Case history
A 43 year old woman with no significant medical history arrived at the emergency room complaining of several days of right-sided headaches, and worsening neck stiffness. Two days prior to coming into the emergency room, she had noticed some “bumps” on her posterior scalp, which her hairdresser looked at for her and confirmed the presence of a rash. Physical exam revealed a low-grade temperature of 100.6F. A small rash on the right side of the head was seen, consisting of a few erythematous patches and vesicles. A lumbar puncture was performed revealing clear and colorless cerebrospinal fluid, and the patient was given doses of ceftriaxone, vancomycin, and acyclovir.
Laboratory diagnosis
Analysis of the CSF was as follows:
- Glucose: 45
- Total Protein: 148 (H)
- RBC Count: 15
- Nucleated cell Count: 314
- Neutrophils: 5%
- Lymphocytes: 72%
- Monocytes: 20%
- Eosinophils: 3%
PCR of the CSF was positive for varicella-zoster virus (Figure 1).
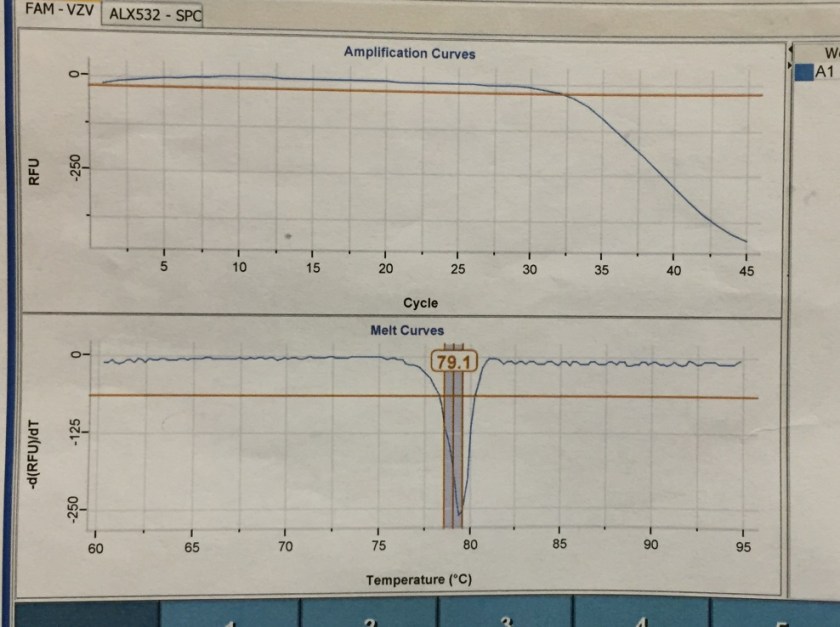
Figure 1. Real-time PCR amplification curves and melting curves for Varicella Zoster Virus in patient’s CSF sample. The amplification curve demonstrates decrease of Relative Fluorescence Units (RFUs) corresponding with detection of viral DNA. The melting point is determined by the composition of nucleic acids, and is unique to VZV, confirming the identity of the virus detected.
Discussion
Primary infection with VZV causes the classic chickenpox illness characterized by a widespread vesicular rash, with lesions of varying ages. Herpes zoster (shingles) occurs when latent varicella-zoster virus (VZV) in a sensory ganglion becomes reactivated, resulting in a painful vesicular rash typically in a dermatomal distribution. By 7-10 days after eruption, the vesicles of the rash will crust over and are not considered infectious. However, in immunocompromised patients, new vesicles may continue to erupt. The predominant complaint is pain along the site of the rash, in 75% of patients.
Even in immunocompetent patients, there is a risk of aseptic meningitis and even encephalitis with VZV reactivation. This can occur from the virus spreading centrally, to the CNS, rather than distally down a spinal nerve. Some studies have even indicated that subclinical meningitis (defined as CSF pleocytosis) occurs in up to 50% of individuals with herpes zoster. In one study, 0.5% of patients with herpes zoster developed meningitis. The typical findings of zoster meningitis on lumbar puncture include elevated protein as well as lymphocytosis.
Antiviral therapy (either with valacyclovir, famciclovir, or acyclovir) is often advised for the treatment of shingles if patients present within 3 days of symptom onset; it has the benefits of shortening the duration of skin lesions and acute neuritis, though its effects on post-herpetic neuralgia are less clear. After three days, the clinical benefit of antiviral treatment is debatable; however, it is recommended for patients with neurologic complications or with compromised immune systems.
The patient had chickenpox when in college. Although she had her zoster outbreak for 4 days by the time of presentation, because of the meningitis a course of oral Valtrex was prescribed. She was discharged home as she was clinically stable.
-Alison Krywanczyk, MD is a 3rd year anatomic and clinical pathology resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Assistant Professor at the University of Vermont.