Case History
A 56 year old male presented to his PCP complaining of sinus congestion, rhinorrhea, night sweats, decreased appetite and fevers of up to 101-102 every evening. Hematologic evaluation revealed a neutropenia and a lymphopenia. An infectious disease work up was negative. His LDH was elevated. Physical examination reveals an enlarged left axillary lymph node. An excisional biopsy was performed.
Biopsy Findings

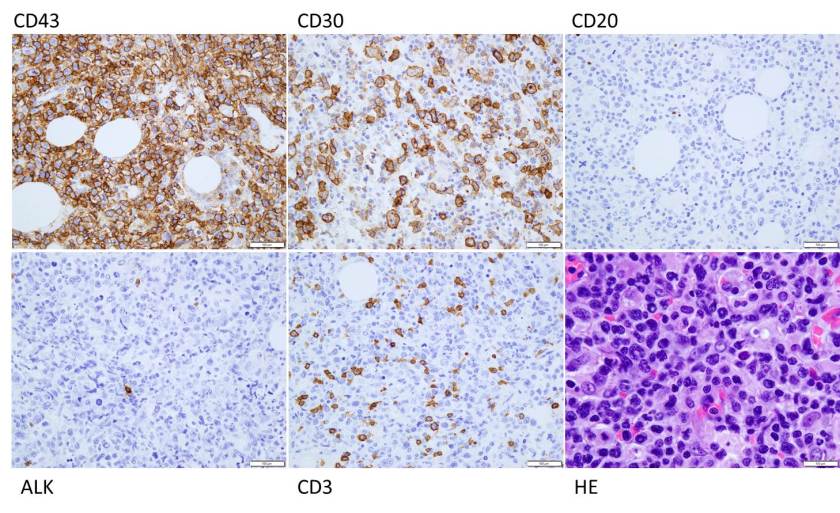
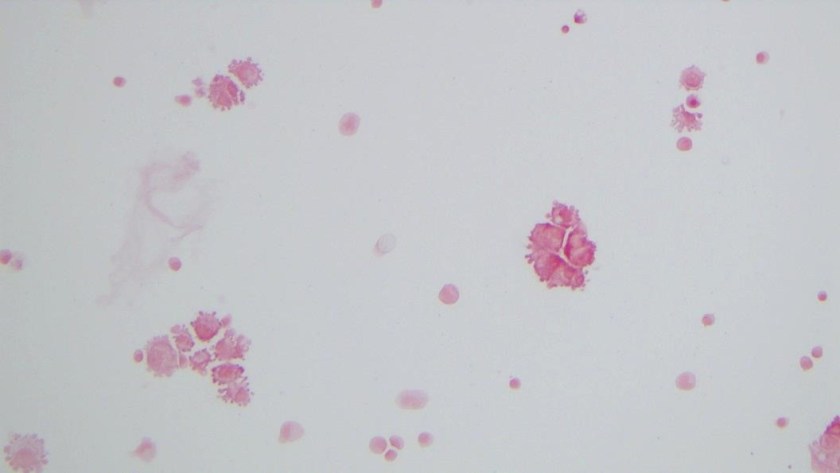
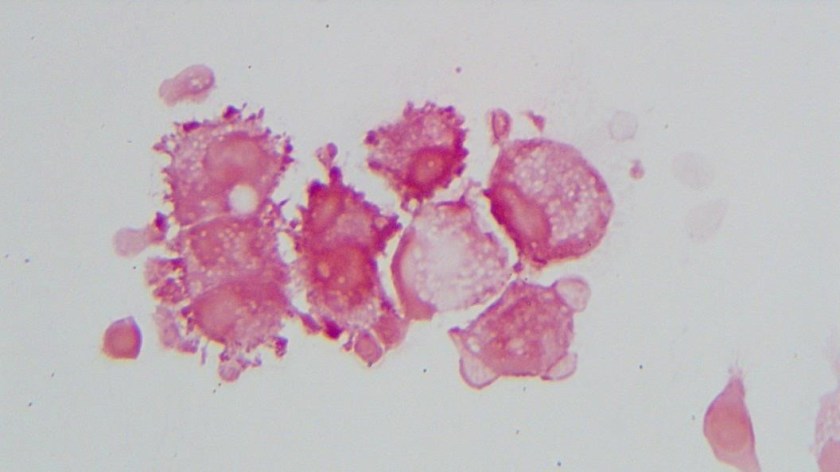
H&E stained sections demonstrate an enlarged node with effaced architecture and scattered residual follicles with small, mature cells. There is a proliferation of intermediate to large, to very large, atypical and highly pleomorphic cells many of which demonstrate bizarre forms, irregular nuclear morphology and acidophilic nucleoli. The lymphoma cells are noted to focally traverse through adipose tissue. Occasional hallmark cells are appreciated.
To further characterize the infiltrate, immunohistochemical stains were performed and interpreted with appropriate controls. The lymphoma cells were diffusely positive for CD45 (LCA), CD43, and CD30 (membranous and Golgi) with a Ki-67 of 80-90%. These cells were negative for CD20, PAX-5, CD3, CD4, CD8 (mostly), CD5, D10, BCl-2, BCl-6 and ALK1.
The morphologic features and immunophenotype of the cells was diagnostic of anaplastic large cell lymphoma, ALK negative.
Discussion
Anaplastic Large Cell Lymphoma (ALCL), ALK-negative (ALK-) is defined as a CD30+ T-cell neoplasm that morphologically resembles ALK-positive ALCL, but lacks ALK protein expression. It most commonly affects adults (aged 40-65 years), and has a slight male preponderance with a male-to-female ratio of 1.5:1. T. Most patients present with advanced disease (stage III-IV), lymphadenopathy and B symptoms. The most common differential diagnosis is ALK-positive ALCL.
The molecular deciphering of ALCL began in the 1990s with the discovery of a recurrent t(2;5) (p23;q35) translocation fusing the ALK gene and the nucleophosmin gene generating a NPM-ALK fusion protein, as well as other ALK translocations resulting in a high ALK kinase activity. This triggers the major oncogenic pathway in ALK-positive ALCL. Pharmacologic therapy has been developed to target ALK, and has shown efficacy. Thus, compared with ALK-negative cases, ALK-positive occurs in younger patients and has a better prognosis. ALK-negative ALCL also tends to involve both lymph nodes and extranodal tissues, although extranodal sites are less commonly involved than in ALK+ ALCL.
