Case History
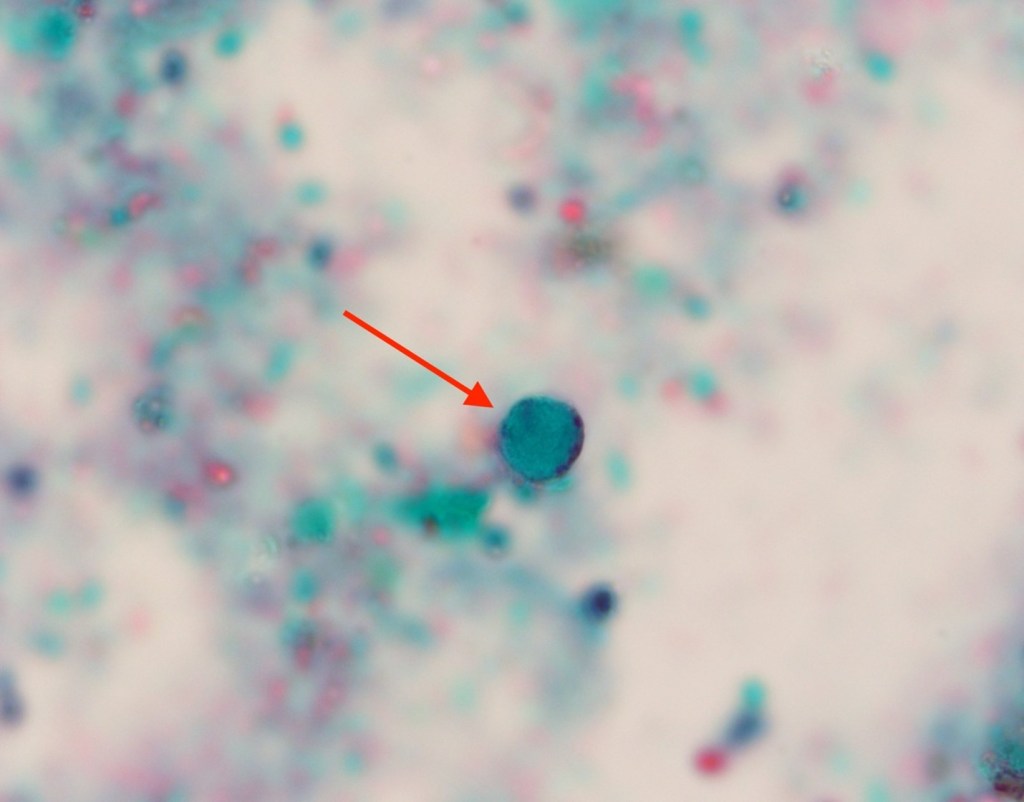
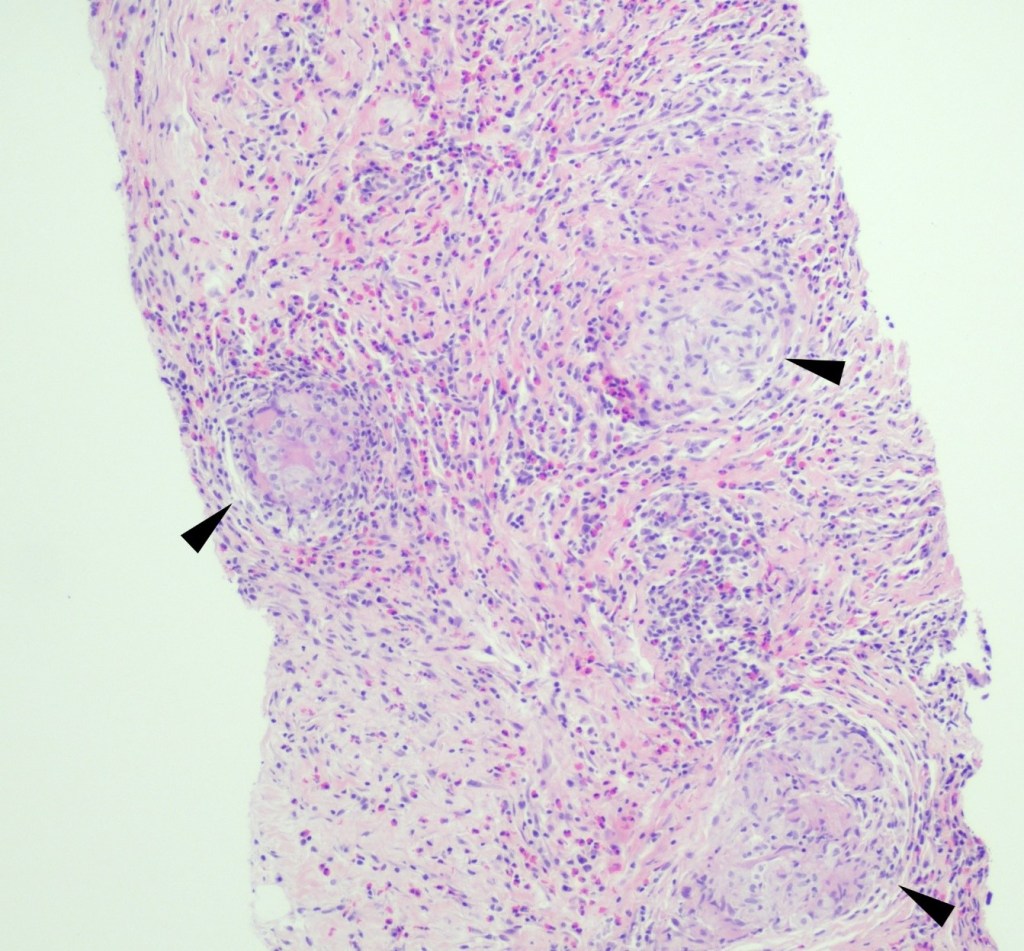
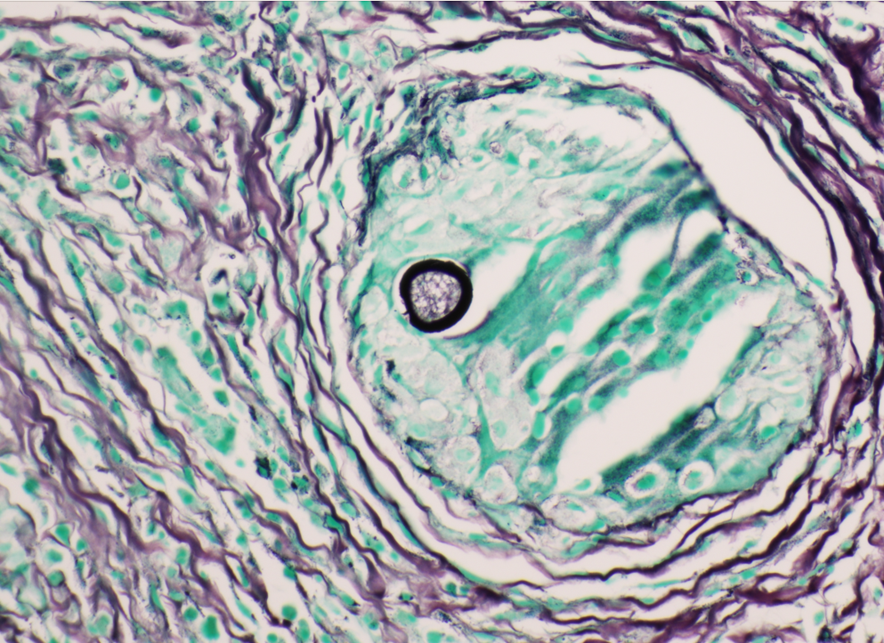
A 75 year old man came to the Emergency Room because of intractable hiccups. He had a medical history of esophagitis, gastroesophageal reflux disease, gastric metaplasia diagnosed during a previous esophago-gastroduodenoscopy (EGD), and a significant episode of hiccups for several years. His esophagogastroduodenoscopy revealed diffuse edema and erythema on the duodenal mucosa. Histopathological examination of the duodenal biopsies (Figures 1-3) showed the presence of Strongyloides stercoralis within a few crypts of the duodenum, and adjacent eosinophil-rich inflammatory infiltrate within the mucosa. These findings provided an incidental finding of the parasite’s presence in the duodenal mucosa.



