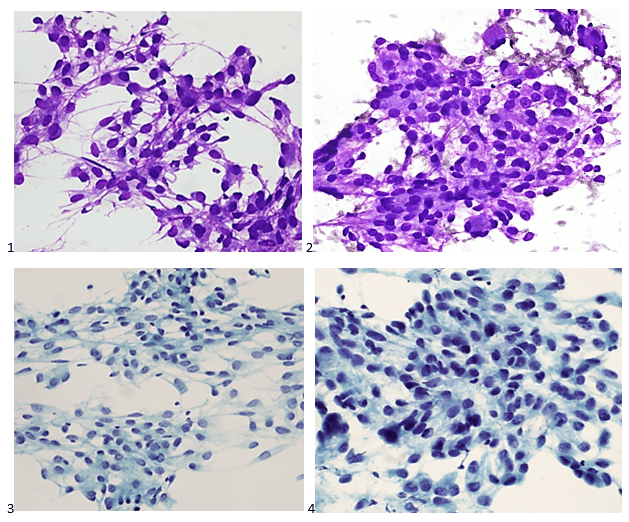
Back in my Master’s program at Jefferson, I fondly remember the week we covered central nervous system (CNS) tumors. I was fascinated by the mnemonic tools we would use to identify different CNS tumors, such as “fried eggs” for oligodendrogliomas, perivascular pseudorosettes in ependymomas, and the whorling associated with meningiomas. Fortunately, for our patients, and unfortunately, for our diagnostic curiosity, we rarely see CNS tumors at my institution. Brain lesions resulting from metastatic carcinomas are typically well-identified via imaging and treated appropriately by the surgical, medical, and radiation oncology teams, but cytologists are available to screen cerebrospinal fluids (CSFs) for CNS involvement. For primary CNS tumors, however, we’re left recollecting the core memory of the second semester of our didactic phase. When a metastatic CNS tumor made its way into our lab, our cytology team swooned with excitement. (Yes, I know, but please introduce me to a lab professional who doesn’t embrace their quirks.) A 27-year-old male patient presented to radiation oncology three years after surgical debulking of a brain tumor at an outside institution. The patient, who was referred to radiation oncology at to treat the residual tumor at the original institution, did not follow up and developed an 8 centimeter recurrence a year after the initial resection. At this point, the patient experienced complete vision loss and underwent a biparietal-occipital craniectomy. A repeat brain MRI was performed a year later, and once again, a large enhancing extra-axial mass was identified along with multiple smaller masses also increasing in size. The patient received radiation after worsening difficulty with ambulation. After almost completing the planned fractions of radiation, the patient elected to stop their radiation therapy due to worsening seizures. A left neck mass was identified six months prior, and while the mass had not grown or caused pain, the patient was referred to head and neck surgical oncology for evaluation. Surveillance imaging demonstrated an enlarged left level 5A lymph node, suggestive of metastatic disease. Multiple ultrasound-guided fine needle aspiration biopsies were obtained from the lymph node, and ROSE was performed. The Diff-Quik-stained and concurrent Pap-stained smears demonstrated lesional tissue, although everything from epithelioid histiocytes to spindle cell melanoma to a renal primary were considered as a differential. Based on the location, a salivary gland primary was also a possibility for this case. The streaked cytoplasm and pseudoinclusions in both smears were concerning for a metastasis of the patient’s primary CNS tumor, but we were still hesitating to make the call.
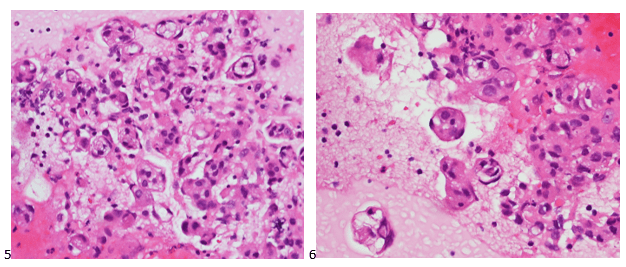
The following morning, the H&E-stained FFPE cell block sections demonstrated the characteristic whorls expected for the patient’s primary, although the idea of metastasis was uncanny.
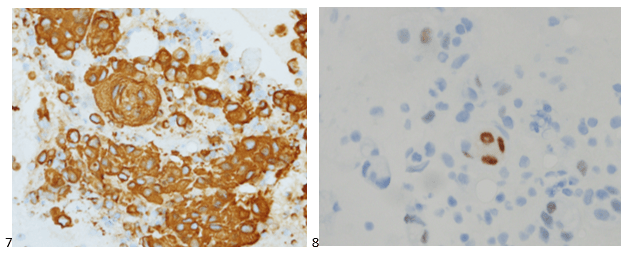
We then used immunohistochemical studies to confirm our morphologic diagnosis. Immunostains performed on the cell block slides with adequate controls show that the tumor cells are positive for vimentin and PR (focal), while negative for AE1/AE3, EMA, CK7, CK20, TTF-1, Napsin A, p40, Pax8, synaptophysin, and S-100. The Ki-67 proliferation index fell at 18%, which is consistent with intermediate aggressive disease in a WHO Grade 2 atypical meningioma.
The patient had next gen sequencing performed on his tissue, which demonstrated an NF-2 mutation, indicating he may benefit from MTOR inhibitors, but he elected not to pursue systemic therapy.
Where meningiomas account for 36% of primary brain tumors, atypical meningiomas comprise only 5-15% of all meningiomas (Cai et al., 202. Extracranial metastasis of atypical meningioma is a rare event, with only a few cases documented in the literature. While meningioma metastases are uncommon, a thorough collaboration between clinical impression and pathologic interpretation is necessary to ensure the possibility is not entirely excluded.
References
Cai C., Kresak J.L., Yachnis A.T. (2021) Atypical meningioma. Pathology Outlines. Retrieved October 11th, 2022, from https://www.pathologyoutlines.com/topic/cnstumoratypicalmeningioma.html.
P.S. I’d like to take this opportunity for a shameless plug. My Doctor of Health Science (DHSc) research survey is live now through November 23rd, 2022. If you’re a medical laboratory professional or pathologist, please consider contributing to our field of laboratory medicine! Click the following link to read the consent form and take the one-time anonymous survey. Thank you for your time!
https://www.surveymonkey.com/r/leadinglabs


-Taryn Waraksa-Deutsch, MS, SCT(ASCP)CM, CT(IAC), has worked as a cytotechnologist at Fox Chase Cancer Center, in Philadelphia, Pennsylvania, since earning her master’s degree from Thomas Jefferson University in 2014. She is an ASCP board-certified Specialist in Cytotechnology with an additional certification by the International Academy of Cytology (IAC). She is also a 2020 ASCP 40 Under Forty Honoree.
