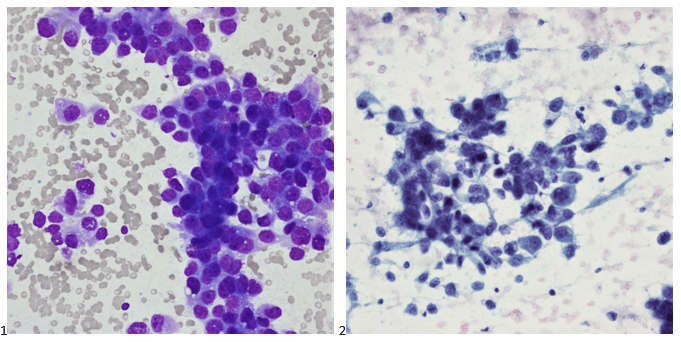
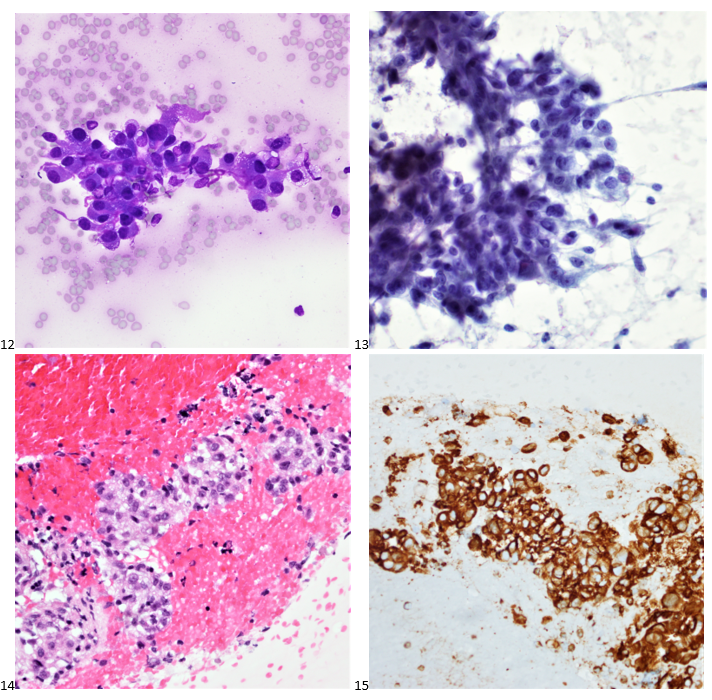
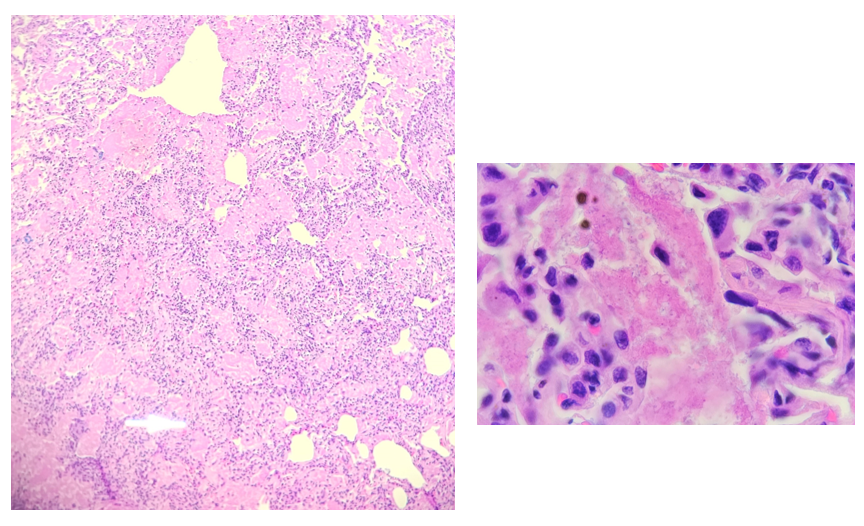
A 61 year old Black woman with a diagnosis of sickle cell beta thalassemia presented to the ER with fatigue, dyspnea, and back and leg pain with some swelling in the hands and feet. Splenomegaly was noted on exam. The patient has a history of moderate to severe symptomatic anemia and is being followed by a hematologist. Her baseline Hgb is 9-10 g/dL. Her treatment plan includes Hydroxyurea, 500 mg daily, and transfusions, as needed. Her last sickle cell crisis was 2 years ago. CBC was ordered. Hgb on admission was 6.1 g/dL. Her RBC morphology showed polychromasia, target cells, sickle cells, anisocytosis, and numerous nucleated RBC forms.
