Case history
A 73 year old woman presented with shortness of breath and was found to have bilateral pleural effusions. She had a history of marginal zone B-Cell lymphoma involving the bone marrow, which was diagnosed 3 months before this presentation and was treated with Rituximab.
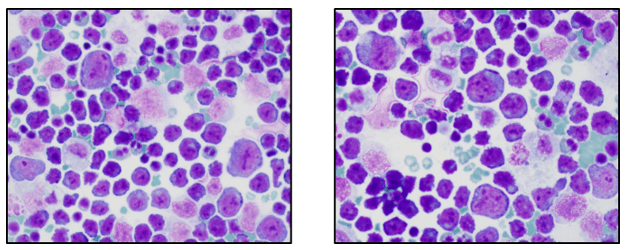
Thoracentesis revealed an atypical lymphoid population comprised of intermediate and large sized cells with eccentrically placed nuclei, multiple prominent nucleoli and scant to moderate amounts of basophilic cytoplasm (Image 1). Initial evaluation of the cytology material was concerning for large-cell transformation of the patient’s previously diagnosed marginal zone B cell lymphoma. A representative portion of the fine needle aspirate sample was sent for flow cytometric immunophenotyping.
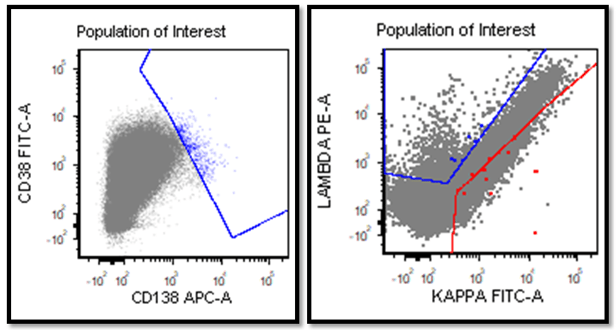
Flow cytometric immunophenotyping showed a distinct population of atypical cells with moderate CD45 expression and increased side scatter in keeping with cytoplasmic complexity (Figure 1, black arrows). On an initial screening B cell lymphoma panel these cells were negative for CD19 and positive for CD30 (partial), and CD44 (Figure 2).

The population of interest lacked expression of CD10, CD20, CD22 and surface immunoglobulin light chains and CD138 (Figure 2 and 3).

CD30 expression prompted the investigation of additional T-cell markers to rule out a T cell lymphoma (Figure 4). This population showed dim expression of CD7 but was otherwise negative for pan T cell markers (CD2, CD3, CD5) as well as CD4 and CD8 (Figure 4).

Given the unusual immunophenotype of the neoplasms, a diagnosis of diffuse large B cell lymphoma (transformation of the known marginal zone lymphoma) seemed less likely and other possibilities were considered.
The presence of CD30 expression and the plasmablastic morphologic features together with the clinical presentation with effusions raised the possibility of primary effusion lymphoma. IHC for anti-HHV8 was performed on the cell block sample (Image 2).

