Case History
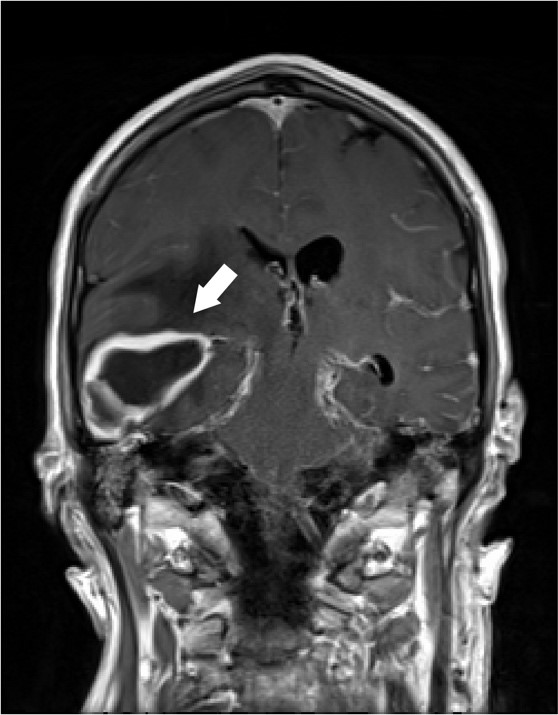
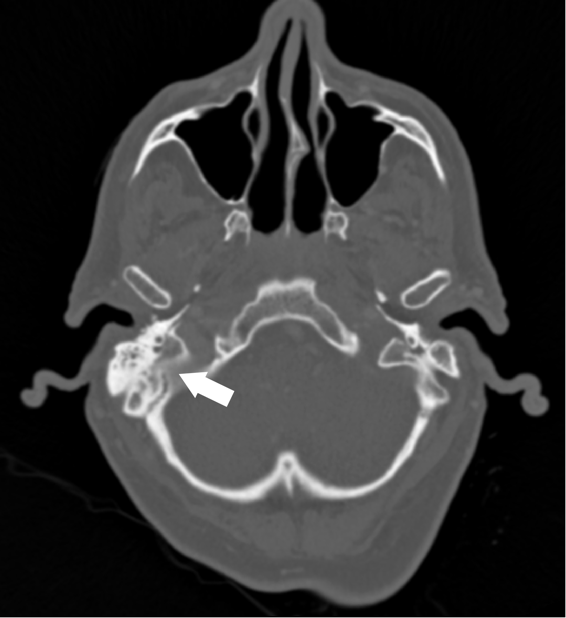
An 18 year old healthy female presented to the emergency department of a tertiary care hospital in Minnesota for headache, vomiting, and sore throat. She did not have any significant past medical history. Due to meningitis concerns, lumbar puncture and head computed tomography (CT) imaging were performed. The CT scan showed an accumulation of fluid in the posterior right frontal sinus with scattered mucosal thickening. However, her cerebrospinal fluid (CSF) profile was insignificant, with normal protein and glucose levels. CSF culture was ordered, and two sets of blood cultures were drawn.
Based on the examination and presenting symptoms, pharyngitis was suspected, and she was discharged with Amoxicillin (500mg Q6H for five days). However, her strep throat screening returned negative. Her blood culture was negative. CSF culture was also negative. Cryptococcal antigen and Enterovirus PCR were performed; however, both results were negative.
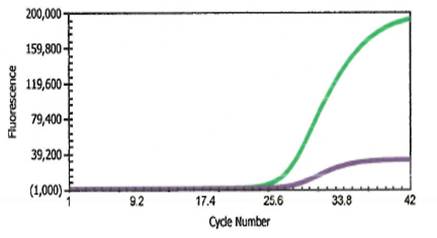
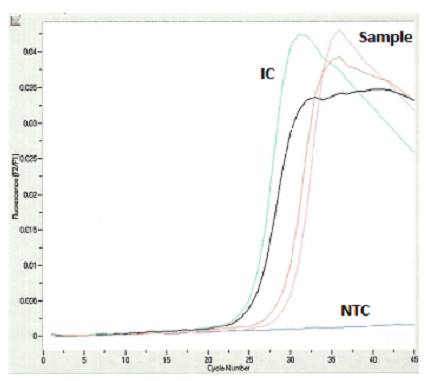
She returned to the ED two days later for a worsening headache and newly developed photophobia. Additional history revealed that she went swimming in a lake two weeks prior to her first presentation at the ED. Her CSF was sent for the Ova and Parasite (O&P) exam for suspicious parasitic meningitis. The CSF O&P test was negative. CSF PCR for amoeba was also performed at a reference laboratory, and the results came back positive with Balamuthia mandrillaris. The patient was then given flucytosine, fluconazole, and azithromycin.
