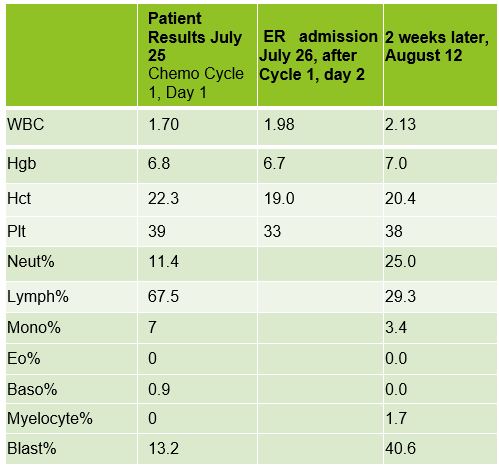
A 75 year old male first presented earlier this year with abnormal CBC results. The patient has a history of Type 2 diabetes, high blood pressure and atrial fibrillation. He was diagnosed with non-small-cell lung cancer (NSCLC) 6 years ago. His stage II NSCLC was completely removed with surgery. Surgery was followed up with adjuvant cisplatin-based chemotherapy to reduce the chance that the cancer would return. In June, he was referred to the hematology oncology department following consecutive CBCs that revealed leukopenia and thrombocytopenia. The CBC results from these specimens are shown below in Table 1.
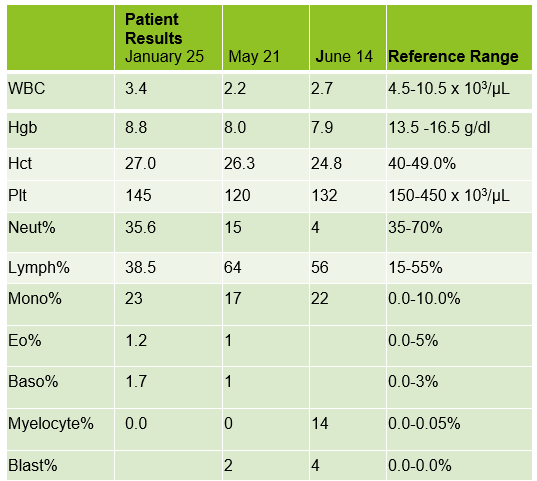
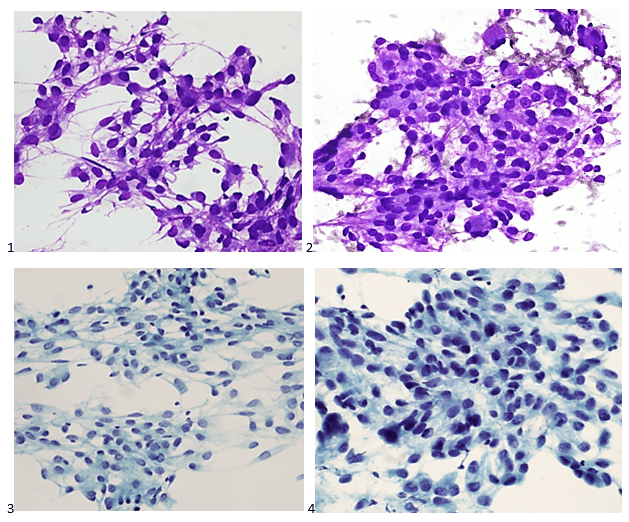
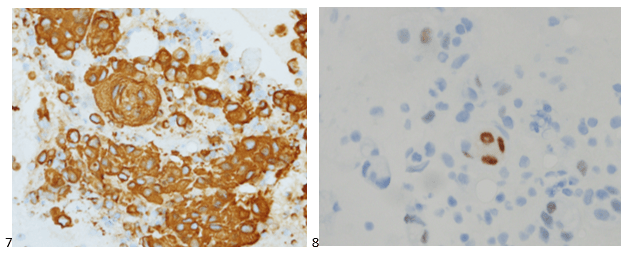
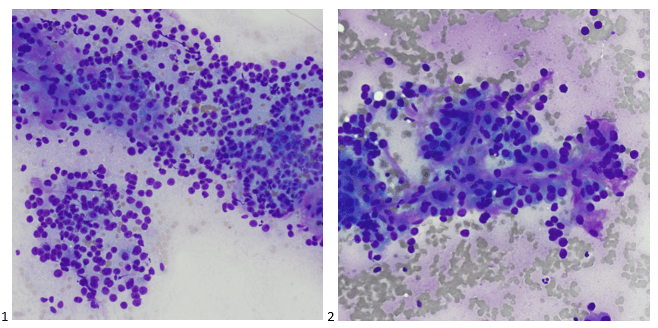
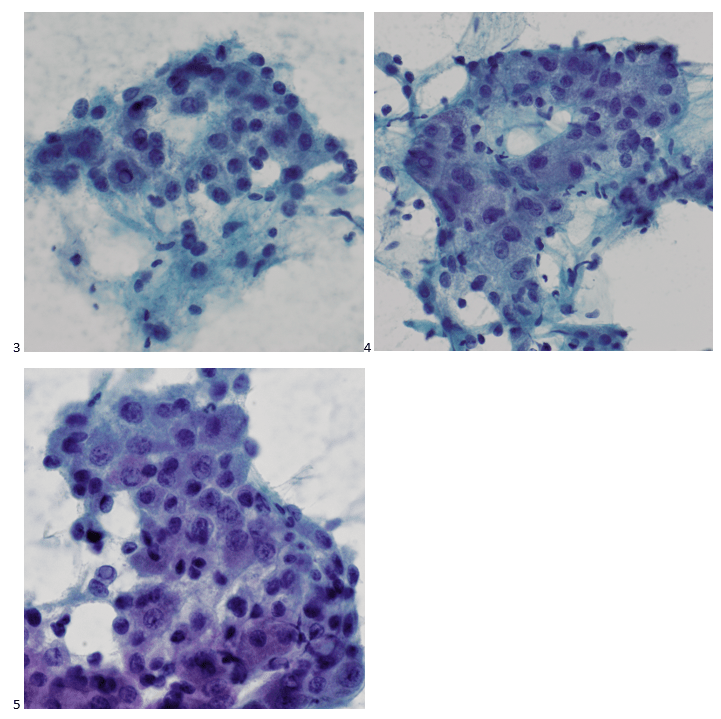
The peripheral blood sample from June was sent for flow cytometry. A leukemia/lymphoma phenotype was performed. Result comments noted proportionately decreased granulocytes with a left shift and 4% blasts. The blasts were CD34+, CD117+, HLA-DR+, CD13+ and CD33+ and were identified as myeloblasts. There were proportionately increased