Hello again everyone and welcome back!
If you caught my previous post it was a rather long twofer for my March rotation at the Mayo Clinic’s Department of Pathology and Laboratory Medicine as well as a great case study of a patient’s therapy-related AML in the setting of Li-Fraumeni Syndrome. Now, as I inch closer and closer to my last months of medical school, I’m doing another pathology clerkship at Danbury Hospital in southwestern Connecticut. It’s an excellent community-based pathology program with a great staff. As you’ve read in my posts before, community hospitals are no stranger to the leading edge of laboratory innovation. A fellow medical scientist in a recent ASCP membership video on social media said, “Laboratory medicine is at the precipice of change.” As a beacon of translational medicine, labs turn routine medical unknowns into answers. Often, they act as leaders in the lab medicine community because of certain population-specific testing and reporting that goes on at their institutions. You might recall my discussion of Bronx-Care Hospital leading the charge in New York City with the newest, 5th generation troponin testing or my experiences at Swedish Covenant Hospital in Chicago with lab automation, software innovation, and CQI.
But back to medical school: the coordinators of this rotation asked be about my interests in pathology and we discussed my past as a medical laboratory scientist. As such, they offered some special projects for me to be a part of in their lab! Specifically, and in addition to my subspecialty objectives and observership, I’ve been helping them with three small projects. First, I assisted in calibrating a freshly validated second Bio-Rad Bioplex2200 analyzer to correlate to a second instrument for some very interesting testing. Second, I’m helping gather ongoing inter-instrument data for a Sebia serum protein electrophoresis instrument. And finally, I’ve been assisting the histopathology section with cross-instrument validation of immunohistochemical stains as well as gathering data for the validation of a great new IHC that would replace PSA. All of these have both used skills from my MLS foundational experience but taken that one step further into the scope of a pathologist by going over the clinical implications and testing outcomes provided by these analyses.
The Syphilis Shuffle
I already mentioned that Danbury’s lab uses the Bioplex2200, so let me tell you a bit about the analyzer and a bit more about an interesting way to do syphilis testing. It’s an interesting immunoassay instrument that uses something called “multiplex” technology. I won’t go into the details about footprint, throughput, and timing because, well, I’m not exactly expecting any Bio-Rad checks over here. But what I do want to talk to you about is the testing methodology. So, what exactly is “multiplex” testing. Basically, by using magnetic beads with fluorescent dyes associated with various tests you can create bead-set assays. Those beads are coated with detection proteins for each assay and exist in a single reagent pack. That allows numerous analytes to be detected at the same time from a single aspirated specimen. A laser in the instrument detects these immunologic events and reports the values. Now, in my experience with immunoassays for tests like syphilis, hepatitis, EBV, or HIV, there are usually honorable mentions of Abbott’s ARCHITECT and Siemens ADVIA Centaur alongside the Bioplex2200. I’m much more familiar with the former two, so getting a chance to work with the latter was great. But what did I learn?


First of all, other analyzers like the ADVIA Centaur use chemiluminescent immunoassay technology and the Abbott ARCHITECH use photometric, potentiometric, or turbidimetric detection for immuno and chemistry tests. There are published demonstrations of how the three instruments compare regarding various metrics and detection statistics. One of those papers from 2017 demonstrated the sensitivity and specificity for HIV testing across the analyzers. Overall, it said the ARCHITECT was the best performing instrument (spoilers: this study was funded by Abbott), though each had their strengths—read the paper here. The multiplex method, although similar in principle, is unique to the BioPlex. And one of the tests I find interesting is their syphilis assay. On the BioPlex, its tested as a total immunoassay with RPR that improves accuracy and precision in real-time. It’s a dual treponemal/non-treponemal test. Those bead packs contain two types of beads that qualitatively detect IgG and IgM antibodies to Treponema pallidum while also qualitatively detecting RPR antibodies in serum or plasma. It comes with its own internal control and verification beads for internal QC. Fast, simple, easy, unique and just as accurate as most syphilis testing—that’s what I’m talking about.

