Hello again everyone! And special thanks to the readers who read, commented, shared, and reached out to me from my last post “A Serious Aside,” talking about physician burnout and health worker suicide. Numerous people had so much to say in support of this topic—and it’s well deserved—sharing their personal stories and relating their own connects, so I truly appreciate it.
This time, how about something different? In the past few months, I’ve been working through my clinical rotations at a major community hospital in New York City, in the Bronx. A CDC-sponsored screensaver image keeps appearing at terminals throughout floors, services, and clinics; and it directly addresses healthcare professionals to monitor hygiene practices to eliminate Candida infections. I’ll have to admit—innocuous stuff—I’ve been seeing health-message PSA-like screensavers at work for years, about a myriad of topics. Who hasn’t seen those? “Keep beds out of the hallways,” “Protect you and your patients from MRSA,” “Make sure lab requisitions are filled out properly…” the list is endless. But having seen my aforementioned screensaver about Candida one too many times, I had to find out what this was about. You might have thought that, since I spent time working in an HIV clinic, this was a simple PSA for those patients otherwise immunocompromised. Right? Nope.
This particular PSA from the CDC warns about Candida auris, a true blue (or pinkish gold, rather) member of everyone’s favorite budding, germ-tube positive, yeast family. C. auris has been in literature for roughly the past decade. Having etiologic origins in southeast Asia and spreading west through the Middle-East, all throughout Africa, and even the UK, this bug has caught the eyes of epidemiologists around the world. Two years ago, the CDC1 and Public health England2 issued warnings about this pathogen, its multi-drug resistance, and its virulence in healthcare-associated infections. Last fall, the NY State Department of Health published their official update for guiding clinicians and laboratory staff.3 In this report, they discussed infection control, prevention, and detection limitations.


So what’s so scary about C. auris? The two most challenging features of this emerging pathogen are its multi-drug resistance and its relatively difficult identification.
This yeast has been shown to show resistance to many antifungal/antimicrobial agents including fluconazole, voriconazole, amphotericin-B, echinocandins, and even flucytosine. Even more concerning is that nearly half of the C. auris strains collected in research done in Asia, Africa, and South America demonstrated multi-drug resistance patterns to two or more combination therapies. These are most of our first-line standard of care therapies for invasive candidiasis in patients!
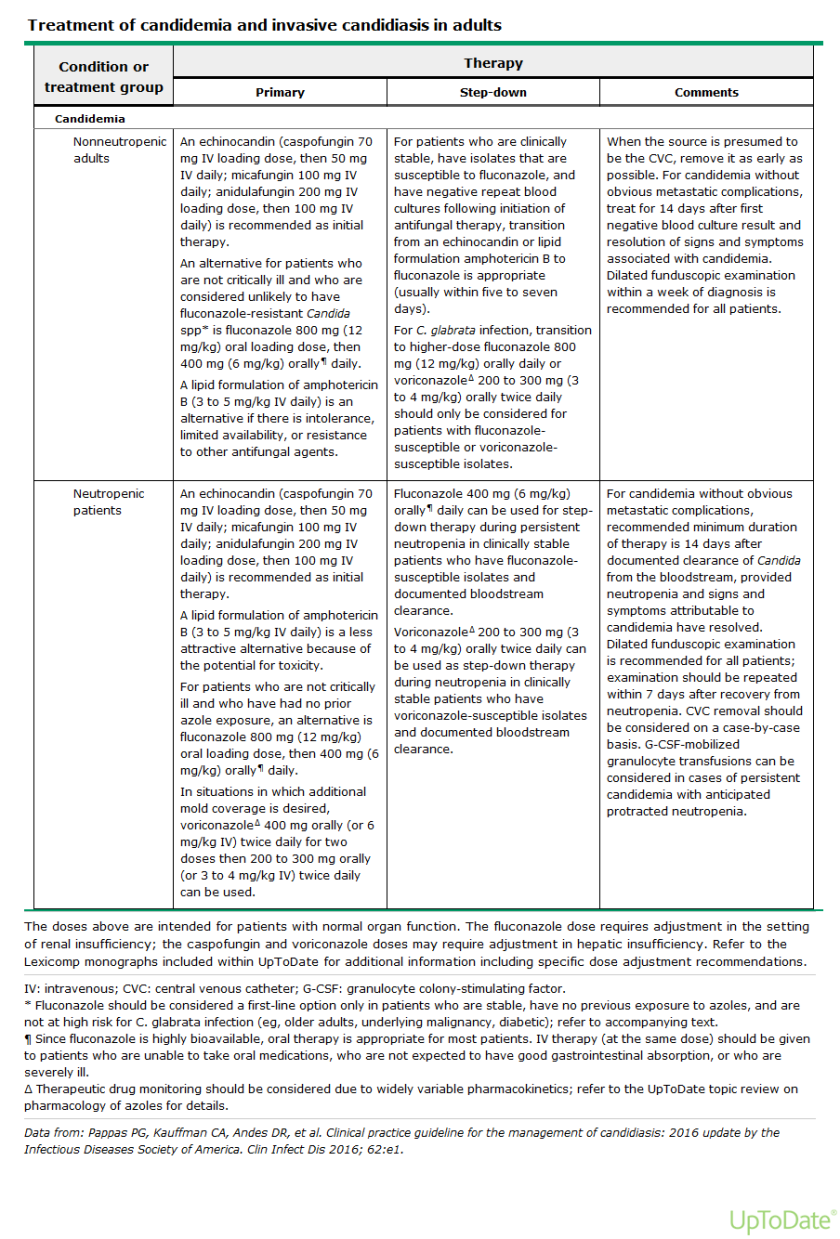
There are various other recommendations regarding therapies to C. auris specifically, as its potential for resistance are known, but infection control along with empiric therapy seem to be the current standard.