The patient was admitted to the hospital. Type and crossmatch for 2 units of packed red blood cells was ordered. CT imaging was performed and revealed severe osteopenia and vertebrae deformities consistent with her history of sickle cell disease. Chest CT showed hypoinflated lungs and areas of consolidation in the lower lobes consistent with acute chest syndrome due to sickle cell beta thalassemia. She was transfused with 2 units of pRBCs and treated for sickle cell crisis. The patient remained stable and was discharged 3 days later.


Sickle cell anemia (HbSS) and thalassemia are the world’s most common single gene disorders. Both are inherited in an autosomal recessive manner, and result in hemolytic anemias. But what happens when you inherit one gene for sickle cell and one gene for beta-Thalassemia (β-thalassemia)?
Sickle cell disease is caused by mutations in the HBB gene that provide instructions for making beta-globin. Sickle cell anemia is a hemoglobinopathy, a qualitative defect in the structure of globin chains, resulting in the production of abnormal hemoglobin. Normal adult Hemoglobin A has 2 α chains and 2 β chains (α2β2). Hb S results from the substitution of valine for glutamic acid at position 6 of the β globin chain. The resultant Hb S has reduced solubility at low oxygen tensions. Patients with sickle cell anemia have a moderate to severe chronic hemolytic anemia with recurrent painful sickle cell crisis.
Sickle cell disease is inherited in an autosomal recessive pattern from parents who have at least one mutated gene. Anyone with a sickle cell gene can pass this gene on to their children. Sickle cell anemia (HbSS) is the homozygous expression of a sickle gene from both parents and is the world’s most common inherited hematological disease. A heterozygote inherits a sickle gene from only one parent. This person is a carrier of sickle cell (HbSs), often referred to as sickle cell trait. HbSs persons do not generally exhibit symptoms or may exhibit only a mild anemia. However, under stressful conditions, such as at high altitudes, they may experience vaso-occlusive sickle crisis.
While hemoglobinopathies are a qualitative defect due to structural changes in the normal amino acid sequence of globin, thalassemias result from an imbalance in the synthesis of the globin chains that make up the hemoglobin molecule. Thalassemias involve the rate of globin chain synthesis leading to a quantitative defect. Thalassemia is divided into α-thalassemias and β-thalassemia. α-thalassemias involve genes for the α chains on chromosome 16. In α-thalassemia, the deletions involve the α1 and/or the α2 globin genes and result in decreased production of α chains. β-thalassemias mainly affect β chain production. They are disorders of reduced globin chain production from the globin chain cluster on chromosome 11.
(Since this case involves a known diagnosis with a compound heterozygous state involving a β-thalassemia gene mutation, the discussion of α-thalassemia has been limited here. Watch for a case involving α-thalassemia in a future blog!)
Beta thalassemia occurs when the beta globin chains are either produced inadequately or not at all. There are many mutations in and around the β globin gene that result in decreased β chain production. Mutations that result in the complete absence of β chain production are designated as β0. In the most severe form of β-thalassemia the patient is homozygous β0/β0 and does not produce any β chains. Without β chains there is no Hb A (α2β2). β+ is used as the designation for any mutations of the β globin gene that cause a partial deficiency of β chains (5-30% decrease) and therefore result in a decrease in production of Hb A. The βsilent designation is used for carrier state gene mutations that result in only a mildly decreased β chain production. The degree of decrease in the β chain production is related to the degree of anemia and the severity of clinical disease.
Thalassemia, like sickle cell anemia, is a hereditary anemia inherited in the autosomal recessive manner. β-thalassemia is divided into categories based on clinical severity of disease. In β-thalassemia major a child inherits a copy of a β-thalassemia gene mutation from both parents. There are various mutations that cause genes with these mutations and different variants may be inherited from each parent. A person with thalassemia major may be homozygous β+/ β+, homozygous β0/ β0, or the compound heterozygous state β+/ β0. Hb A is only produced in patients with the β+ mutation. β-thalassemia major patients have the most severe hemolytic anemia and symptoms. β-thalassemia intermedia is characterized as homozygous βsilent or heterozygous βsilent with β+ or β0 and mild to moderate disease. β-thalassemia minor, also called β-thalassemia trait or carrier state, presents with mild but asymptomatic hemolytic anemia. These patients are heterozygous with normal β globin and have slightly decreased Hb A.
In people with sickle cell disease, at least one of the beta globin is replaced with hemoglobin S. In homozygous sickle cell anemia, both beta globin subunits in hemoglobin are replaced with hemoglobin S. In compound sickle cell diseases, one beta globin is replaced with hemoglobin S and the other beta globin is replaced with a different abnormal variant. Examples of this are Hb SC disease, and Hb SD syndrome. Compound heterozygosity is the inheritance of two different mutated genes that share the same locus. If mutations that produce hemoglobin S and beta thalassemia occur together, individuals have hemoglobin S-beta thalassemia disease. (sickle cell beta-thalassemia, Hb S β thal or sickle-β-thal). Sickle cell beta thalassemia patients have hemoglobin S (α2β26Glu→Val) and either β0 or β+.