Final diagnosis
Primary effusion lymphoma (HHV8 positive).
Discussion
Primary effusion lymphoma (PEL) is a large B-cell neoplasm usually presenting as serous effusions without a detectable tumor mass [1]. It is universally associated with the human herpesvirus 8 (HHV8). It usually occurs in the setting of immunodeficiency [2]. Some patients with PEL secondarily develop solid tumors in adjacent structures such as the pleura [3-5].
Immunophenotype of PEL:
POSITIVE: CD45, HLA-DR, CD30, CD38, VS38c, CD138, EMA, HHV8 (LANA1).
NEGATIVE: pan- B-cell markers (CD19, CD20, and CD79a), surface and cytoplasmic Ig, and BCL6.
PEL is usually negative for T/NK-cell antigens, although aberrant expression of T-cell markers may occur. PEL is usually positive for EBV-encoded small RNA (EBER) by in situ hybridization but negative for EBV latent membrane protein 1 (LMP1) by IHC. This could be explained by EBV virus latency. It is ability of a pathogenic virus to lie dormant (latent) within a cell, denoted as the lysogenic part of the viral life cycle. EBV expresses its genes in one of three patterns, known as latency programs. EBV can exhibit one of three latency programs: Latency I, Latency II, or Latency III. Each latency program leads to the production of a limited, distinct set of viral proteins and viral RNAs. The Epstein-Barr virus encoded RNAs (EBERs): EBER1 and EBER2 are expressed during all latency forms [6], whereas LMP1 is expressed only in latency 2 and 3 rendering it a less sensitive marker for detection of EBV infection. EBV-negative PEL is common in elderly, HIV-negative patients from HHV8-endemic regions (Mediterranean) [7].
Differential Diagnosis
Most common cavities involved by PEL: pleural, pericardial, and peritoneal [8-10].
It was thought that PEL can involve an artificial cavity related to the capsule of a breast implant [11] although it was described only in one case report without appropriate HHV8 staining and before recognition of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL), which this case probably was presenting [12].
Primary effusion lymphoma (PEL) Prognosis
The prognosis is very unfavorable. Median survival is < 6 months. Rare cases have been reported that responded to chemotherapy and/or immune modulation [13].
Flow Cytometry Utility
The importance of utility of flow cytometry in establishing a diagnosis of PEL has been previously shown by others [14]. In the series by Galan et al. the authors described a case of PEL in an 88-year-old HIV-negative female with right-sided pleural effusion without significant lymphadenopathies or other effusions. The cytological study of the pleural fluid revealed a dense proliferation of large plasmablastic cells. A six-color multiparametric flow cytometry immunophenotyping study revealed 45% of large in size and high cellular complexity cells positive for CD45 (dim), CD38, CD138, CD30 and HLA-DR; and negative for CD19, CD20, cytoplasmatic CD79a, surface and cytoplasmic light chains Kappa and Lambda, CD3, CD4, CD5, CD7, CD8, CD28, CD56, CD81, and CD117. In situ hybridization for EBV-encoded small RNA was negative and immunohistochemistry for Kaposi sarcoma herpesvirus (HHV8) confirmed the diagnosis of PEL. These results in addition to the current case highlight the utility of flow cytometry in the diagnosis of lymphomas involving body cavities.
In Summary
PEL is associated with a proliferation of large B-cells which are positive for HHV8, CD45 (dim), CD30, CD38, and CD138 and negative for lineage defining B cell markers (CD19, CD20, and CD79a). Although PEL is a very rare lymphoma, it is important to consider it in patients with pleural, pericardial, and peritoneal effusions by sending a sample for cytological examination and flow cytometric immunophenotyping. Due to the absence of a mass lesion, cytology and flow cytometry are essential for establishing the diagnosis of PEL.
References
- Said, J.a.C.E., Primary effusion lymphoma, in WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (Revised 4th edition), C.E. Swerdlow SH, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Editor. 2017: Lyion. p. 323–324.
- Song, J.Y. and E.S. Jaffe, HHV-8-positive but EBV-negative primary effusion lymphoma. Blood, 2013. 122(23): p. 3712.
- Dotti, G., et al., Primary effusion lymphoma after heart transplantation: a new entity associated with human herpesvirus-8. Leukemia, 1999. 13(5): p. 664-70.
- Jones, D., et al., Primary-effusion lymphoma and Kaposi’s sarcoma in a cardiac-transplant recipient. N Engl J Med, 1998. 339(7): p. 444-9.
- Luppi, M., et al., Molecular evidence of organ-related transmission of Kaposi sarcoma-associated herpesvirus or human herpesvirus-8 in transplant patients. Blood, 2000. 96(9): p. 3279-81.
- Khan, G., et al., Epstein Barr virus (EBV) encoded small RNAs: targets for detection by in situ hybridisation with oligonucleotide probes. J Clin Pathol, 1992. 45(7): p. 616-20.
- Dupin, N., et al., Distribution of human herpesvirus-8 latently infected cells in Kaposi’s sarcoma, multicentric Castleman’s disease, and primary effusion lymphoma. Proc Natl Acad Sci U S A, 1999. 96(8): p. 4546-51.
- Otsuki, T., et al., Detection of HHV-8/KSHV DNA sequences in AIDS-associated extranodal lymphoid malignancies. Leukemia, 1996. 10(8): p. 1358-62.
- DePond, W., et al., Kaposi’s sarcoma-associated herpesvirus and human herpesvirus 8 (KSHV/HHV8)-associated lymphoma of the bowel. Report of two cases in HIV-positive men with secondary effusion lymphomas. Am J Surg Pathol, 1997. 21(6): p. 719-24.
- Beaty, M.W., et al., A biophenotypic human herpesvirus 8–associated primary bowel lymphoma. Am J Surg Pathol, 1999. 23(8): p. 992-4.
- Said, J.W., et al., Primary effusion lymphoma in women: report of two cases of Kaposi’s sarcoma herpes virus-associated effusion-based lymphoma in human immunodeficiency virus-negative women. Blood, 1996. 88(8): p. 3124-8.
- Lyapichev, K.A., et al., Reconsideration of the first recognition of breast implant-associated anaplastic large cell lymphoma: A critical review of the literature. Ann Diagn Pathol, 2020. 45: p. 151474.
- Ghosh, S.K., et al., Potentiation of TRAIL-induced apoptosis in primary effusion lymphoma through azidothymidine-mediated inhibition of NF-kappa B. Blood, 2003. 101(6): p. 2321-7.
- Galan, J., et al., The utility of multiparametric flow cytometry in the detection of primary effusion lymphoma (PEL). Cytometry B Clin Cytom, 2019. 96(5): p. 375-378.
This case was previously presented by authors as eCSI Case on International Clinical Cytometry Society website. For more information please follow: https://www.cytometry.org/public/newsletters/eICCS-10-1/article7.php

-Dr. Loghavi is an Assistant Professor of hematopathology and molecular pathology MD Anderson Cancer Center in Houston, TX. She received her MD degree from the Azad University in Tehran, Iran. Shen then completed an Anatomic and Clinical Pathology residency training at Cedars Sinai Medical Center in Los Angeles, CA, followed by Surgical pathology, Hematopathology and Molecular pathology fellowship training at the University of Texas, MD Anderson Cancer Center. Dr. Loghavi is passionate about medical education. Her clinical and research interests are focused on hematologic malignancies, with particular focus on myeloid neoplasm and the applications of flow cytometric immunophenotyping and molecular methods in detection of minimal/measurable residual disease. She has authored 100 peer-reviewed articles, 5 book chapters, and numerous abstracts in the fields of hematopathology and molecular pathology.

-Kirill Lyapichev, MD, FASCP, is a board-certified anatomical and clinical pathologist who completed 2 years of hematopathology fellowship at MD Anderson Cancer Center. He is currently a molecular genetic pathology fellow at MD Anderson Cancer Center. Additionally, he is interested and involved in other research projects including neoplastic as well as non-neoplastic entities: MALT lymphoma, Castleman Disease, Kikuchi-Fujimoto Disease, and others. In 2020 he was selected as one of ASCP’s 2020 Top 40 Under Forty. Follow him on Twitter: @KirillLyapichev.
