Hi everyone, and welcome back!
I’ll get straight to the point this time: there’s a legitimate crisis happening in our world that is highlighting a multitude of things. Just to name a of few: the complex intersection of state-sponsored healthcare as a human right, the availability of resources to clinical personnel, the logistics and implications of public health measures to the average person, our global connectivity, and lack of damn tests!
And that’s our stop. Please take a minute to locate your exits, as they may be behind you, and get ready to talk about this SUPER CRITICAL aspect of the COVID-19 pandemic. What are clinical tests?
I know, this “viral” topic right now will be shared and spread to a vast array of audiences. So, I’m going to try something new here. The last four years of posts on this blog have been aimed somewhere between laboratory professionals and working clinicians. I open a rare window into our clinical world from time to time, but this pandemic is something affecting everyone. Today, we’ve got a major problem about what’s being said in the media and who understands which parts of it…so, as I discuss some major points below, I will take short pauses to highlight clinically relevant information as well as put things in plain, simple terms for all of us to get up to speed together.
(If you are one of my clinical colleagues, from the bench to the bedside, your notes will be in BLUE. If you are not in the healthcare world, or just want some simple clarification, your notes are going to be GREEN. Let’s run an example so you can see what I mean…)

Last month I talked about the background and biology of the SARS-CoV-2 virus as a novel, emerging pathogen and potential pandemic and discussed how we must contextualize these epidemics with proper understanding of data and statistics. Numbers are important and if we’re going to combat a super bug, we’ve got to know what it is, how it acts, where it came from, and what’s happening in the field. A subsequent and fantastic post right after mine by Dr. SoRelle at UTSW, discussed how to validate a COVID-19 test in your laboratory. Basically, he presented a snapshot of the current climate of academic hospital labs now depending on LDTs to respond to the pandemic and discussed where to read more, what limitations testing might have, and how to address things like designing/purchasing primers, cross-reactivity, detection capabilities, and EUAs under the new FDA guidance.

But back to business, this month we’re talking about testing, and if you just google search “COVID-19” in the states, you’re met with a myriad of failure stories…
“As US fumbles COVID-19 testing, WHO warns social distancing not enough” (source)
“Limited access to COVID-19 tests frustrates patients, health professionals” (source)
“What went wrong with coronavirus testing in the US” (source)
“Why coronavirus testing in the US is so delayed” (source)
“America’s shamefully slow coronavirus testing threatens all of us” (source)

I’ll stop torturing you. You’re forced to watch and/or read enough news as it is nowadays (you’re social distancing, right? …right?) I mean, even Norway took a jab and called us “underdeveloped” citing our poorly developed healthcare delivery system—ouch. So, what’s the big problem with testing? What stark revelation exists between all these different news sources and highlights our “laboratory failures” in America? Well, luckily nearly all of these lab-fail stories aren’t aimed at our labs at all, they’re aimed at governmental decisions and red tape traps. But that’s hard to get to in a sea of clickbait. So instead of giving you more rabbit-hole lab talk, let’s talk about two main tenets of this narrative that everyone should be aware of: what laws affect lab tests and what does it mean to create a new lab test? After we clear the air for those two key points, we’ll come back around and tell you what COVID-19 looks like in the medical laboratory.
The Law of the Lab
There are SO MANY regulations and accrediting bodies that govern the way labs operate, deliver results, and function in clinical settings. Let’s name some! Various federal, state, and local laws ensure the highest calibers of safety and accountability for clinical and research laboratories; laws from OHSA, the FDA, recommendations from the CDC and local health departments; regulations for mechanical, noise, chemical, exposure, pathogen/infectious safety, pollution, radiation, blah blah blah! Trust me, there are a ton. What feels ever present to us laboratorians are the accrediting and inspection agencies that grant us authority to continue performing our clinically vital roles. Joint Commission, FDA, and other inspections make sure hospitals are up to snuff. Laws that govern what labs can do what tests fall under CLIA. They help us define high complexity to waived (not complex) testing. There’s a massive difference in a clinical office’s medical assistant obtaining a point of care (POCT) glycated hemoglobin (Hgb A1c) than a medical laboratory scientist conducting a hemoglobin electrophoresis study, and that difference highlights specialized training, laboratory ability, safety and quality requirements.

Accreditations and recommendations can come from many professional societies like the CAP, ASCP, AABB, AACC, ASM, ASH, and many subspecialized groups. Laboratorian trigger warning: I’m a certified CAP inspector. No; you don’t have to put away your desktop coffee…for now. But I’ve certainly—along with all of you—been wholly immersed in the endless accreditation and regulation protocols we follow. We know exactly why these regulations exist and we should talk more about them, since they define our role as such a critical one.
Feeling Validated Yet?
Some of you lost a breath when I said CAP inspector. The rest of you may now spit out your coffee and run for the hills because I’m talking about VALIDATIONS. Rotating MLS students sometimes do their capstones or theses on validating a test or instrument, but we do them because that’s just a part of our job! So what goes into validating a test? The short answer: a whole lot. The long answer: a whole lot more! Let’s take PCR testing for example, it’s topical because we’re talking about COVID-19 testing and that’s the prime modality so let’s explore that validation.