The major risk factors for C. auris infections include the relative status of individual patients: intensive care, acute renal failure, immunocompromised status, localized or systemic infections, and colonization. Simply being hospitalized is an associated risk. On my current service of patients I’m part of a nephrology/medicine team. There are several chronic infection, ESRD, immunocompromised, or otherwise applicable patients to these risk stratifications. No wonder we’ve got those screensavers!
Concerns for identifying C. auris take us back to the lab. Detecting this bug is not as simple as a couple microscopic morphologies and a yeast API strip—sorry to my old mycology professors. C. auris based on chemical tests like these can produce confounding results. Even VITEK identification (unless you’re running Vitek 2 with Biomerieux software) or culture growth can yield non-specifics like C. haemulonii or Saccharomyces cerevisiae. C. auris has a very high salt and temperature tolerance, and with no particular morphologic identifiable features, it remains a challenging identification. It can be grown on dulcitol agar or CHROMagar, but you do not get clear results. What’s the way to get the ID then? Ultimately MALDI-TOF, PCR, and molecular testing is the answer. There are already available C. auris sequences you can obtain for in-house validation if you’re using MALDI already. And when it comes to susceptibility, fear not: as far as I’ve been able to read E-Tests still work.
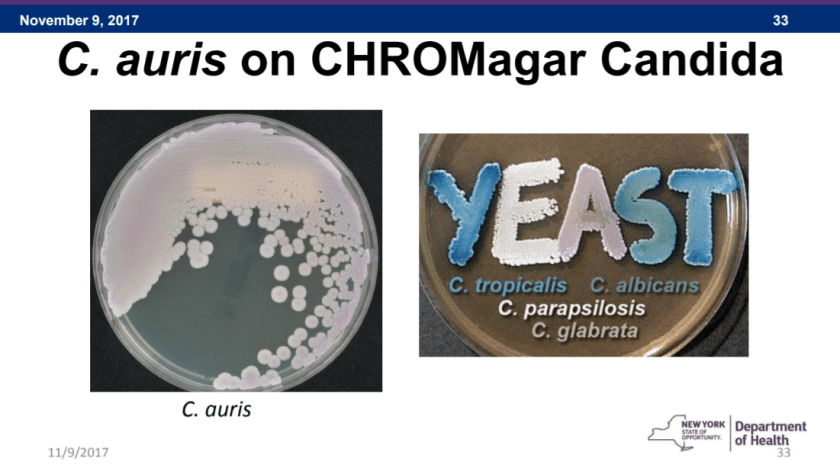
I was very impressed with MALDI when I was working in Chicago, and a community hospital I was with just finished validating when I left for medical school. I am glad to see it again with this emergent pathogen, and it definitely demonstrates the next wave of instrumentation. Extremely rapid and very accurate.
The variable drug susceptibility, virulence, and ability to thrive in the environment actively threaten those with long inpatient stays. This microorganism is treated with standard precautions and infection control measures. Currently NY leads the nation by far in purported cases of C. auris. So … please wash your hands. A lot. I know I am.
Thanks! See you next time!
References
- Centers for Disease Control and Prevention. Clinical Alert to U.S. Healthcare Facilities – Global Emergence of Invasive Infections Caused by the Multidrug-Resistant Yeast Candida auris. https://www.cdc.gov/fungal/diseases/candidiasis/candida-auris-alert.html
- Public Health England. Candida auris identified in England. https://www.gov.uk/government/publications/candida-auris-emergence-in-england/candida-auris-identified-in-england
- NY State Department of Health https://www.health.ny.gov/diseases/communicable/c_auris/docs/c_auris_update_for_lab_staff.pdf

–Constantine E. Kanakis MSc, MLS (ASCP)CM graduated from Loyola University Chicago with a BS in Molecular Biology and Bioethics and then Rush University with an MS in Medical Laboratory Science. He is currently a medical student at the American University of the Caribbean and actively involved with local public health.
