Case
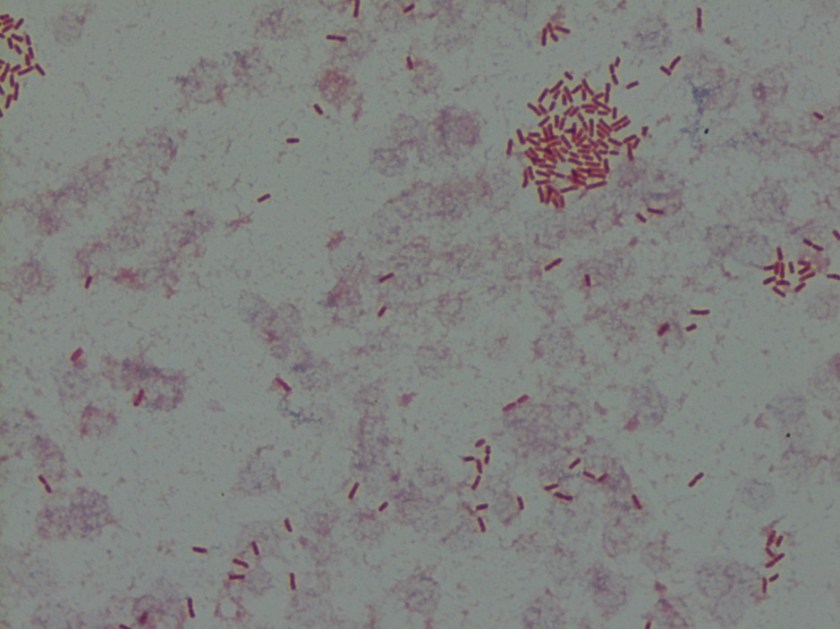
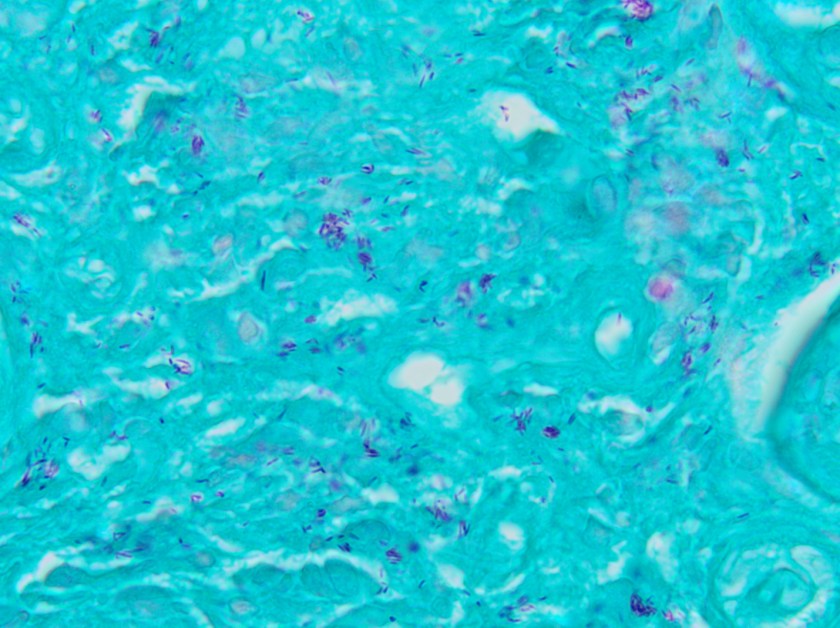
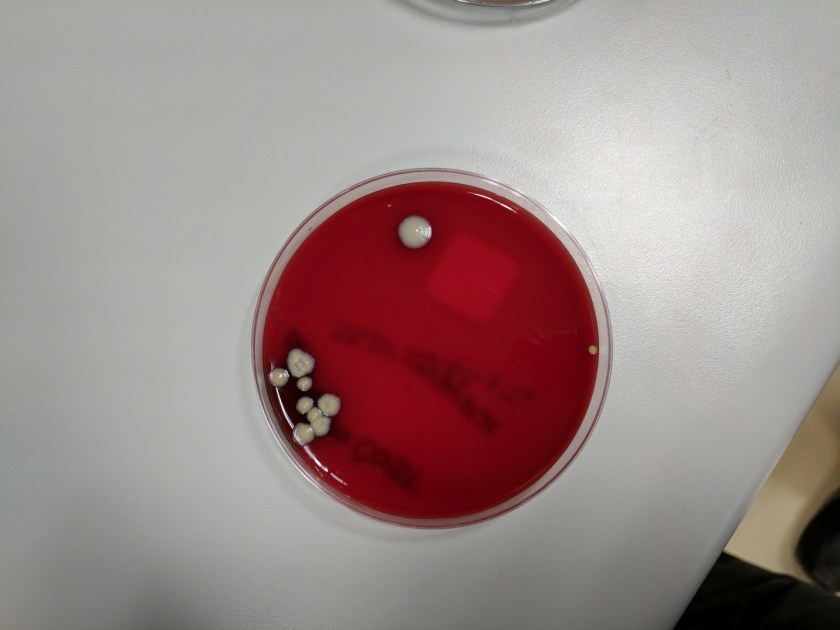
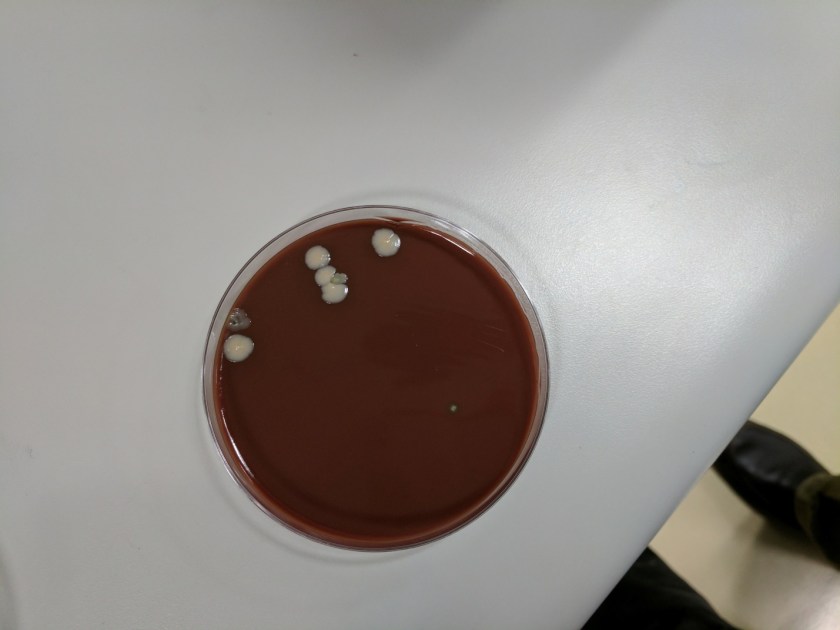
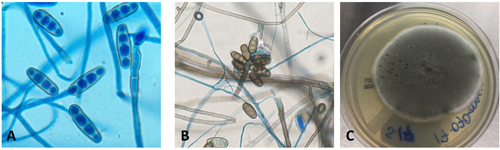
A 16-year-old male presented with recurrent sinusitis and rhinitis. He had a history of left sinus surgery two years ago, and at that time pathologic examination of the tissue demonstrated eosinophilia and fungal culture grew Curvularia, consistent allergic fungal sinusitis. The patient was doing well without allergy or immunotherapy management until three months ago when he could not breathe out of the right nostril and began snoring loudly. He underwent bilateral endoscopic frontal sinusotomy with tissue removal of the ethmoid and sphenoid sinuses. Tissue was sent to the laboratory for fungal culture. After five days, fungal cultures grew mold on inhibitory mold agar with gentamicin. The surface was a gray speckled color (Image 1C). The reverse color of the mold colony was dark brown to black. The microscopic appearance can be seen in Image 1A-B.
