Case history
A 60-year old woman residing in Vermont presented to the dermatology clinic for a routine annual skin exam. She had no complaints. On physical exam, a pink papule was seen on the patient’s back, with a centrally embedded tick (Figure 1). The tick was removed and sent for identification, with the plan to give a single prophylactic dose of doxycycline if identified as Ixodes scapularis.
Laboratory Identification
The tick was examined and noted to be less than 1 mm in size, with six legs (Figure 2). This was identified as the larval stage of Ixodes scapularis.
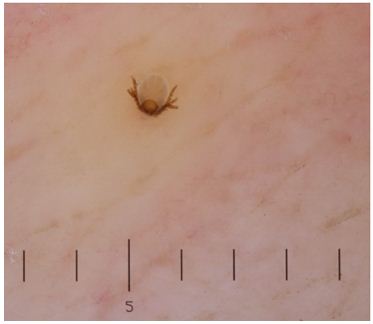
Figure 1. Photograph of Ixodes scapularis larva embedded in the patient’s back. Six legs are visible.

Figure 2. Photograph of the tick larva received in the laboratory, demonstrating a light tan-brown color.
Discussion
I scapularis, also known as the blacklegged tick, deer tick, or bear tick, is most clinically significant for its ability to transmit the pathogens Borrelia burgdorferi, Babesia spp., and Anaplasma phagocytophilum. It has four separate life stages (egg, larva, nymph, and then adult), spanning approximately 2 years. Each of these stages feeds on different preferred host animals.
Eggs are deposited on the ground by blood-engorged females in the late spring, where they subsequently hatch into 6-legged larvae. Because they have not yet fed, larva forms generally do not carry or transmit B. burgdorferi or other tick-borne pathogens. Trans-ovarial transmission of Borrelia, Anaplasma, or Babesia from adult I. scapularis females to eggs of is not a significant mode of pathogen transmission; however, in a similar tick species, I. ricinus (prominent in Europe), trans-ovarial transmission of Babesia divergens does occur, and so infection may be transmitted by larvae. The I. scapularis larvae will take their first blood meal from small mammals and birds, and then when engorged fall to the ground and molt into nymphs.
The nymph forms, which have already taken a blood meal, can carry pathogens and in fact are more likely to transmit pathogens to humans than the adult form of the tick. This is because the nymph form is much smaller (<2mm in size) than the adult form, and therefore is likely to go undetected when it attaches to a host. The nymphs are dormant over the winter, and re-activate the following spring to take their second meal. By fall, nymph forms have molted into adult ticks, which prefer to feed on white-tailed deer. However, while these deer support the tick population, they are not a large reservoir for Lyme disease. Rather, it is the white-footed mice preferentially fed upon by larvae and nymph forms that act as the main reservoir for B. burgdorferi, B. microti, and A. phagocytophilum. The female adults of I. scapularis are red to orange and larger than males, around 1/8 of an inch long, with a dark brown to black dorsal shield. If females do not feed in the fall, they can remain dormant over the winter and may emerge if the weather gets temporarily warmer (so the onset of cold weather does not necessarily mean the risk of tick exposure is over). Male adults do not take blood meals, and so do not transmit blood-borne pathogens.
To be considered in the differential diagnosis is Dermacentor variabilis, or the American dog tick. This tick species is larger than Ixodes spp., and adult forms have a white-to-gray collar on their backs. D. variabilis have more rectangular-shaped head and mouth parts than the deer tick. Both nymph and larvae forms are yellow-brown in color before feeding, and then turn gray once engorged. It is extremely uncommon for nymph and larval forms of D. variabilis to feed on humans, in contrast to I. scapularis. D. variabilis does not transmit Lyme disease, though in endemic areas it may transmit Rickettsia rickettsii or Francisella tularensis.
Because the tick in the presented case was identified as an I. scapularis larva, the patient was not treated with antibiotics as there was an exceedingly low risk of pathogen transmission.
-Alison Krywanczyk, MD is a 3rd year anatomic and clinical pathology resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Assistant Professor at the University of Vermont.