atypical monocytes with CD23 expression. Lymphocytes were also proportionately increased and included an increased population of CD57+, CD3+ T cells consistent with T-cell large granular (LGL) expansion. Immunophenotypic findings raised a concern for a myelodysplastic process. The hematologist discussed the findings with the patient and the patient was scheduled for a bone marrow biopsy. The procedure was performed 3 weeks later. CBC results on the day of the procedure are shown below in Table 2.
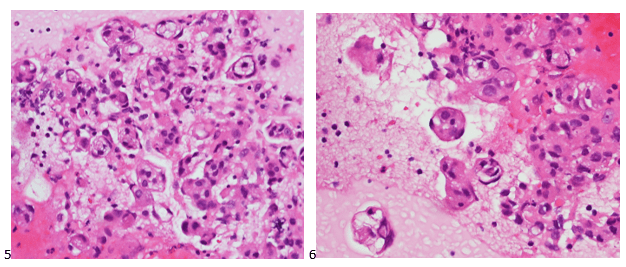
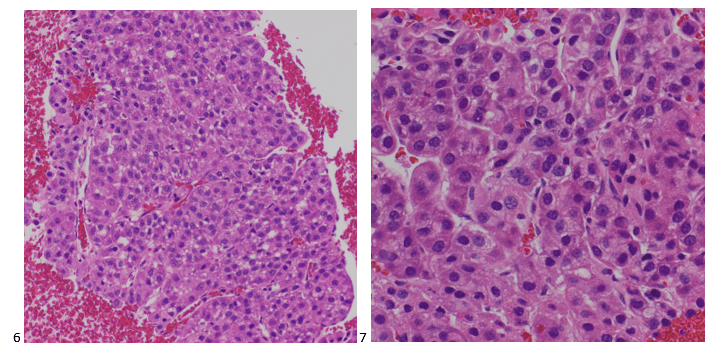
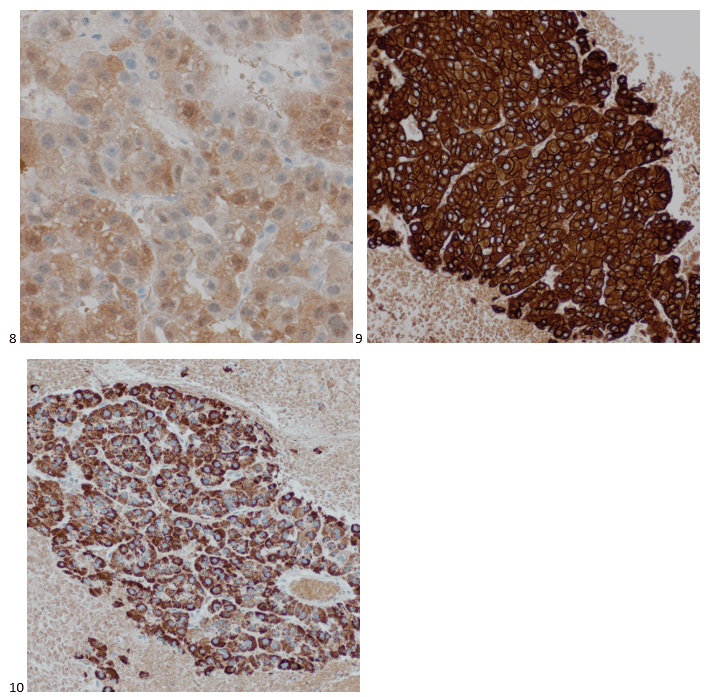
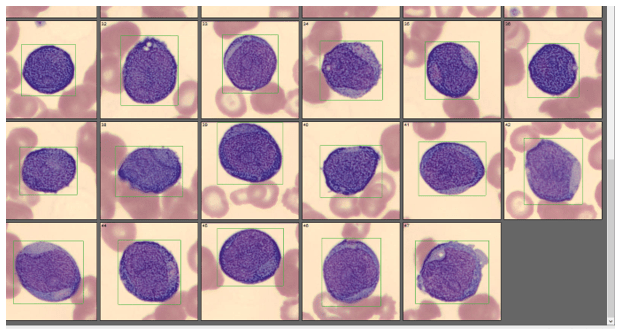
Bone marrow aspirate showed markedly increased myeloblasts (55%), consistent with acute myeloid leukemia (AML), nonacute promyelocytic leukemia (APL) type. The phenotype of the blasts was CD13+, CD33+, CD117+ and HLA-DR+. Blasts were negative for CD34. Several genomic variations were found in the specimen. These included variations in IDH2, SRSF2, STAG2 and ASXL1. Diagnosis: Increase in myeloblasts consistent with AML, nonAPL type.
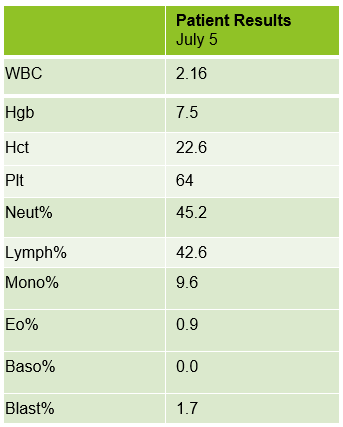
In July, 20 days after the bone marrow procedure and AML diagnosis, the patient was scheduled to begin his first cycle of Azacitidine (Vidaza). Based on his critical hemoglobin, the patient received 1 unit of packed RBCs followed by his first Vidaza injections. This Cycle 1, Day 1 chemotherapy was well tolerated, and he returned home. The following day he returned for his second treatment. His CBC showed good response to the previous day’s transfusion and his Cycle 1, Day 2 Vidaza was administered without incident. However, that evening the patient presented to the ER with nausea, vomiting and nose bleeds. The patient was admitted to the hospital and received another RBC transfusion. For the next several days the patient continued to do poorly, requiring additional RBC transfusions, and the Vidaza treatments were deferred, then discontinued. The patient had several ER visits and hospital admissions with transfusions over the next 2 weeks. During this time, we saw his blast% on his differential peak at over 60%. The patient was transferred to the palliative care team with care and comfort measures. CBC results from Cycle 1, Day 1 and subsequent CBC results are shown below. Note the sharp increase in blasts over a 2-week period.
