’s used in many labs today still. It’s got great stability and has room to interpret ACS risk stratification based on population data in each location. People understand those results, too. But exactly how much room between, let’s say 0.10 ng/mL and >0.50 ng/mL, is there to stratify that risk? Complex decision algorithms then become hybrids of institutional cardiology recommendations, American College of Cardiology (ACC) recommendations, emergency department input, and, of course, laboratory management recommendations.
Here at Bronx Care Hospital Center (BCHC), I spoke with a laboratory manager about rolling out this brand new high-sensivity troponin (hs-cTn), and we discussed their vaildation and policies. Along with this new project, the hospital has been a vocal part of American College of Cardiology ACC17 Acute and Stable Ischemic Heart Disease program addressing topics including marijuana use and ACS, stable angina risk stratification, NSTEMI sex differences in revascularization and outcomes, treating cocaine related ACS with beta blockers, and research Anticoagulation Therapy After Anterior Wall ST-Elevation Myocardial Infraction in Preventing LV Thrombi. So, it seems fitting that this is as good as any a pilot location for cardiology departments city-wide to watch and learn from the hs-cTn roll-out!
Tell me more about this new test…
This hs-cTn assay is an electrochemiluminescence immunoassay (ECLIA) that uses two monoclonal antibodies against human cardiac troponin T. At this institution, they are using this immune sandwich assay on a Roche Cobas E with a similar biotin-streptavidin coated microparticle complex as with the previous generation testing. This is a short test with similar reagent use and stability as before, and only for lithium heparinized samples. With a relatively quick turn-around in less than 10 minutes, the new hs-cTn offers critical information for clinical correlations on the fly from potential STEMI codes coming into the ED.
Without going into horrific details about validating data on old and new troponins across patient populations, there is something interesting to note here which came up in discussion with the lab manager: new reference ranges. Now, with testing sensitivity, cross-reactivity, ranges, and interfering substances, the ranges are no longer the decimal-place values reported that we’re all used to. The ranges we work with now at BCHC are cutoff between <12 ng/L for positives and >52 ng/L for critical values suggestive of acute coronary syndrome. The analytical measuring range is much larger now between 6-10,000 ng/L. With the adjustments for limits of detection and blanks on instrumentation, the specificities of these values are normalized on a larger index for reflecting differences in male and female cardiac enzyme activity to a common cutoff of that <12 ng/L value. But more so than just a value, a new part of trending troponins becomes much more important: the delta values. These are more acutely indicative of the cardiac necrosis and/or condition of other non-specific heart tissue damage en vivo occurring in patients. Taking all this into account, you now have a much wider and broader range of values to interpret and incorporate into your clinical decision making, which brought up a few questions when I spoke with lab staff as well as cardiologists.
So, what’s wrong with the new troponin?
Okay, that’s a fair question at this point. And my answer is still: probably nothing. Sorry to be so inconclusive, but it’s still early days. There is a lot of data to support moving toward newer generation hs-cTn testing since it has been available outside of FDA-approval in Europe before January 2017. Research done in the Department of Internal Medicine and Central Institute for Medical Laboratory Diagnostics at Innsbruck Medical University in Austria show that new troponins might not be that different (read: better) than their 4th generation counterparts, at least with regard to emergency room visits. T. Ploner, et. al, argue that diagnosing AMI in the ED doesn’t really benefit from the heightened sensitivity offered by the new Roche 5th gen assay (Figure 1, Figure 1). But, when they compared the detection of other cardiac disease including AMI, unstable angina, heart failure, arrythmias, pulmonary emboli, pulmonary disease, anemia, renal disease, and several other entities, the 5th generation assay could more readily detect changes early and provide clinicians with critical data quickly (Figure 1, Figure 2).
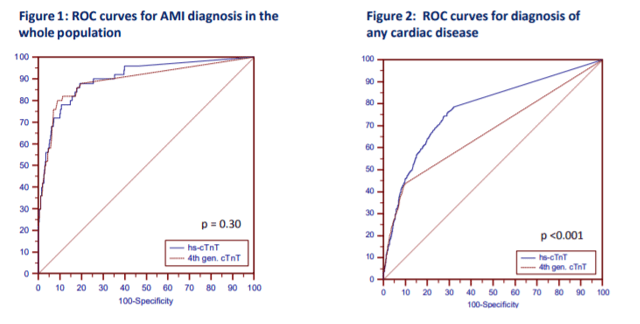
(Source: Ploner et al. (2017) High-Sensitivity cardiac tropnonin assay is not superior to its previous 4th cTnT assay generation for the diagnosis of acute MI in a real-world emergency department, link: http://spo.escardio.org/eslides/view.aspx?eevtid=48&fp=P4177)
Finally, I think a review paper from the American Journal of Medicine last year summarized it best. Coming from the Department of Cardiovascular Diseases and Department of Laboratory Medicine and Pathology at Mayo Clinic in Rochester, MN, the authors discussed concern over the troubling over-sensitivity and potential pitfalls of reaching too far with hs-cTn testing. Their bottom line: collaborating on data and accuracy between cardiology, laboratory medicine, and emergency medicine, there is a great potential for this super sensitive test to provide clinicians with very useful data in the near future. We just have to process that data correctly. As always, results should be interpreted in conjunction with clinical presentation including medical history and laboratory data. But in the case of new and coming advances in critical care, there seem to be some common themes between my conversations here at BCHC and in what I read in the literature regarding how to ensure we move forward appropriately.
- Multidisciplinary educational efforts are critical. The ER, the lab, and the cardiology department at each institution utilizing 5th gen troponins need to understand the new ranges, the new triaging cutoffs, the clinical correlations for consulting the ICU/CCU, and how to understand the deltas for their patient populations.
- Create clear communications for your laboratory values. Will you normalize for gender or provide sex-specific confidence interval reporting? Will you provide tables for suggested value correlations with AMI/ACS protocols?
- Order sets and in-service training. You’d be surprised how much the nitty gritty details of lab draws and ordering appropriate tests/tubes could slow down your institution’s advancement.

(Source: Sandoval, Jaffe (2017) Using High-Sensitivity Cardiac Troponin T for Acute Cardiac Care, The American Journal of Medicine (2017) 130, 1358–1365, doi:10.1016/j.amjmed.2017.07.033)
At the very end of the day, it’s up to the institution. Clinical centers have to follow their own guidelines for cardiac pathology. ACC/ESC/AHA guidelines and Universal MI definitions are for clinical correlation across locations, but a single roll-out of a fancy new test can’t make a better ER. It really does take communication, collaboration, and accountability. We all have to push the envelope and practice at the top of our scopes in order to make health care better every day. One of the ways we might be able to do that now is by considering these new high-sensitivity troponins as a useful new clinical tool to improve patient outcomes.
Thanks for reading! See you next time!
Disclosure: I am no longer affiliated with Swedish Covenant Hospital in Chicago as an employee, and any recount of policy and/or procedure(s) specifically regarding their cardiology protocols and troponin resulting are a historical and anecdotal account of my time working there in the past. I have no affiliations with SIEMENS, Advia, Roche, or any other medical laboratory instrumentation institution. I am only affiliated with Bronx Care Hospital System as a current rotating medical student and my account of their transition to 5th gen testing is anecdotal from discussions with in-house staff, cardiologists, and laboratory management.

–Constantine E. Kanakis MSc, MLS (ASCP)CM graduated from Loyola University Chicago with a BS in Molecular Biology and Bioethics and then Rush University with an MS in Medical Laboratory Science. He is currently a medical student at the American University of the Caribbean and actively involved with local public health.