So, you want to add a qPCR to your lab’s test menu, eh? You first have to decide quantitative or qualitative. If its quantitative you’re in for a treat—yay statistical variant analysis! You have to prove applicability, practical application, specificity, sensitivity, efficiency, correlatable translation into useful clinical data. Detecting a number or degree of mutations? Make sure its accurate and precise (and repeatable). Buying commercial kits or making your own in-house assay? What about a qualitative test, just a simple yes or no. Easier right? Not really, you need to prove most of the same data and statistical soundness depending on methodology used, instrumentation, nature of variation, etc. AND: it all must be documented, proven, up to regulatory standards, and you better have a database FULL of data that proves your test works. Now, I have good news and bad news.
The good news: this is pretty routine, and pretty easy to follow even for simple new tests your incorporating from commercial vendors with ready-to-validate test kits. The bad news: COVID-19 is not this type of test. Say what you will about the US’s decision to NOT utilize the WHO testing model available weeks ago, the CDC responded in kind here by providing a two-control kit that SLOWLY started to break out. Using this as a model, many labs in academic hospital pulled the trigger on their LDT work. (LDT = laboratory developed tests) I’ve already revealed myself as a CAP inspector (pshh, not even my final form!) and I can tell you that there are hundreds of citations for how to validate already existing tests, compare tests across different instruments, compare them across the same instrument, prove your statistical ability to ensure quality results, and even how to make your own test from scratch! Talk about extra work!


Image 5. [MLS]: You’re a good analyzer, a smart analyzer. We all appreciate your efforts and you’ve really contributed to a positive work environment. We appreciate you, so we got you these reagents. They’re nice and fresh, what do you think? [Instrument]: *BEEP* *BEEP* *ERROR* *MORE VALIDATION REQUIRED*, [MLS]: okay, okay, have you lost weight?…**RESULTS VERIFIED** (Image source)
Running COVID-19 testing alongside B. burgdorferi: Corona…with Lyme?
Now we’ve come to the heart of the current discussion. Dr. SoRelle talked about validating a COVID-19 assay last month, so I’m letting him take point on that—read his piece, its great! What we’re talking about today is what exactly regulations and validations have to do with COVID-19 testing. Remember the regulations that say how laboratories run tests and operate clinically? Okay, well there are issues with this narrative. You’ll see plenty of news stories about the regulatory red tape that has hindered lab professionals from providing COVID testing. This makes it sound like because of “rules” we just can’t give them out to everyone, or we can’t set up testing for whatever regulatory reason. This is not the case. This is how the relationship between those aforementioned regulatory bodies work. The FDA makes sure labs operate up to federal standards and, and specifically, enforce regulations outlined in CLIA. While the FDA would like to call tests/results products of analyzers/devices to fall in their prevue, the interpretation and translation of that clinical data is not a print out but a MEDICAL SPECIALTY—hi, I’m pathologist #2465827, part of the FDA regional lab result response team—nope. Lab developed tests (LDTs) are not FDA products, but CLIA certified processes that only CLIA certified laboratories can perform. No CLIA no LDT—it’s as simple as that. You now understand that a lot goes into a single test, and that not every lab can perform high-complexity testing, thus, not every lab is CLIA certified to do LDTs… which means no COVID testing for them.

Okay, the other half of the narrative says that because of a lack of supplies from industry vendors, commercial kits were unavailable either from them or the CDC and if they got them, their reagents were not good. Frustrated, scorned immunology/chemistry lab specialists decide, “we’ll show them…we’ll make our own test! *evil laugh*” A bit dramatic, because it is dramatic. That’s not how LDTs work. How, then, do we get to LDTs? That answer is simple: regardless of politics, resources, or any other social concerns, medical laboratorians and clinical pathologists are part of the essential healthcare team that strives to care for the sick and hopes to prevent unnecessary infection or illness. We’re in this together and if one system can’t provide tests for the whole country, hundreds of thousands of hospitals will rise to the occasion and share partial LDTs or create their own. Just like with any other test, labs know full well how to get this done and work together to do it.
But what about the stories that say some tests take 8 hours vs others that take 4 days to turn around results? How come some kits came with 2 swabs, and some with 3? Why did other countries like South Korea do a lot better than us in testing their population? Did we wait too long to set up LDTs? Can’t I just go to my doctor and get tested now? Okay, okay, I hear you. This is where it gets heavy, especially in the media. The takeaway can be a simple comparison: look back to the Zika epidemic. Zika is an Arbovirus/Flavivirus that we knew about for roughly 60 years. As such, we had time to understand it and create tests quickly that measured exposed patients’ antibody response to acute or resolved infections. Even simpler, Zika is part of a family of mosquito-borne illnesses that we generally understand pretty well. SARS-CoV-2 is not like Zika. The word “novel” is used because it’s just that: brand spankin’ new. And, even though it’s a cousin of its predecessor in the old SARS epidemic, we’ve been racing the clock to create accurate and reliable testing. That’s why there’s so much variability happening across commercial industry to academic hospital laboratories.


