Case Description
A 62 year old male with unknown past medical history was dropped off at the emergency department by EMS after being found altered with concern for IV drug use. On presentation he was febrile to 104.5o F, tachycardic, and although he was initially responsive, his mental status deteriorated. Labs were drawn and broad-spectrum antibiotic coverage with vancomycin, cefepime, and metronidazole was initiated in the ED. He then had a tonic-clonic seizure event and was given intravenous levetiracetam. A CT brain showed a right inferior temporal lobe lesion, initially interpreted as likely glioblastoma multiforme, causing subfalcine and uncal herniation. MRI revealed a ring-enhancing mass measuring 3 cm x 3 cm x 3 cm in the right temporal lobe with significant surrounding edema. CT of the temporal bones also revealed right mastoiditis (Figures 1 and 2).
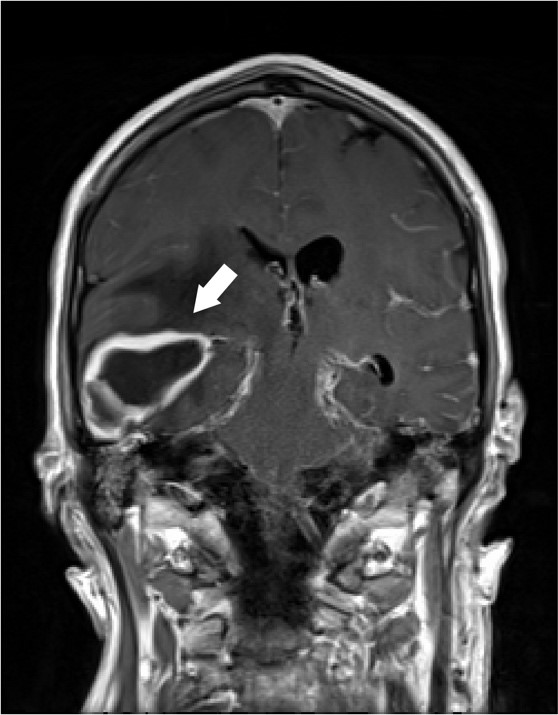
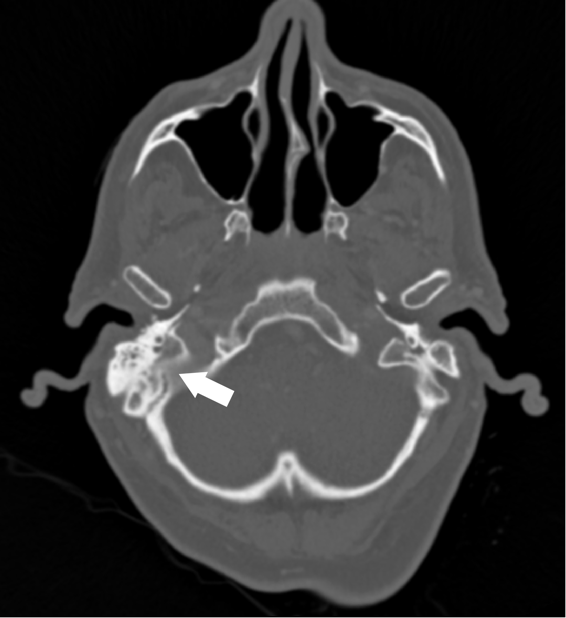
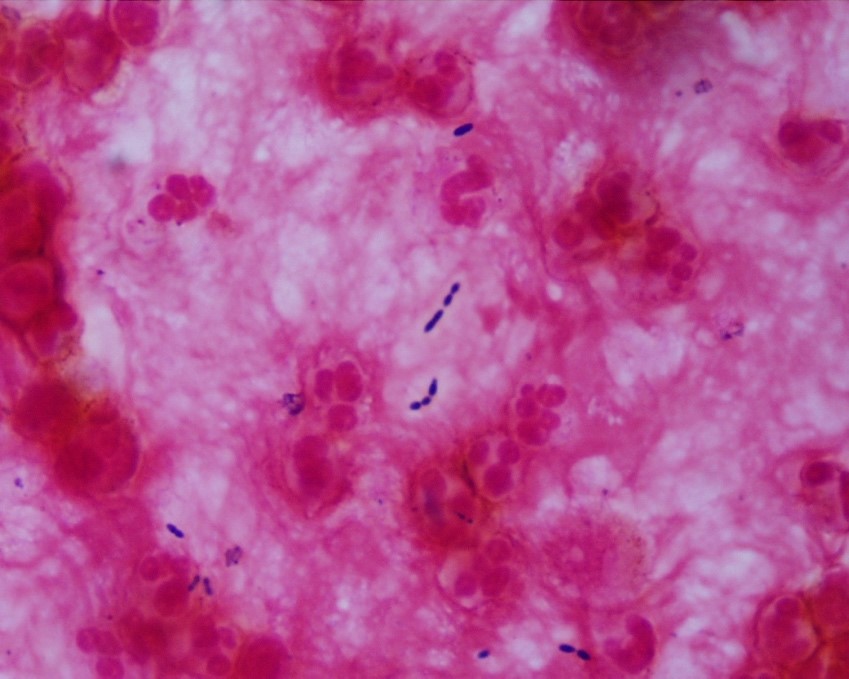
Neurosurgery evacuated 17 mL of fluid from the mass and a ventricular drain was placed. Gram stain of the evacuated fluid identified many white blood cells and few gram-positive cocci in pairs, chains and clusters (Figure 3). Postoperatively, the patient was mechanically ventilated and medicines were used to support his blood pressure in the ICU. Broad-spectrum antibiotics were continued for CNS penetration and activity against possible oral flora.