(Immuno) Fixing Everyone’s Problems
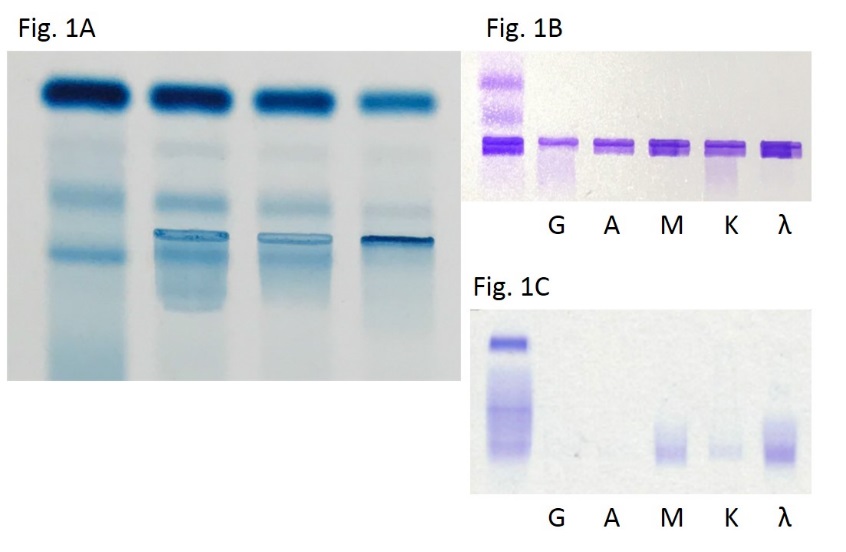
I remember learning a lot about gel electrophoresis, serum/urine protein electrophoresis, and immunofixation in MLS school but never really spent too much time with gels. I didn’t get to do much work with electrophoresis because I spent my time in labs either on a microscope doing diffs, a blood bank shaking tubes, or monitoring chemistry outputs and validating reports. Despite spending years with an ID badge that often said “chemistry” on it, gels were a less common test and not just for me. The primary care clinician usually only orders SPEP and subsequent gels in the investigation of multiple myeloma or other paraproteinemia from a vast array of disease process. As such, the results are often more challenging to interpret and require reporting and education from the pathology department. There are, however, a myriad of interpretable patterns and information within the gels of an SPEP. The principle is standard—proteins separate in media based on charge and mass—and particular patterns tell us more information than one might realize. For example, chronic inflammatory processes might increase production of acute phase reactants like alpha-1-antitrypsin or haptoglobin. Where do the peptide building blocks for those new proteins come from? Wherever the most protein is: Albumin, obviously for those playing at home. So your friendly normal neighborhood SPEP in a chronic inflammatory process might morph into something with a fainter albumin band and some extra attenuation in the alphas 1 and 2. If you see that, you might correlate with an ESR or CRP (or hs-CRP, you fancy laboratorian) …


NKX3.1, the Lexus of Prostate IHCs
Now for something different. Let’s talk about prostates. Fun! More specifically, let’s talk about prostate-related immunohistochemical stains. The first one you’ll think off right away is probably …drumroll… PSA, and you’d be absolutely correct. That’s a standard IHC for detecting prostatic adenocarcinoma and is especially useful in finding metastases from a prostate primary. Though not brand new, there is another stain for prostatic IHC detection that, in some recent studies, has been shown to be more sensitive for prostate malignancy than PSA by about 5%. It’s called NKX3.1 and it has PSA’s sensitivity of 94.2% beat at around 98.6%–that could translate to plenty of earlier diagnoses and better outcomes for patients.

What’s that got to do with my current pathology rotation? Well, I’ve gotten a lot of anatomical pathology exposure in my time here and I even helped correlate IHC stain quality across two instruments. With that done, I’m currently collecting specimens of saved tissue blocks that were both positive and negative for the lab’s current prostate IHC, PSA, and retesting them all with NKX3.1 in order to switch protocols to the new, more sensitive test. At the very least, the addition of a secondary validated prostatic stain would be useful. What’s important in gathering specimens for this kind of correlation is understanding when and where this new stain would be positive and negative and making sure it behaves in your patient population the way you would expect. NKX3.1 is supposed to be positive in nearly 99% of prostatic adenocarcinomas whether they’re primary or metastatic. It is a Chromosome 8 protein which is expressed in the prostate and testis and can even be found in the salivary glands, bronchial submucosal glands, and regions of the ureters. It can be positive in 27% of invasive lobular carcinoma, 25% of metastatic lobular carcinoma, 2-9% of invasive ductal carcinoma, and 5% of metastatic ductal carcinoma. (Source: Pathology Outlines) While I’m looking over specimens with historical orders for PSA IHCs, not all are positive, and not all are prostatic tissue. Conducting validation studies like these in pathology really require a good understanding of how to clinically correlate data with useful decision-making and tailor it to your patient population.
I wrote about a lot of topics this month, I know, but I think there’s a common theme. As a medical laboratory scientist, like many of you, I’ve worked out countless QC problems and instrument validations per protocol. Now that I’m making the transition to medicine in pathology, there’s a lot of forethought and planning that goes into validating or calibrating any test. In chemistry you need to get your limits of detection just right and match your throughput with the test volume your population needs. In hematology, you better know exactly how cells get detected by your analyzers and have a solid algorithm for working up and understanding aberrant flags. When it comes to anatomic pathology, speaking a common language of morphology and pattern-recognition is vital to reporting reliable and critically important data. Laboratory medicine always exists at the forefront of medical testing and methodology, and what that translates to on a day-to-day basis is being able to know how to find, make, or confirm a good, reliable test. As for me, medical school is full of unique experiences and rewarding opportunities to learn. This month, I couldn’t be happier to use my skills in the lab to connect my time at the bench to my work learning, calibrating, and validating for the next step.
And, after all, aren’t we all looking for a little validation now and then?
Thanks to Danbury Hospital’s Department of Pathology and Laboratory Medicine for having me this month and thank you all for reading.
See you next time!

–Constantine E. Kanakis MSc, MLS (ASCP)CM graduated from Loyola University Chicago with a BS in Molecular Biology and Bioethics and then Rush University with an MS in Medical Laboratory Science. He is currently a medical student actively involved in public health and laboratory medicine, conducting clinicals at Bronx-Care Hospital Center in New York City.