Discussion
Balamuthia mandrillaris belongs to a group of free-living amoebae, including Acanthamoeba species and Naegleriafowleri, that cause fatal encephalitis.1 Balamuthia mandrillaris is the only known species of the genus Balamuthia that causes infections in humans. Encephalitis caused by B. mandrillaris is known as granulomatous amoebic encephalitis (GAE). GAE is characterized as a subacute to a chronic infection that can last several months to years.2 GAE differs from primary amoebic meningoencephalitis (PAM) caused by Naegleria fowleri, which typically causes an acute onset lasting a few days.
While ecological niches of B. mandrillaris are not well understood, they have been reported to be isolated from dust, soil, and water.r Both trophozoite and cyst forms can enter the body through the nasal passage or ulcerated/broken skin; however, the trophozoite stage causes associated disease manifestation and represents a diagnostic stage.2
Brain-eating amoebas are traditionally difficult to diagnose. Hematology and chemistry profiles of CSF of affected individuals are generally unremarkable, although, sometimes, increased monocytes and lymphocytes, along with increased protein levels, are seen in some cases of GAE.3
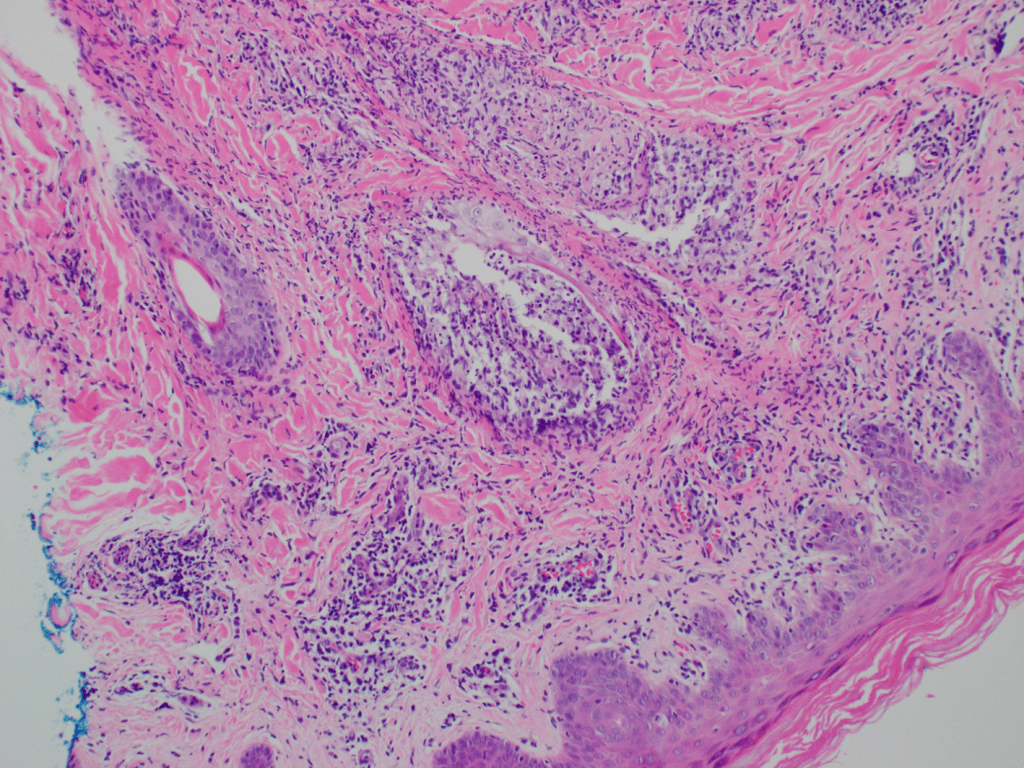
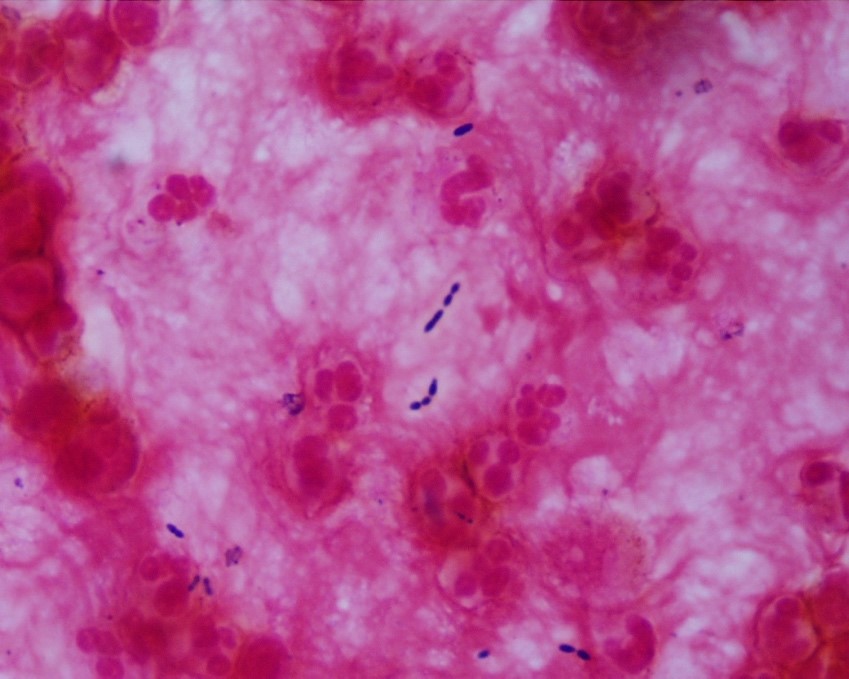
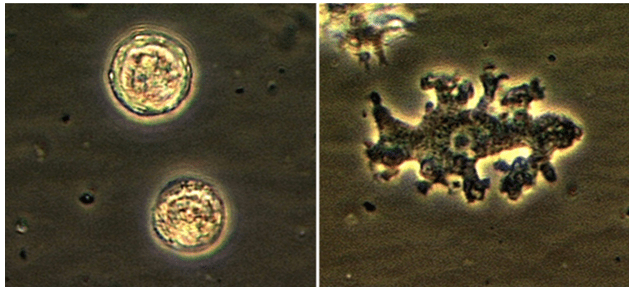
The most common method of laboratory diagnosis of B. mandrillaris is a microscopic examination of CSF wet mount (Figure 1) or via immunohistochemical staining of CSF or brain biopsy.1 With advancements in technology, species-specific nucleic acid amplification tests (NAAT) can be performed to diagnose B. mandrillaris infection accurately. However, there is no commercially available NAAT for the free-living amoeba. Only very few laboratories, such as State departments of health laboratories, Centers for Disease Control (CDC) and Prevention, or commercial reference laboratories, develop these tests as a laboratory-developed test (LDT). Histological assessment of biopsies from brain lesions may reveal tumor-like appearance or perivascular monocytic necrosis of affected areas.1 While there have been significant technological advancements, the prognosis stays at less than a 5% survival rate,6 with only roughly 25% of cases diagnosed antemortem. One possible reason for delayed laboratory diagnosis is the challenges in performing the microscopic examination in clinical microbiology laboratories since it requires expertise for accurate identification of the organism. Additionally, most clinical microbiology laboratories do not readily have an in-house LDT for free-living amoeba NAAT. Therefore, the turnaround time for diagnosing B. mandrillaris or any free-living amoeba is typically longer when specimens have to be sent out to reference laboratories.