Pandemic Proportions
I love to make puns but let me be clear: this is a serious global pandemic, as defined by the CDC and WHO and we must understand this fully and appropriately. We’re still in the dark about a lot of data, mostly because of missing numbers. What we do know continues to inform our daily-changing climate of public health awareness in the US. The following images speak for themselves and I’ve collected them as a resource and snapshot for you to look through, just to get your SARS-CoV-2 bearings.

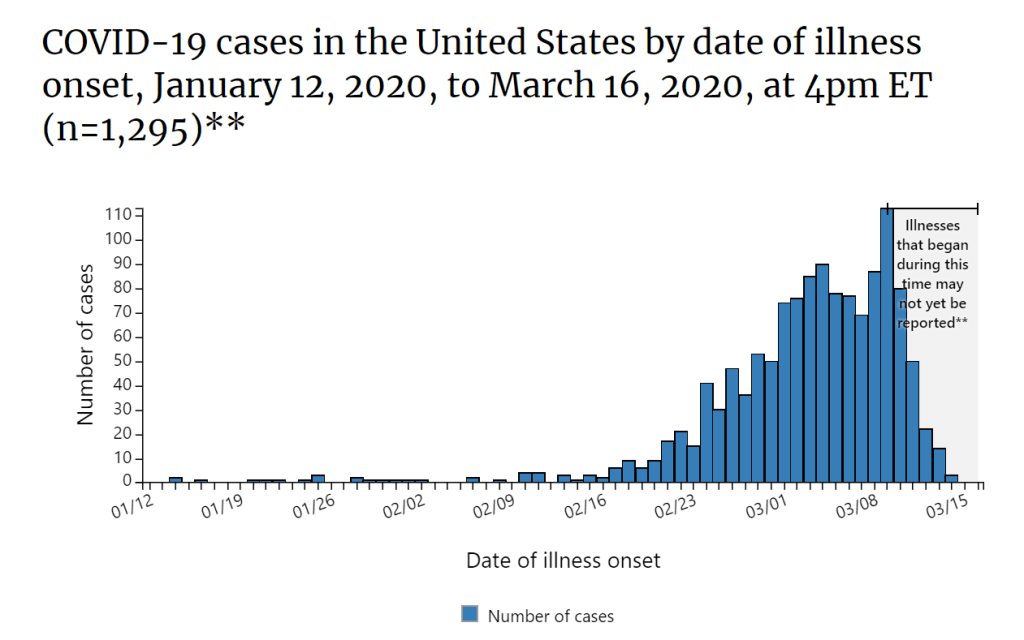





What can I say? It’s hard to write a conclusion about something that’s changing every day. I’ve been doing my best editing to keep this as up to date as it could possibly be for publication, but there are a million more resources sitting in the queue that I wish I could walk you all through. But I think, at the end of the day, what we need to remember is one simple thing: we’ve been here before and we’ll be here again. Public health epidemics and our responsiveness is one of the more serious aspects of laboratory medicine and pathology, not to mention healthcare at large. I said it last month that we’re lucky that this isn’t an influenza-type virus, because that would look a lot different. There are still some highly serious infectious etiologies out there more lethal than COVID-19, but this pandemic is bringing to light a lot of public health awareness and an opportunity to examine our population’s medical/health literacy, our policy priorities, our strength and resolve, as well as our ability to adapt.



In the coming days, weeks, months you’re going to most likely hear about municipal infrastructures cutting back on things in order to prevent the spread of the virus. Listen to these warnings and heed the advice of the medical community. If you think you’re being a hero by braving the storm, you’re no better than a mosquito that carries malaria with no symptoms itself. Wash your hands, practice appropriate social distancing, continue your daily life with your necessary trips out being mindful of what those of us in healthcare live with on a daily basis. Practice compassion as well, just because we’re social distancing to “flatten the curve” doesn’t mean its time for martial law and cleaning supply hoarding. I know these are serious times, but humor and compassion can be infectious too.
Thank you for reading.
Please remember to follow the most updated, verified, and trusted sources on this starting with the CDC and your local public health organizations.
Wash your hands. Change your clothes. Minimize exposures. Take time to reflect and keep as close to your normal routine as possible while remembering things will get better.
If you have specific questions, comments, or concerns leave them in the comments below. Feel free to contact me anytime, and follow my social media (@CEKanakisMD on Twitter) for more/updated material.
Take care, see you next time!

-Constantine E. Kanakis MD, MSc, MLS (ASCP)CM is a new first year resident physician in the Pathology and Laboratory Medicine Department at Loyola University Medical Center in Chicago with interests in hematopathology, transfusion medicine, bioethics, public health, and graphic medicine. His posts focus on the broader issues important to the practice of clinical laboratory medicine and their applications to global/public health, outreach/education, and advancing medical science. He is actively involved in public health and education, advocating for visibility and advancement of pathology and lab medicine. Watch his TEDx talk entitled “Unrecognizable Medicine” and follow him on Twitter @CEKanakisMD.