Case History
A 72 year old female with past medical history of stage IV ovarian adenocarcinoma treated with chemotherapy and interval debulking surgery, presented to emergency room with a one week history of confusion and worsening balance.
CT scan of the head showed new communicating hydrocephalus. A magnetic resonance imaging couldn’t be performed initially because of patient’s uncontrolled agitation. Lumbar puncture (LP) was performed. Following this procedure the patient’s mental status showed some improvement and therefore neurosurgery team decided to insert ventriculoperitoneal (VP) shunt to treat her hydrocephalus and prevent recurrence of seizures.
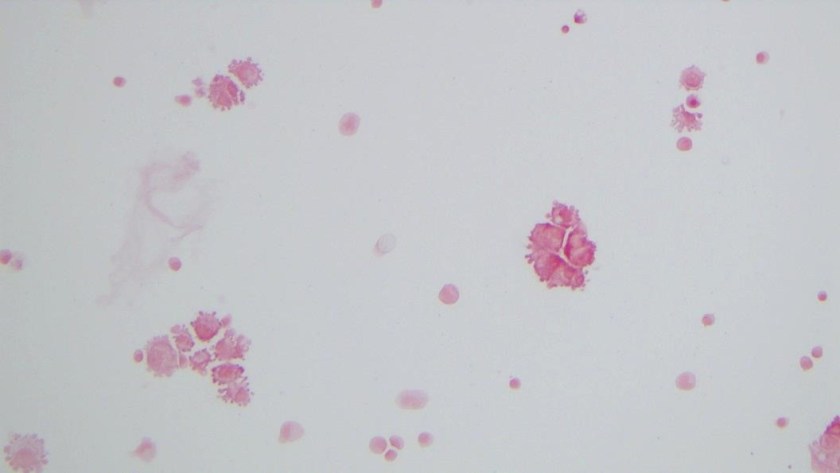
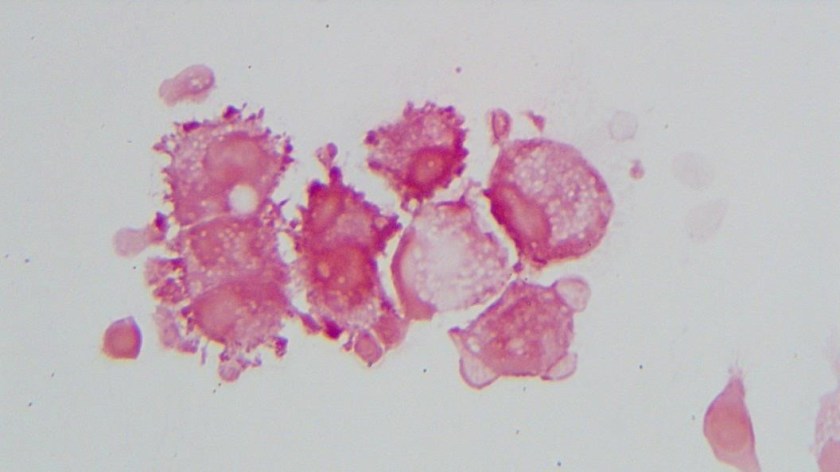
It was Friday afternoon when a microbiology technologist brought the patient’s cerebrospinal fluid (CSF) gram stain to be reviewed. It was confirmed that no inflammatory cells and organisms were present. However, cells in the background looked very atypical (Image 1a, b).
Discussion
The gram stain is used to provide preliminary information about the microorganism present in the specimen. Gram stain differentiates bacteria into two fundamental varieties of cells. Bacteria that retain the initial crystal violet stain (purple) are said to be “Gram-positive,” whereas those that are decolorized and stain red with carbol fuchsin (or safranin) are said to be “Gram-negative” (1). An adequate examination of a gram-stained smear includes observing numerous representative fields and the fields containing neutrophils yield the most information (2). Gram stain provides information about number of bacteria present, gram reaction and shape of the bacteria. In background we can also see epithelial cells and inflammatory cells. However, it’s a good practice to also appreciate and investigate any odd looking findings.
To investigate further, we visited the hematology laboratory to view their CSF slide to determine if these cells were a processing artifact. After it was confirmed that hematopathology saw the same atypical cells, a cytopathologist was requested to review the gram stain since patient’s CSF cytology specimen was to be processed after the weekend. Cytopathologist favored our suspicion and decided to process the cytology specimen late in the day on Friday and it was confirmed that those atypical cells were consistent with the metastatic adenocarcinoma.
Neurosurgery team was immediately contacted to reconsider insertion of the VP shunt as the shunt would drain fluid from the CSF into the peritoneal cavity and thus there was concern for transferring of malignant cells from central nervous system into abdomen/pelvis. However, after consulting oncology team it was later decided to proceed with the procedure since patient’s primary tumor originated in abdomen/pelvis and current intraabdominal tumor burden was not significant as compared to the symptoms driven by CNS involvement. Proceeding with this procedure was considered to be palliative and the best course of action to improve the patient’s quality of life.
References
- Beveridge TJ. Use of the gram stain in microbiology. Biotech Histochem.2001 May;76(3):111-8.
- Barenfanger J, Drake C. Interpretation of gram stains for the nonmicrobiologist. 2001 July;32(7):368–375.

-Kiran Manjee, MD, is a 1st year anatomic and clinical pathology resident at University of Chicago (NorthShore).

-Erin McElvania, PhD, D(ABMM), is the Director of Clinical Microbiology NorthShore University Health System in Evanston, Illinois. Follow Dr. McElvania on twitter @E-McElvania.