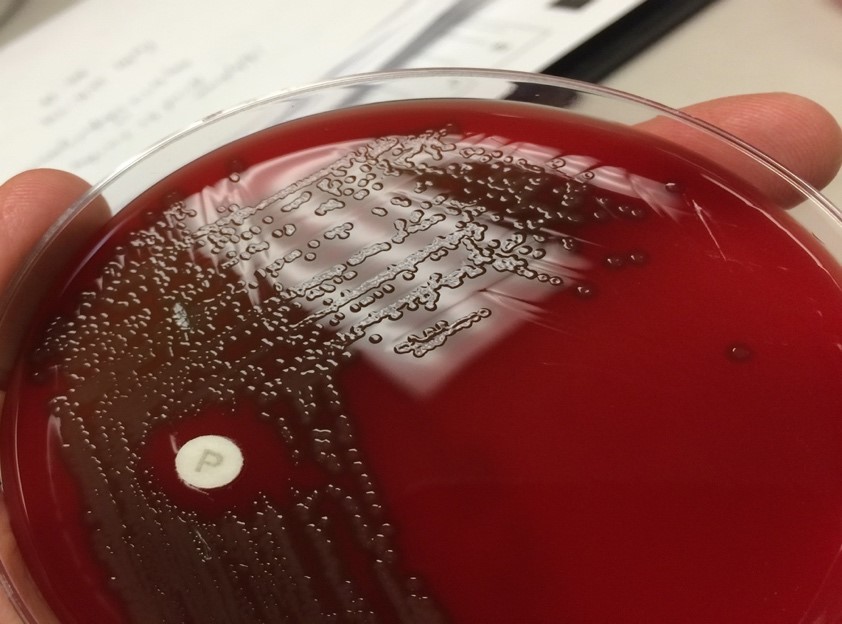
An aerobic culture of drained contents from the brain ultimately grew characteristic alpha-hemolytic colonies with central umbilication (Figure 4) which were subsequently identified by MALDI-TOF and optochin disk as Streptococcus pneumoniae. Admission blood cultures also grew Streptococcus pneumoniae with characteristic “bullet-” or “lancet-shaped” gram-positive cocci in pairs on Gram stain. Fungal and acid-fast bacillus cultures had no growth. Following susceptibility testing; antibiotic coverage was narrowed to IV Penicillin G.
The patient remained unresponsive and required continued intensive medical support. Although blood cultures were sterilized, he continued to have fevers and persistent leukocytosis. Gram stain of ventricular drainage re-demonstrated gram positive diplococci. The patient was transitioned to comfort care and expired on day 5 of hospitalization, the cause of death was sepsis.
Discussion
This case of a brain abscess demonstrates an unusual intracranial complication of Streptococcus pneumoniae. S. pneumoniae (or pneumococcus) is a commensal of the upper respiratory tract (URT) and important opportunistic pathogen. Up to 65% of children and less than 10% of adults are colonized by S. pneumoniae. Dissemination of S. pneumoniae beyond its niche in the nasal mucosa leads to a spectrum of disease including lobar pneumonia, meningitis, sepsis, sinus infections and middle ear infections.1 Local dissemination of S. pneumoniae to the central nervous system (CNS) is the most common intracranial complication of otitis media and mastoiditis. These patients can present with fulminant “otogenic” meningitis. About a third of these cases require myringotomy or mastoidectomy.2 Focal parenchymal brain infection by pneumococcus, however, is uncommon.
This patient presented with signs of mass effect due to a large temporal lobe abscess warranting emergent neurosurgery. Broadly, focal parenchymal brain infections arise either by hematogenous dissemination of organisms or contiguous spread from an adjacent infection. The age, immune status, and any underlying disease present in the patient help predict the pathogen. Brain imaging is also helpful. Hematogenous spread, usually from endocarditis, tends to produce multiple lesions at the grey-white matter junction,3 while direct seeding causes solitary lesions.4,5 In this older patient with a relatively intact immune system and a possible history of intravenous drug use, hematogenous spread of bacteria was considered. However, a large single lesion in the temporal lobe with a plausible adjacent nidus (opacified mastoid air cells) is most consistent with contiguous spread.