When a qualitative hemoglobinopathy is inherited with a quantitative disorder of hemoglobin synthesis, the severity of the compound disorder is dependent on the β gene mutation. Patients with β0 produce no Hb A and have moderate to severe symptoms comparable to that of Hb SS patients. β+ patients will produce some β chains and therefore have some Hb A and milder or no symptoms.

Newborn screening can diagnose β0-thalassemia at birth by detecting a complete absence of hemoglobin A. However, it is not possible to make a definitive diagnosis of β+-thalassemia in the newborn because newborns have Hb F, and the reduced amount of hemoglobin A overlaps the range for normal babies. In adults with Hb S – β thal the amount of Hb S is variable. There is some Hb A in β+ patients but no Hb A detected in β0. Hb A2 and Hb F are increased. In addition to hemoglobin electrophoresis, molecular testing may also aid in the diagnosis by identifying genetic mutations. Beta globin gene sequencing can identify beta thalassemia alleles that are caused by point mutations in the beta globin gene. As well, structural variants of the beta globin gene such as Hb S can be identified with this technique. This can lead to a better understanding and clinical management of the disease.
Case Study, continued: This patient inherited a Hb S gene from one parent and a β-thal gene from the other, resulting in sickle cell beta thalassemia. This compound heterozygosity affects red blood cells both by the production of structurally abnormal hemoglobin, and by the decreased synthesis of beta globin chains. Clinical manifestations depend on the amount of beta globin chain production. Symptoms may include anemia, vascular occlusion, acute episodes of pain, acute chest syndrome, pulmonary hypertension, sepsis, ischemic brain injury, splenic sequestration crisis and splenomegaly.
Hemoglobin electrophoresis was sent out to a reference lab and results are shown in Table 2. Based on the Hemoglobin electrophoresis, is this patient Hb S- β0-thal or Hb S- β+-thal?

Hemoglobin electrophoresis results for Hb S beta thalassemia patients are expected to show 60-90% Hb S and 10-30% Hg F. This patient’s Hgb S at 74% is within this range. This result reflexed a sickle solubility test, which was positive. As well, the elevated Hb F and Hb A2 are consistent with this diagnosis. It was noted in the discussion above that Hb S- β+-thal mutations cause a decrease of 5-30% in beta chains and therefore a decrease in Hb A. This patient’s Hb S is greater than the Hb A and her Hb A concentration is 14.8%, which is consistent with this diagnosis. Hb S- β0 mutations produce no Hb A. In this case there is some Hb A on electrophoresis but not as much as would be expected in a β+-thal mutation. Also, of note it that this patient was recently transfused with 2 units of pRBCs. Interpretations of hemoglobin electrophoresis assume that the patient has not been transfused in the last 3 months. The Hb A in this patient can be explained by these recent transfusions. Therefore, it can be concluded that the hemoglobin pattern and concentrations are consistent with transfusion of a Hb S beta0 thalassemia patient. The β0 mutation is also consistent with this patient’s moderately severe symptomatic anemia.
References
- Keohane, Elaine, et al. Rodak’s Hematology, Clinical Principles and Application, 5th ed, Elsevier, 2016
- McKenzie, Shirlyn. Clinical laboratory Hematology. Pearson Prentice Hall, 2004.
- McGann PT, Nero AC, Ware RE. Clinical Features of β-Thalassemia and Sickle Cell Disease. Adv Exp Med Biol. 2017;1013:1-26. doi: 10.1007/978-1-4939-7299-9_1. PMID: 29127675.
- Origa R. Beta-Thalassemia. 2000 Sep 28 [Updated 2021 Feb 4]. In: Adam MP, Mirzaa GM, Pagon RA, et al.,editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1426/
- Turgeon, Mary Louis. Clinical Hematology Theory and Procedures, 6th ed, Jones and Bartlett Learning, 2017.

-Becky Socha, MS, MLS(ASCP)CMBBCM graduated from Merrimack College in N. Andover, Massachusetts with a BS in Medical Technology and completed her MS in Clinical Laboratory Sciences at the University of Massachusetts, Lowell. She has worked as a Medical Technologist for over 40 years and has taught as an adjunct faculty member at Merrimack College, UMass Lowell and Stevenson University for over 20 years. She has worked in all areas of the clinical laboratory, but has a special interest in Hematology and Blood Banking. She currently works at Mercy Medical Center in Baltimore, Md. When she’s not busy being a mad scientist, she can be found outside riding her bicycle.