While writing my last blog, I asked “What is your least favorite test to do in Hematology?” (I’m not ignoring our favorite tests! I will get to those in another blog.) And then, I started thinking about why we may not like certain testing. Is it because they are time consuming, or repetitive? Is it because they hurt our eyes, or necks, or fingers? Or is it because it’s a test that we perform but we may not be sure what the test is for, or we don’t understand the theory behind it? I started thinking about my coworkers and other tests that could be on those lists and I immediately decided that a good candidate in our lab is the T4T8 panel. Probably the primary reason is that the instrument we do these on has given us many problems over the last year. The instrument has spent most of the year with an “instrument out of service” sign on it. Service has been here many times, but the instrument just appears to have exceeded its life expectancy. In normal times, when the instrument was in its prime, setting up and running a T4T8 panel does require a number of steps, and some time. In the last year we have had to add lots of coaxing, even more time, and some luck to get the test to run. This can be frustrating in any lab situation, but is particularly frustrating when we are short staffed, trying to train new staff, and very busy. So, I don’t think it’s the test itself that techs dislike, it’s the time it takes, not being comfortable with setting up the test, juggling our other work while struggling with another instrument, and the fact that even after we get results, a percentage of the samples have results that don’t meet our criteria and still need to be sent out to the reference lab.
Another reason why this test may be a little intimidating is its unfamiliarity. It’s not a test that is done in every lab. I have worked as a Medical Laboratory Scientist for many years. I’ve worked in 6 labs since the mid 1980’s and the introduction of CD4 testing for human immunodeficiency virus (HIV) patients, yet my current place of work is the first place that we have done these in house. Before this job, if you asked me or any of my coworkers what a T4T8 panel was, we probably would have answered “a send out test”. A few weeks ago, we had a call from a doctor asking questions about his patient’s T4T8 assay results. The tech answering the phone got a blank look on their face and quickly handed the phone to me. This told me that techs, and even doctors, may not really understand what this test is testing and what the results mean. This further confirmed to me that the lack of knowledge about these tests may be another reason why these don’t win any popularity contests in our lab.
So, what exactly is a T4T8 panel?
Some other names for the test are a Lymphocyte subset panel, an Immuno T-cell (CD3/4/8) assay, T-Cell subsets Percent and Absolute panel or T-Lymphocyte Helper/Suppressor Panel. As a quick review, we know that lymphocytes are either B-lymphocytes or T-lymphocytes. Immunotyping lymphocytes can provide information for disease diagnosis and monitoring. All T-lymphocytes express CD3 antigens on their surfaces, which can be used to differentiate B-cell disorders from T-cell disorders. T-lymphocyte subsets include T-helper/inducer cells which express both CD3 and CD4, and T-cytotoxic/suppressor cells, which express CD3 and CD8. In a T4T8 panel we are concerned with identifying T-lymphocytes, and the percentage of each subset both individually, and compared to one another.
The test we perform uses monoclonal antibodies, anti CD3, anti CD4 and anti CD8, which recognize specific human lymphocyte subsets. Our reagents come as antibody containing tubes and are run on the Cell-Dyn Sapphire. After performing a CBC on the sample, the instrument is programmed to add an aliquot of the sample to the CD3 +CD4 reagent tube and a second aliquot to the CD3 + CD8 reagent tube. Immunophenotyping is performed by flow cytometry on these 2 aliquot tubes. The CD3 antibody in both tubes separates out all T-lymphocytes, and the addition of the CD4 in the first tube identifies the cells which are also CD4 positive, the T4 or helper cells. The CD3 + CD8 tubes identifies the percentage of T cells that are T8 or suppressor cells. The assay uses the CBC results and the immunophenotyping runs to calculate the helper/suppressor ratio, also known as T4/T8 ratio or CD4/CD8 ratio.
Why is this test performed?
After the discovery of lymphocyte subset abnormalities in human immunodeficiency virus (HIV) patients in the 1980s, lymphocyte immunophenotyping has become widely used in this patient population for the evaluation of their prognosis, immune deficiency status, response to therapy, and diagnosis of AIDS. The test is most often done to assess HIV infection status but may also be useful in the diagnosis and monitoring of other diseases or after organ transplantation. Some examples of conditions in which this assay may be useful include other viral and bacterial infections, severe combined immunodeficiency, Hodgkin disease, certain leukemias, multiple sclerosis, and myasthenia gravis. A newer application of CD4/CD8 ratios are as potential biomarkers of cancer progression. The most interesting new use of T-cell subset testing that I have read about has been with the recent COVID-19 pandemic. Several studies have shown that CD4 and CD8 T- cell counts reflected disease severity and can predict clinical outcomes of COVID-19 infection. These studies have concluded that COVID-19 patients presenting with relatively low CD4 and CD8 T-cell counts are more severely infected and may have a worse prognosis. The Abbott test we use was designed to be used to monitor immune status in (HIV)-infected individuals. It is not intended for screening for leukemic cells or for phenotyping samples in leukemia patients.
What do the results mean?
The absolute CD4 count and CD4/CD8 Ratio can be used as a snapshot of immune system health. Normal absolute CD4 counts are 600 to 1200 /mm3. In immune suppression, values drop below 500/mm3 and in advanced infection, values of less than 200/mm3 are consistent with a definition of acquired immunodeficiency syndrome (AIDS). In advanced disease, some patients may have a normal CD4 count but experience a weakening immune system. Or the immune system can become exhausted and unable to produce sufficient T-cells. The CD4/CD8 ratio is useful for judging the strength of the immune system. A normal CD4/CD8 ratio is between 1.0 and about 3.0-4.0.
T-helper cells start the defensive immune response by signaling other cells that infectious pathogens are present. At initial infection with HIV, T-suppressor cells increase in an effort to destroy infected cells. We see an increase in CD8 cells as the CD4 cells are destroyed. These events result in a low CD4/CD8 ratio. When HIV antiretroviral therapy (ART) is initiated, the ratio will usually, gradually return to normal. However, if ART is not started or if the immune system is severely affected, the body may not be able to make adequate new CD4 cells and the ratio may never return to normal.
With the availability of very effective therapies available for the treatment of HIV, the CD4/CD8 ratio has become more important in patients with long term HIV infection. Recent studies have suggested that people with a low CD4/CD8 ratio who have been on treatment for years are at an increased risk from non-HIV illnesses such as cardiovascular and renal disease.
CD4 counts are important in HIV management and used to guide treatment including the decision to initiate prophylactic treatment against opportunistic infections. It is recommended that CD4 counts be performed every 3-6 months after initiation of ART. After the first 2 years on ART, CD4 monitoring can be decreased in frequency to every 12 months for people whose CD4 count is between 300 and 500 and may be considered optional for those with CD4 counts over 500. Table 1 and 2 shown below are examples of patient reports for the T4T8 assay.