A wide range of organisms should be considered when evaluating brain abscesses, though S. pneumoniae is a relatively rare culprit. A meta-analysis of 9,699 patients with brain abscesses found that S. pneumoniae was isolated from only 2.4%.6 The most common organisms were streptococci of the viridans group (34%) and Staphylococcus spp., most commonly S. aureus (18%). Even among patients with otogenic intracranial abscesses, S. pneumoniae is rarely implicated. Interestingly, the pathogen most frequently isolated from otogenic brain abscesses is Proteus mirabilis. 7,8
Once S. pneumoniae was identified, susceptibility testing was required to rule out acquired resistance to beta lactam and cephalosporin antibiotics, which is mediated by altered penicillin-binding proteins (PBPs).9 A more stringent susceptibility minimal inhibitory concentration (MIC) breakpoint applies to S. pneumoniae meningitis than other infections to account for drug distribution into the CNS.10 The hospital antibiogram reports that 97% of S. pneumoniae isolates are susceptible to Penicillin at MICs acceptable for treating non-meningitis infection but only 53% are susceptible at MICs for meningitis. Furthermore, 3.3% of all strains reported in the United States between 2001 and 2005 were also significantly resistant to ceftriaxone.11 This patient was covered by broad spectrum antibiotics until susceptibility testing demonstrated sensitivity to both penicillin and ceftriaxone.
References
1 Weiser, J. N., Ferreira, D. M. & Paton, J. C. Streptococcus pneumoniae: transmission, colonization and invasion. Nat Rev Microbiol 16, 355-367, doi:10.1038/s41579-018-0001-8 (2018).
2 Kaplan, D. M., Gluck, O., Kraus, M., Slovik, Y. & Juwad, H. Acute bacterial meningitis caused by acute otitis media in adults: A series of 12 patients. Ear Nose Throat J 96, 20-28 (2017).
3 Bakshi, R. et al. Cranial magnetic resonance imaging findings in bacterial endocarditis: the neuroimaging spectrum of septic brain embolization demonstrated in twelve patients. J Neuroimaging 9, 78-84, doi:10.1111/jon19999278 (1999).
4 Brouwer, M. C., Tunkel, A. R., McKhann, G. M., 2nd & van de Beek, D. Brain abscess. N Engl J Med 371, 447-456, doi:10.1056/NEJMra1301635 (2014).
5 Miller, J. M. et al. A Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2018 Update by the Infectious Diseases Society of America and the American Society for Microbiology. Clin Infect Dis 67, e1-e94, doi:10.1093/cid/ciy381 (2018).
6 Brouwer, M. C., Coutinho, J. M. & van de Beek, D. Clinical characteristics and outcome of brain abscess: systematic review and meta-analysis. Neurology 82, 806-813, doi:10.1212/WNL.0000000000000172 (2014).
7 Duarte, M. J. et al. Otogenic brain abscesses: A systematic review. Laryngoscope Investig Otolaryngol 3, 198-208, doi:10.1002/lio2.150 (2018).
8 Kangsanarak, J., Fooanant, S., Ruckphaopunt, K., Navacharoen, N. & Teotrakul, S. Extracranial and intracranial complications of suppurative otitis media. Report of 102 cases. J Laryngol Otol 107, 999-1004, doi:10.1017/s0022215100125095 (1993).
9 Chen, L. F., Chopra, T. & Kaye, K. S. Pathogens resistant to antibacterial agents. Infect Dis Clin North Am 23, 817-845, vii, doi:10.1016/j.idc.2009.06.002 (2009).
10 Weinstein, M. P., Klugman, K. P. & Jones, R. N. Rationale for revised penicillin susceptibility breakpoints versus Streptococcus pneumoniae: coping with antimicrobial susceptibility in an era of resistance. Clin Infect Dis 48, 1596-1600, doi:10.1086/598975 (2009).
11 Sahm, D. F. et al. Tracking resistance among bacterial respiratory tract pathogens: summary of findings of the TRUST Surveillance Initiative, 2001-2005. Postgrad Med 120, 8-15, doi:10.3810/pgm.2008.09.suppl52.279 (2008).
–Miles Black, Ph.D. is a fourth-year medical student in the Medical Scientist Training Program at UT Southwestern Medical Center. His background is in enzyme biochemistry and Legionella pathogenesis.
–Denver Niles, MD is the Medical Microbiology fellow at UT Southwestern Medical Center. Prior to his Medical Microbiology fellowship, he completed pediatric infectious disease training at Baylor College of Medicine/Texas Children’s Hospital.
–Dominick Cavuoti, D.O. is a professor of Pathology at UT Southwestern Medical Center who specializes in Medical Microbiology, ID Pathology and Cytology.

-Clare McCormick-Baw, MD, PhD is an Assistant Professor of Clinical Microbiology at UT Southwestern in Dallas, Texas. She has a passion for teaching about laboratory medicine in general and the best uses of the microbiology lab in particular.