Discussion
These features are consistent with the identification of Bipolaris. Microscopic examination using lactophenol cotton blue tape prep demonstrated oblong conidia characteristic of Bipolaris (Image 1 A-B). The conidia are ellipsoidal with pale brown pseudoseptations that contain three to five septa. Four septa are the most common. Bipolaris is a dematiaceous fungus, meaning the cell walls contain dark brown melanin pigment. This can be seen by microscopic observation of the fungal cell wall which contains pigment (Image 1B) and is also demonstrated by the dark reverse color of the fungal colony.
To distinguish Bipolaris from Drechslera and Exserohilum, the Germ tube test can be utilized. When conidia are incubated with a drop of water on a glass slide for 8-24 hours, they will begin to form Germ tubes. Bipolaris species germinate from both poles of the oblong conidium at a 180 degree angles (hence the name “Bipolaris”), whereas Exserohilum germinate from just one pole at a 180 degree angle and Dreschslera species germinate at a 90 degree angle from the central cells of the conidium. Dreschslera can be confused for Bipolaris based on colony appearance and microscopic appearance, but unlike Bipolaris, Dreschslera is not associated with human disease.1
Pathogenic strains of Bipolaris include Bipolaris australiensis, Bipolaris hawaiiensis, Bipolaris maydis, Bipolaris melanidis, Bipolaris speicifera, and Bipolaris sorokiniana.2 The most common cause of infection is Bipolaris spifcifera. Bipolaris species are the most common cause of fungal sinusitis in immunocompetent individuals which often presents as allergic rhinitis. Allergic rhinitis could also be a risk factor for acquiring Bipolaris. Treatment often consists of prompt surgical excision to prevent expansion, superficial deformity and dissemination. If fungal chemotherapy is pursued, itraconazole and amphoterin B have been reported as effective agents.3
Bipolaris is one of the most common causes of allergic fungal sinusitis, typified by nasal polyps and mucus plugs consisting of eosinophils, fungal hyphae and Charcot-Leyden crystals. It is a type 1 and 3 hypersensitivity reaction mediated process due to high levels of mold-specific IgE.4 Skin prick testing is also positive in patients with allergic fungal rhinosinusitis (AFRS) which further indicates that the pathophysiology is an immunologic versus infectious process.4 While the exact process of fungal allergic sensitization has not been codified, chitin, a structural fungal protein has been shown to elicit a Th2 immune response.5 It will be interesting to see how this research evolves so that we might one day see why fungi can cause both erosive infections and allergies within human patients.
References
- Fothergill AW. Identification of Dematiaceous Fungi and Their Role in Human Disease. Clin Infect Dis. 1996; 22 (S2): S179-84.
- Shafili SM, Donate G, Mannari RJ, Payne WG, Robson MC. Diagnostic Dilemmas: Cutaneous Fungal Bipolaris Infection. Wounds. 2006; 18(1):19-24.
- Saenz RE, Brown WD, Sanders CV. Allergic Bronchopulmonary Disease Caused by Bipolaris hawaiiensisPresenting as a Necrotizing Pneumonia: Case Report and Review of Literature. The American Journal of Medical Sciences. 2001; 321(3):209-12.
- Manning SC, Holman M. Further evidence for allergic pathophysiology in allergic fungal sinusitis. Laryngoscope. 1998;108(10):1485–1496.
- Reese TA, Liang HE, Tager AM, Luster AD, Van Rooijen N, Voehringer D, Locksley RM. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature 2007; 3;447(7140):92-6.

-Jeffrey SoRelle, MD, is a 1st year Clinical Pathology Resident at UT Southwestern Medical Center.

–Erin McElvania TeKippe, PhD, D(ABMM), is the Director of Clinical Microbiology at Children’s Medical Center in Dallas Texas and an Assistant Professor of Pathology and Pediatrics at University of Texas Southwestern Medical Center.