Patient History: Case 1
A 55 year old Asian woman presented to an emergency department in southern New England in September with complaints of a high fever with chills for the past 5 days. She noted feelings of excessive tiredness, muscle aches, and headache. She also described a decrease in appetite and nausea with vomiting and diarrhea. On physical exam, she was febrile (103.8°F) and scleral icterus was identified. Laboratory workup revealed findings suggestive of hemolysis including increased LDH (401 U/L) and increased unconjugated bilirubin (1.7 mg/dL), despite hemoglobin & hematocrit values in the normal range (13.7 g/dL & 39.3%, respectively). Elevated liver enzymes were also noted; AST 81 U/L and ALT 72 U/L. When questioned regarding traveling history, she reported a trip to Spain and Portugal 5 months earlier. Though she acknowledged living in a rural area of the Northeastern U.S. and indicated that her husband was diagnosed with Lyme disease one year earlier, she denied both recent time outdoors and arthropod or mosquito bites.
Patient History: Case 2
A 31 year old African American woman with a history of sickle cell trait presented to an emergency department in southern New England in September complaining of fevers of 5 days duration. She described being asymptomatic in the mornings followed by high spiking fevers with muscle aches and dull frontal headaches in the evenings. A physical exam revealed a fever (103°F), but no evidence of meningismus. Laboratory workup revealed a mild, microcytic anemia (hemoglobin & hematocrit: 10.7 g/dL & 32.5%, MCV: 76.3 fL), a decreased absolute lymphocyte count and increased band neutrophils. When questioned regarding recent travel, she reported having returned from Africa 10 days earlier. While abroad, she had primarily been in Nigeria’s capital, but she had also visited rural areas. She did not recall having been bitten by mosquitos, but she did not take any anti-malarial prophylaxis. Further, she denied both recent travel to the woods in the Northeastern U.S. and recent arthropod bites.

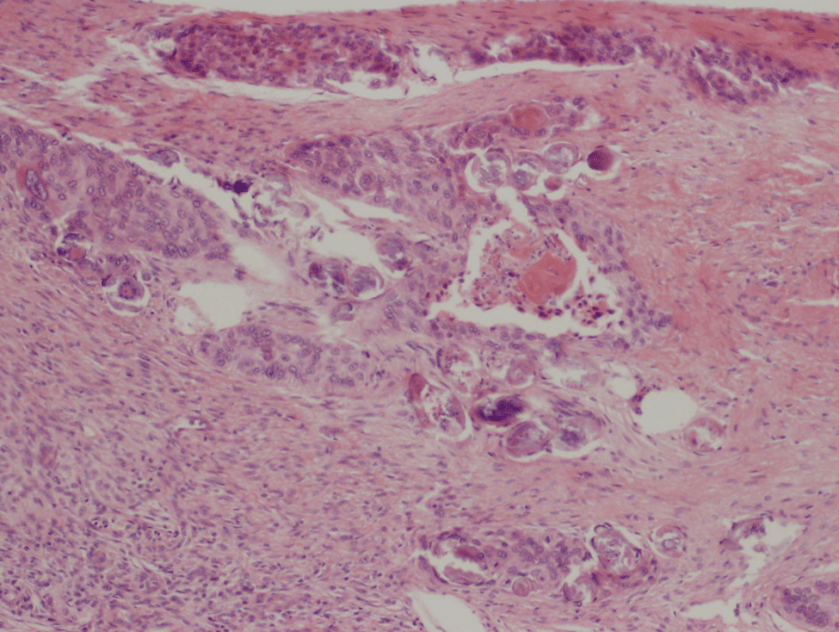
Figure 1. Peripheral blood smear from patient 1 showing ring-like forms which contain a small amount of cytoplasm and a chromatin dot as illustrated by the arrows. Both intra-erythrocytic and extra-cellular forms are present. Platelets are denoted by arrowheads.
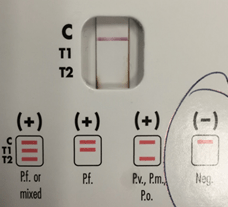
Figure 2. BinaxNOW lateral flow assay from patient 1 is negative for the various Plasmodium spp.