AML is the most common acute leukemia in adults. In AML with minimal differentiation, evidence of bone marrow failure is characterized by anemia, neutropenia, and thrombocytopenia. The median age for patients with AML in the US is 66-67, and those who are older than 55-65 at diagnosis often have challenges and lower odds for long term survival. These older patients tend to have poor tolerance to traditional aggressive chemotherapy because of other health issues. This patient was likely not a good candidate for strong chemotherapy because of his age and health history. In these more fragile patients, Vidaza may be used. Vidaza is a class of drug called a hypomethylating agent that works by switching off DNA methyltransferase. This switches on genes that stop the cancer cells growing and dividing. The goal is to reduce the number of abnormal blood cells and to control cell growth.
As you can see from the CBC results, the onset of this patient’s AML was very abrupt, and the disease progressed rapidly. He has several risk factors that made him more likely to be diagnosed with AML. Older age is a risk factor for AML, and AML is more common in males than females. He has a history of smoking which is a behavioral risk factor associated with AML. Additionally, patients with cancer who are treated with certain chemotherapy drugs are more likely to develop AML in the years following treatment. This patient was treated with cisplatin following lung cancer surgery. Cisplatin is an alkylating agent which has been linked to an increased risk of AML.
Also interesting is the note on the peripheral blood phenotype interpretation that a T-cell large granular lymphocyte (LGL) expansion was present. These are an increased population of CD57+, CD3+ T cells. LGL clones have been described in AML and a hallmark of this association is cytopenia, as is observed in this patient. The patient’s poor prognosis can partly be attributed to the p.Gly646TrfsTer12 alteration in the ASXL1 gene, identified in the bone marrow interpretation. This alteration is associated with decreased overall survival and poor prognosis which was observed in this patient.
I work in a hospital with a large hematology/oncology practice, and we see a lot of adult leukemia patients. Many of the patients we see regularly have Chronic Lymphocytic Leukemia (CLL). We feel like we get to know these patients, because even though we never see them, we see their CBCs every week, sometimes for many years. This was an interesting case because it reminded me of the sudden onset and rapid progression of AML. It was amazing to see the differentials change so dramatically in a matter of weeks. This patient is currently receiving care and comfort end of life measures.
References
Fattizzo, B, Bellani, V, et al. Large Granular Lymphocyte Expansion in Myeloid Diseases and Bone Marrow Failure Syndromes: Whoever Seeks Finds. Front. Oncol., Sec. Cancer Immunity and Immunotherapy. 01 October 2021.
Pratcorona M, Abbas S, Sanders MA, Koenders JE, et al.Acquired mutations in ASXL1 in acute myeloid leukemia: prevalence and prognostic value. Haematologica. 2012 Mar;97(3):388-92. doi: 10.3324/haematol.2011.051532. Epub 2011 Nov 4. PMID: 22058207; PMCID: PMC3291593.
https://www.cancer.net/cancer-types/lung-cancer-non-small-cell/types-treatment
Thomas XG, Dmoszynska A, Wierzbowska, et al. Results from a randomized phase III trial of decitabine versus supportive care or low-dose cytarabine for the treatment of older patients with newly diagnosed AML. Journal of Clinical Oncology 29:2011
Turgeon, Mary Louis. Clinical Hematology Theory and Procedures, 6th ed, Jones and Bartlett Learning, 2017.

-Becky Socha, MS, MLS(ASCP)CMBBCM graduated from Merrimack College in N. Andover, Massachusetts with a BS in Medical Technology and completed her MS in Clinical Laboratory Sciences at the University of Massachusetts, Lowell. She has worked as a Medical Technologist for over 40 years and has taught as an adjunct faculty member at Merrimack College, UMass Lowell and Stevenson University for over 20 years. She has worked in all areas of the clinical laboratory, but has a special interest in Hematology and Blood Banking. She currently works at Mercy Medical Center in Baltimore, Md. When she’s not busy being a mad scientist, she can be found outside riding her bicycle.