Case History:
A 45 year-old woman was hospitalized for a bilateral subarachnoid hemorrhage with right-sided intraventricular hemorrhage secondary to a basilar artery aneurysm. On hospital day 2, after endovascular coiling of her aneurysm, she developed seizure-like activity. She was found to have new bilateral thalamic and brainstem infarcts with increased hydrocephalus and had an external ventricular drain placed.
She showed some minimal neurological improvements over the next 2 weeks until hospital day 17 when her mental status acutely declined and she developed a fever, leukocytosis, and meningeal signs. 8cc of cloudy, yellow fluid was collected via lumbar puncture. Analysis of the fluid showed pleocytosis (cell count of 54K) with a neutrophil predominance and markedly elevated protein.




Laboratory Identification:
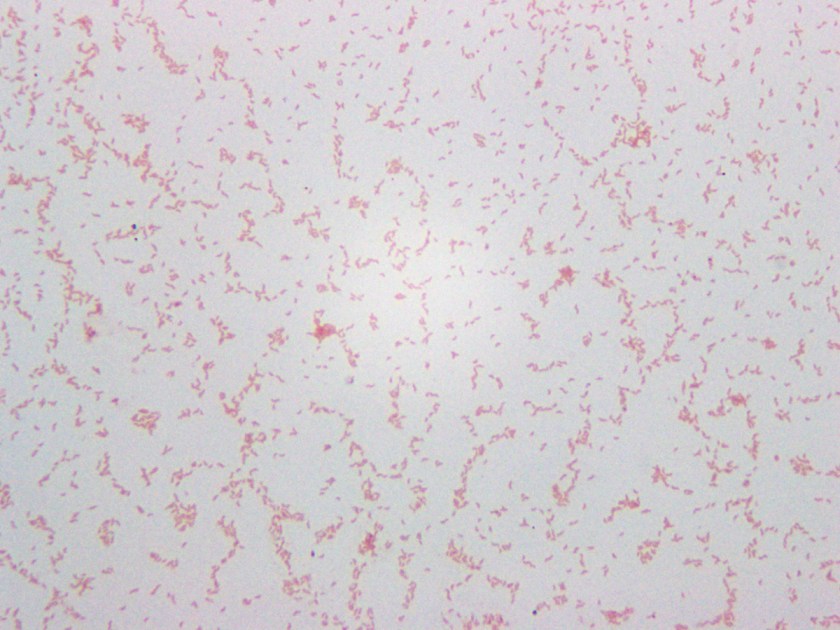
Initial review of the spinal fluid gram stain showed many polymorphonuclear leukocytes and what appeared to be paired gram negative cocci suspicious for Neisseria meningitidis. However, many organisms lacked the more characteristic “kidney-bean” shape. Further inspection of the gram stain revealed many gram negative organisms which more closely resembled bacilli. Given the patient’s history and clinical course, it was determined the pathological agent was most likely a gram negative coccobacillus. On culture, the bacteria formed smooth, round, opaque colonies on Blood and Chocolate agar and was lactose non-fermenting on MacConkey agar. The bacterial colonies were also oxidase negative.
Mass spectrometry identified the organism as Acinetobacter radioresistens.
Rare gram positive appearing organisms of similar shape were also located on the gram stain. This demonstrates that Acinetobacter is known to occasionally retain the crystal violet stain leading to cases of initial misidentification.
Discussion:
Acinetobacter radioresistens is one of about 30 species of bacteria included in the Acinetobacter genus. Acinetobacter is characterized as a gram negative, aerobic coccobacillus which is non-motile, non-fermentative, and oxidase-negative. It grows well on standard aerobic media and typically forms smooth, round, mucoid colonies at 37°C. Acinetobacter is a water organism which preferentially colonizes aquatic, humid, and tropical environments; perhaps accounting for the increased incidence of Acinetobacter infections between the months of July and October.
While there have been reported cases of community-acquired Acinetobacter pneumonia in Southeast Asia and Australia, in most areas of the world Acinetobacter is known primarily as an agent of nosocomial infections. Studies show that an estimated 33% of healthcare workers are colonized with Acinetobacter and that it is one of the most prevalent bacteria isolated from the white coats of medical students. Despite its ubiquity in hospitals, Acinetobacter infections are relatively rare. Many patients may be colonized with it, but Acinetobacter only usually causes disease in immunocompromised and/or critically-ill patients with long hospitalizations. At particular risk are ventilated patients supported with multiple lines, drains, and catheters. Acinetobacter is reported as the pathological agent in a small percent of ventilator-associated pneumonias, central line-associated bloodstream infections, catheter-associated urinary tract infections, and surgical site infections. It is also recognized as a cause of nosocomial meningitis in neurosurgical patients with external ventricular drains, especially those with a history of intracranial hemorrhage and recent prior antibiotic therapy.
Acinetobacter infections are of particular concern because several species demonstrate resistance to many antimicrobials. Acinetobacter baumanni, the species responsible for the majority of Acinetobacter infections, has demonstrated resistance to 1st-3rd generation cephalosporins, macrolides, penicillins, and aminoglycosides. Because these infections are robust and difficult to treat, patients with Acinetobacter infections have a 25-75% mortality risk depending on the site of their infection and their baseline cardiopulmonary and immune status. Currently, carbapenems are considered the gold standard treatment.
-Elaine Amoresano, MD, is a 1st year anatomic and clinical pathology resident at the University of Vermont Medical Center.
-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Assistant Professor at the University of Vermont.