Diagnosis of B. mandrillaris encephalitis solely based on clinical symptoms is often challenging due to similar presentation in other causes of infectious encephalitis. B. mandrillaris can affect both immunocompetent and immunocompromised individuals.1,3,4 The first B. mandrillaris case was reported in a deceased baboon in the San Diego Zoo in 1986.1 The majority of patients were diagnosed postmortem.1 While most B. mandrillaris infections are actively acquired through nasal passages or skin penetration, rare post-mortem cases of passive transfer of the organism from organ transplantation have been reported.6 With technological advancement, there have been successes in pre-mortem diagnoses in recent years.1,4 According to the known cases, individuals of Latin American origin are more likely to contract the disease; it is unknown if it is due to increased exposure or a genetic predisposition.1 Similar to other free-living amoebae, B. mandrillaris can be generally found in warmer climates or tropical regions. Of approximately two hundred cases reported worldwide, about 34 were reported in Latin America, from Mexico to Brazil, while some were from Japan, New Zealand, England, and other European countries. The Southwestern United States also contributes 30 cases, mostly in Arizona, Texas, and California.1 In the United States, there have only been 109 cases directly reported to CDC from 1974 to 2016.2,7 We believe that this is the first case of Balamuthia reported in Minnesota. The number of exact cases would be difficult to be determined due to misdiagnosis and rare occurrence of the disease or cases not reported to CDC or the state department of health.
While investigational drugs for B. mandrillaris GAE are in development, combination therapy of flucytosine, fluconazole, pentamidine, and azithromycin or clarithromycin has shown successes.2 Our patient was successfully treated with flucytosine, fluconazole, and azithromycin.
References
- Matin A, Siddiqui R, Jayasekera S, Khan NA. Increasing importance of Balamuthia mandrillaris. Clin Microbiol Rev. 2008 Jul;21(3):435-48. doi: 10.1128/CMR.00056-07. PMID: 18625680; PMCID: PMC2493082.
- Centers for Disease Control and Prevention. (2019, August 23). CDC – Dpdx – free Living Amebic Infections. Centers for Disease Control and Prevention. https://www.cdc.gov/dpdx/freelivingamebic/index.html.
- Kofman A, Guarner J. Free Living Amoebic Infections: Review. J Clin Microbiol. 2021 Jun 16:JCM0022821. doi: 10.1128/JCM.00228-21. Epub ahead of print. PMID: 34133896.
- Pietrucha-Dilanchian, P., Chan, J. C., Castellano-Sanchez, A., Hirzel, A., Laowansiri, P., Tuda, C., Visvesvara, G. S., Qvarnstrom, Y., & Ratzan, K. R. (2011). Balamuthia mandrillaris And Acanthamoeba Amebic Encephalitis With Neurotoxoplasmosis Coinfection in a patient with Advanced HIV Infection. Journal of Clinical Microbiology, 50(3), 1128–1131.
- Ong TYY, Khan NA, Siddiqui R. 2017. Brain-eating amoebae: predilection sites in the brain and disease outcome. J Clin Microbiol 55:1989 –1997. https://doi.org/10.1128/JCM. 02300-16.
- Centers for Disease Control and Prevention. 2011. Balamuthia mandrillaris transmitted through organ transplantation—Mississippi, 2009. Am J Trans-plant 11:173–176. https://doi.org/10.1111/j.1600-6143.2010.03395_1.x.
- Jennifer R Cope, Janet Landa, Hannah Nethercut, Sarah A Collier, Carol Glaser, Melanie Moser, Raghuveer Puttagunta, Jonathan S Yoder, Ibne K Ali, Sharon L Roy, The Epidemiology and Clinical Features of Balamuthia mandrillaris Disease in the United States, 1974–2016, Clinical Infectious Diseases, Volume 68, Issue 11, 1 June 2019, Pages 1815–1822, https://doi.org/10.1093/cid/ciy813
-Alejandro Soto, MLS (ASCP)CM is a junior medical technologist who is passionate about clinical microbiology.

-Phyu M. Thwe, Ph.D., D(ABMM), MLS(ASCP)CM is Microbiology Technical Director at Allina Health Laboratory in Minneapolis, MN. She completed her CPEP microbiology fellowship at the University of Texas Medical Branch in Galveston, TX. Her interest includes appropriate test utilization and extra-pulmonary tuberculosis.