The other differential diagnoses of ALK- ALCL includes, primary cutaneous ALCL (C-ALCL), other subtypes of CD30+ T-cell or B-cell lymphoma with anaplastic features and classic Hodgkin Lymphoma. If a single lymph node or cutaneous cases are suggestive of ALK- ALCL, C-ALCL needs to be considered. Any cases that involve the gastrointestinal tract need to be distinguished from CD30+ enteropathy-associated and other intestinal T-cell lymphomas.
Molecular analysis of ALK- ALCL shows characteristic strong expression of CD30, in equal intensity in all the cells. Loss of T-cell markers is frequently seen, however, more than half of all cases express one or more T-cell markers. CD2 and CD3 are more commonly expressed than CD5, and CD43 is almost always expressed. CD4+ is frequently positive, while CD8+ is rare. Many cases also express cytotoxic markers TIA1, granzyme B, and/or perforin.
The genetic profile in ALK-negative ALCL has been found to be pretty heterogenous. Most notably, activating mutations of JAK1 and/or STAT3 have been shown to lead to activation of the JAK/STAT3 pathway. Chromosomal rearrangements of DUSP22 (i.e. chromosomal rearrangements in or near the DUSP22-IRF4 locus on 6p25.3) occur in 30% of the cases, and rearrangements of TP63 occur in about 8% of cases. Neither of the rearrangements have been reported in ALK+ ALCL.
From a prognostic standpoint, studies have shown that the rearrangements have effects on the survival rate. TP63-rearranged cases were shown to have an unfavorable prognosis worse than ALK- ALCL with neither rearrangement, while DUSP22-rearranged cases were shown to have favorable outcomes similar to ALK-positive ALCLs.
References
- Gaulard P, de Leval L. ALK-negative anaplastic large-cell lymphoma. 2016 Jan 14;127(2):175-7.
- Edgardo R. Parrilla Castellar et al., ALK-negative anaplastic large cell lymphoma is a genetically heterogeneous disease with widely disparate clinical outcomes Blood. 2014 Aug 28; 124(9): 1473–1480.

-Brandon Zelman is 4th year medical student at the Philadelphia College of Osteopathic Medicine and an aspiring pathologist. You can follow Brandon on Twitter @ZelmanBrandon.

-Kamran M. Mirza, MD PhD is an Assistant Professor of Pathology and Medical Director of Molecular Pathology at Loyola University Medical Center. He was a top 5 honoree in ASCP’s Forty Under 40 2017. Follow Dr. Mirza on twitter @kmirza.