Case History
A previously healthy 14-year-old female presents to the emergency department with three days of progressive facial and neck swelling. The swelling started on the left side. Two days ago she visited her primary care physician where she had negative monospot and mumps IgM testing. She is fully vaccinated, but was exposed to a mumps outbreak at school.
Discussion
Our patient was diagnosed with mumps by positive RT-PCR from a buccal swab. The mumps virus is a member of the Paramyxoviridae family which includes notable human pathogens parainfluenza, Hendra, and Nipah viruses. Members of this family are enveloped, helical viruses with single-stranded, non-segmented RNA genomes with negative polarity. Mumps is an obligate human pathogen that replicates in the epithelial cells of the upper respiratory tract and subsequently moves to regional lymph nodes. It is spread from person to person via direct contact with respiratory secretions or contact with contaminated fomites. Mumps is a highly contagious disease with as high as 85% of naïve individuals becoming infected after contact with a mumps infected individual. It spreads most efficiently in areas where there is close contact among individuals for prolonged periods of time such as college campuses and close-knit religious communities.
Prior to vaccination for mumps in the 1960s, greater than 150,000 cases of mumps occurred each year in the US. The incubation period for infection is 16-18 days, with the majority of infected persons being asymptomatic or having mild respiratory symptoms. Orchitis causing sterility in post-pubescent males is the main concern of mumps infection but other rare but serious complications include mastitis and oophoritis in females, meningoencephalitis, pancreatitis, and deafness.
Due to sporadic outbreaks of measles since the introduction of the vaccine, the vaccine schedule has been revised from one dose of the MMR (measles, mumps, and rubella) vaccine at age 12-15 months to include another MMR booster at age 4-6 years. We are currently in the middle of yet another outbreak with nearly 6,000 cases of mumps reported to the CDC in 2016 and a high rate of infections reported thus far in 2017 (Figures 1 and 2).
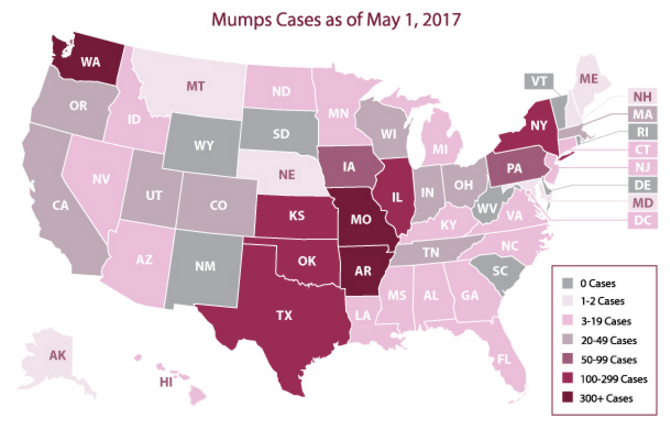
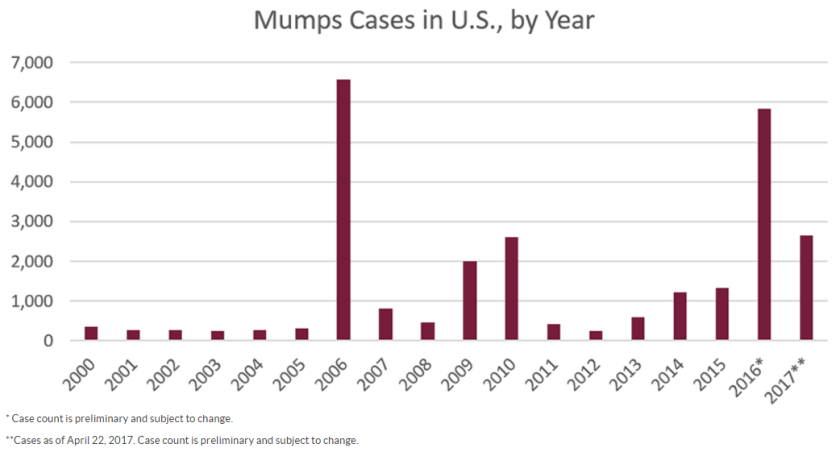
(Figure courtesy of the CDC Mumps website at https://www.cdc.gov/mumps/outbreaks.html. Content source: National Center for Immunization and Respiratory Diseases [NCIRD], Division of Viral Diseases)
Diagnostic Testing for Mumps
Serological testing for IgM and RT-PCR from a buccal swabs are the mainstay of mumps diagnosis. IgM becomes positive in the first 3-4 days after symptom onset and will remain positive for 8-12 weeks. IgG becomes positive 7-10 days following symptom onset and will remain at high levels for many years and detectable for life. In a vaccinated individuals, IgM testing has less utility as it may be non-reactive or weakly positive following a secondary immune response.
RT-PCR from a buccal swab specimen is the most sensitive test for diagnosis of mumps. It should be performed as soon as a patient is symptomatic, as testing by this method is the most sensitive in the first few days following symptom onset and becomes less sensitive as time goes on.
Urine specimens can be used to isolate mumps in viral culture. Urine is not positive for mumps until greater than 4 days post symptom onset and is less sensitive than PCR performed on the bucal swab. For these reasons, viral isolation from urine is no longer a commonly used test for diagnosis of mumps, although viral culture is still considered the gold standard for mumps conformation.
Resolution
The patient and her family were counseled on the infectious nature of mumps. She was instructed to remain in isolation at home for 6 days after resolution of swelling.
References
- Manual of Clinical Microbiology, 11th edition
- CDC Mumps Website (www.cdc.gov/mumps/index.html)

–Erin McElvania TeKippe, PhD, D(ABMM), is the Director of Clinical Microbiology at Children’s Medical Center in Dallas Texas and an Assistant Professor of Pathology and Pediatrics at University of Texas Southwestern Medical Center.