Discussion
Strongyloidiasis is a parasitic infection caused by the nematode Strongyloides – most commonly S. stercoralis. While it is commonly seen in tropical and subtropical regions, cases can also occur in temperate climates. Notably, our patient had a recent travel history to Jamaica, a known endemic region for Strongyloides infection.
The life cycle of Strongyloides stercoralis involves both free-living and parasitic stages. The infectious filariform larvae penetrate the human skin typically after contact with contaminated soil or exposure to infected fecal matter. Subsequently, they migrate to the lungs through the bloodstream, and eventually reach the small intestine, where they mature into adult worms. The adult worms reside in the duodenal and proximal jejunal mucosa, reproducing asexually by parthenogenesis. Some of the eggs hatch within the intestine, releasing rhabditiform larvae into the feces. It causes autoinfection by penetrating the intestinal wall or the perianal skin area.
The diagnosis of Strongyloides is typically accomplished by morphologic identification of larvae in the stool, duodenal aspirate, or sputum in disseminated cases. Strongyloides serologic testing is often performed in transplant patients who have a pertinent demographic and clinical history of potential exposure. The presence of eggs is rarely observed in the stool; therefore, microscopic examination of stool samples may have a lower sensitivity in uncomplicated infection with a low organism burden. In our case, stool samples were not collected for evaluation. Hyper-infection syndromes associated with disseminated Strongyloides could present as subclinical infection in patients under immunosuppression. As the larvae invade other organs, such as CNS, lungs, and blood stream, intestinal flora from the GI tract is carried along with the larvae, which causes super-infections, such as bacteremia and meningitis.
No FDA-cleared molecular testing is available for Strongyloides while some reference laboratories may offer laboratory-developed-tests. Therefore, the laboratory diagnosis frequently relies on the morphologic identification of the filariform larvae or eggs from clinical samples. In our case, the histopathological examination of the duodenal biopsies that were obtained to evaluate persistent hiccups revealed a significant eosinophil-rich inflammatory infiltrate within the mucosa, along with the presence of the larvae within the crypts. While hiccups can be due to various etiologies, including gastrointestinal disturbances and certain medications, and may not be directly related to parasitic infections, the diagnosis of Strongyloides in this case was purely incidental.
References
- Gulwani, Hanni. “Strongyloides Stercoralis.” Pathology Outlines – Strongyloides Stercoralis, Aug. 2012, http://www.pathologyoutlines.com/topic/smallbowelstrongyloides.html.
- Carrada-Bravo, Teodoro. “Strongyloides Stercoralis: Vital Cycle, Clinical Manifestations, Epidemiology, Pathology and Treatment.” Revista Mexicana de Patolog, 1 Jan. 1970, http://www.medigraphic.com/cgi-bin/new/resumenI.cgi?IDARTICULO=16127.
- “Strongyloides Stercoralis.” RCPA, 2023, http://www.rcpa.edu.au/Manuals/RCPA-Manual/Clinical-Problems/S/Strongyloides-stercoralis.
- De la Cruz Mayhua, Juan Carlos, and Bisharah Rizvi. “Strongyloides Hyperinfection Causing Gastrointestinal Bleeding and Bacteremia in an Immunocompromised Patient.” Cureus, 24 June 2021, www.ncbi.nlm.nih.gov/pmc/articles/PMC8310433/.

-Inas Mukhtar, MD, is from Sudan and graduated medical school from University of Khartoum and started a pathology residency in Sudan before applying here to the US. She is currently PGY-2 at Montefiore Medical Center. Her hobbies include watching documentaries and spending time with friends and family.

-Phyu Thwe, Ph.D, D(ABMM), MLS(ASCP)CM is Associate Director of Infectious Disease Testing Laboratory at Montefiore Medical Center, Bronx, NY. She completed her medical and public health microbiology fellowship in University of Texas Medical Branch (UTMB), Galveston, TX. Her interests includes appropriate test utilization, diagnostic stewardship, development of molecular infectious disease testing, and extrapulmonary tuberculosis.