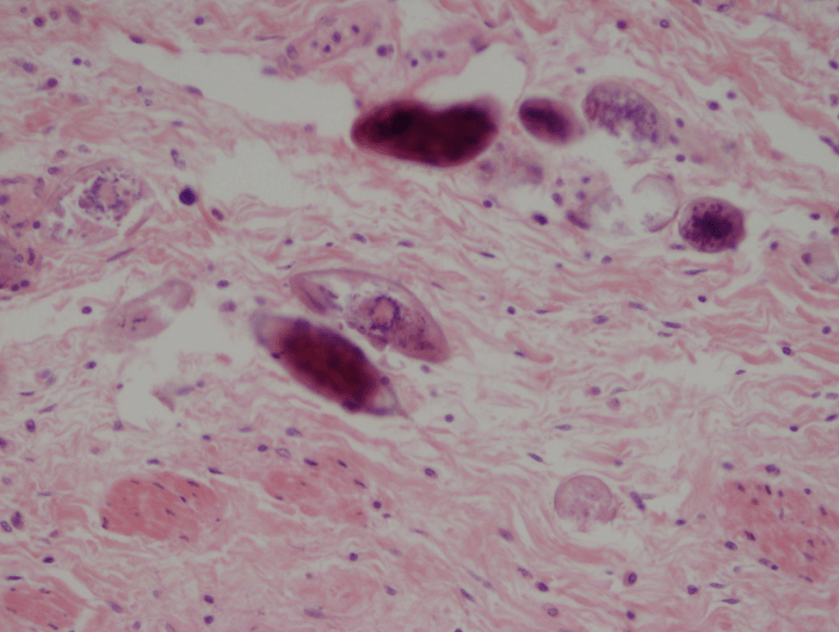
Figure 3. Peripheral blood smear from patient 2 showing ring forms and trophozoites within red blood cells as denoted by arrows. Inset illustrates a scattered gametocyte. Platelets are denoted by arrowheads.

Figure 4. BinaxNOW lateral flow assay from patient 2 is positive for non-falciparum malaria species as indicated by the faint positive reaction denoted by the red arrow in the T2 region.
For patient 1, given the results from the peripheral blood (Figure 1), the negative BinaxNow results (Figure 2) and her lack of recent travel to malaria endemic regions, her illness was attributed to infection by Babesia spp. Further serologic testing was positive for Babesia microti. She was seronegative for Anaplasma phagocytophilum, Borrelia burgdorferi, Ehrlichia chaffeenesis. This finding was confirmed by PCR of her blood, which detected B. microti, but failed to detect B. duncani or B. divergens/MO-1. Approximately 3% of her red blood cells contained intracellular parasites.
For patient 2, her disease was most consistent with an infection by a non-falciparum species of malaria, including P. ovale, P. vivax or P. malariae, given her recent travel to Nigeria and advanced forms seen in the peripheral blood (Figure 3). Further speciation was uncertain due to low parasitemia levels (<1%) and the findings were unable to exclude a mixed infection with a low P. falciparum burden.
Discussion
The clinical and laboratory presentations of babesiosis and malaria are quite similar despite the fact that each infection is caused by a distinct and highly unique microorganism. As seen in the two cases above, both illnesses often begin insidiously with fevers, headache and muscle & joint aches. The non-specific nature of the patient’s symptoms results in an unclear etiology unless key elements of the patient’s history, including exposure to insect and arthropod vectors and travel or habitation in endemic areas, are provided.
Examination of thick and thin blood smears is useful in the diagnosis of these two diseases. While both organisms have a very similar sized lifecycle forms which selectively infect red blood cells and prompt hemolysis, there are a few useful distinguishing characteristics. In the case of babesiosis, which is transmitted by the Ixodes scapularis tick in the United States, there are small ring like structures, both within red blood cells and extra-cellularly. The diagnostic tetrad form, known as a Maltese Cross, is helpful if identified but is not frequently observed in human infections. No advanced forms or pigment is present. In the case of malaria, which is transmitted via the female anopheline mosquito, protozoa are only found within red blood cells and advanced forms, including schizonts or gametocytes, are helpful in further speciation, if present. Other features, such as size of the infected red cell, number of merozoites, level of parasitemia and gametocyte shape, are helpful in the morphologic assessment of the Plasmodium spp.
Due to the pathogenic severity of P. falciparum, it is important that the microbiology laboratory has the ability to make the diagnosis in real time across all shifts. The BinaxNOW is an FDA approved lateral flow assay that is simple to perform and provides rapid diagnostics, though it isn’t as sensitive as microscopy. The test is comprised of two antigens: one specific to P. falciparum (T1) and one antigen common to all Plasmodium spp. (T2). The test will be positive for levels of parasitemia greater than 5,000 parasites per microliter.
As utilized in the above cases, other various laboratory modalities can aid in the diagnosis of babesiosis and malaria, including serologic tests and PCR, however, these tests may not be available in STAT situations. Using a variety of tests and obtaining a thorough travel history, will help the provider arrive at the correct diagnosis of blood protozoa.

-JP Lavik, MD, PhD, is a 3rd year Anatomic and Clinical Pathology Resident at Yale New Haven Hospital.

-Lisa Stempak, MD, is an Assistant Professor of Pathology at the University of Mississippi Medical Center in Jackson, MS. She is certified by the American Board of Pathology in Anatomic and Clinical Pathology as well as Medical Microbiology. Currently, she oversees testing performed in both the Chemistry and Microbiology Laboratories. Her interests include infectious disease histology, process and quality improvement and resident education.