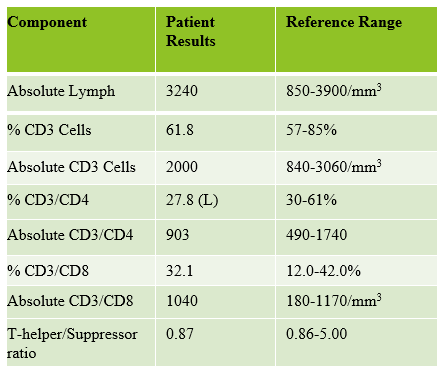
*There are times when the absolute or % CD3T may be less than the sum of the CD4T and CD8T. This is due to averaging of CD3T counts from the 2 monoclonal tubes
In our lab, these tests are performed daily, as they are received, from 7am to 7PM, 7 days a week. There are no commercial quality control materials available for the test, so we must choose negative and positive QC from our patient population. For the QC we choose patients with CBC and WBC differential values within normal ranges, with no flags. There are additional age and diagnosis/treatment related restrictions on samples that can be used as controls. Our in-house patients often have abnormal results, and our patient population also includes our large outpatient hematology/oncology center. Thus, at times, finding appropriate controls can be challenging. I can add this to the list of ‘problems’ with this test and why techs don’t like them. Call me weird, but I actually like doing these! I like the challenge of finding QC, I like that they are ‘different’ from the hundreds of CBCs we perform each day, and I look at them as a little change in routine and a chance to do something unique. Though I wish the instrument would run perfectly every day, I even (sort of) don’t mind troubleshooting when it’s not working. I like solving problems! I enjoy teaching others how to run these, and I enjoy answering questions about the test.
Many thanks to my great coworker Jacky Olive for her assistance always and inspiration for this blog. I know these are not your favorite test!
*There are times when the absolute or % CD3T may be less than the sum of the CD4T and CD8T. This is due to averaging of CD3T counts from the 2 monoclonal tubes
Becky Socha MS, MLS(ASCP)CMBB
References
- Abbott Laboratories, Cell Dyn Immuno T-Cell (Cd3/4/8 )ReagentsPackage Insert. Abbott Park, Il.
- Li Raymund; Duffee Doug; Gbadamosi-Akindele Maryam F.CD4 Count. NIH National Library of Medicine. May 8, 2022
- Domínguez-Domínguez L, Rava M, Bisbal O, et al. Cohort of the Spanish HIV/AIDS Research Network (CoRIS). Low CD4/CD8 ratio is associated with increased morbidity and mortality in late and non-late presenters: results from a multicentre cohort study, 2004-2018. BMC Infect Dis. 2022 Apr 15;22(1):379.
- Liu Z, Long W, Tu M et al. Lymphocyte subset (CD4+, CD8+) counts reflect the severity of infection and predict the clinical outcomes in patients with COVID-19. Journal of Infection. Vol 81, Issue 2. P318-356, AUGUST 01, 2020
- Kagan JM, Sanchez AM, Landay A, Denny TN. A Brief Chronicle of CD4 as a Biomarker for HIV/AIDS: A Tribute to the Memory of John L. Fahey. For Immunopathol Dis Therap. 2015;6(1-2):55-64
- McBride JA, Striker R (2017) Imbalance in the game of T cells: What can the CD4/CD8 T-cell ratio tell us about HIV and health? PLoS Pathog 13(11)
- Sinha A, Mystakelis H, Rivera AS, Manion M, et al. Association of Low CD4/CD8 Ratio With Adverse Cardiac Mechanics in Lymphopenic HIV-Infected Adults. J Acquir Immune Defic Syndr. 2020 Dec 1;85(4)
- Wang YY, Zhou N, Liu HS, Gong XL, Zhu R, Li XY, Sun Z, Zong XH, Li NN, Meng CT, Bai CM, Li TS. Circulating activated lymphocyte subsets as potential blood biomarkers of cancer progression. Cancer Med. 2020 Jul;9(14)

-Becky Socha, MS, MLS(ASCP)CMBBCM graduated from Merrimack College in N. Andover, Massachusetts with a BS in Medical Technology and completed her MS in Clinical Laboratory Sciences at the University of Massachusetts, Lowell. She has worked as a Medical Technologist for over 40 years and has taught as an adjunct faculty member at Merrimack College, UMass Lowell and Stevenson University for over 20 years. She has worked in all areas of the clinical laboratory, but has a special interest in Hematology and Blood Banking. She currently works at Mercy Medical Center in Baltimore, Md. When she’s not busy being a mad scientist, she can be found outside riding her bicycle.

I have actually been to your lab! Small world.