Leukemic Reticuloendotheliosis (LRE) is a term that was first used in 1923 but is a name that most of us would not recognize today. In 1958, Bournocle et al. published a paper that characterized LRE as a separate clinical disorder and described the clinical course, pathologic features, treatment options and prognosis. The study also described an unusual morphology of the malignant cells seen in this condition. The malignant cells were noted to be small mononuclear cells with projections around the circumference of the cytoplasm. Another decade went by before these cells were given the nickname “hairy cells”. At the time, though LRE was considered a fatal disease, splenectomy appeared to be a beneficial treatment, thus pointing to a lymphocytic disorder. Later, in 1976, an article was published that suggested that these hairy cells were monocytic rather than lymphocytic in origin. The true lineage of these hairy cells was unknown until the development of newer immunophenotypic methodologies in the mid to late 1970s. Today, hairy cell leukemia (HCL) is considered a rare, chronic B cell leukemia that comprises 2% of lymphoid leukemias and responds well to therapy.
Patients may be entirely asymptomatic at diagnosis, and the finding of hairy cells on the peripheral smear from a routine CBC prompts further investigation. Patients do not usually require treatment at diagnosis, and many patients live a normal lifespan. Originally, diagnosis was based on clinical and laboratory result correlation: CBC results, observation of the characteristic hairy cells, and splenomegaly. One of the first tests used for diagnosis of HCL was tartrate-resistant acid phosphatase activity (TRAP stain). Today, standard practice is immunophenotyping by flow cytometry. HCL is characterized by the expression of B-cell antigens CD19, CD20, and CD22 in addition to bright CD11c expression with CD103, CD25, CD123 and Annexin A1 (ANXA1) co-expression. Annexin A1 is the most specific immunohistochemical marker for HCL. In 2011, the BRAF-V600E mutation was identified as the genetic causal event of HCL, allowing even more advances in the diagnosis and therapy for HCL.
As the disease progresses, most patients experience increasing cytopenia, including monocytopenia, and persistent splenomegaly. Treatment is usually started when a patient meets certain guidelines, which include a severe cytopenia or pancytopenia, malignant lymphocytosis, increased susceptibility to infection or symptomatic splenomegaly. Historically, the only available treatment was splenectomy. In the 1980’s, interferon therapy was introduced and was able to induce partial responses in some patients. In the 1990’s the purine analogs, cladribine or pentostatin, became available as the preferred first line treatment for HCL. Treatment response is good and offers prolonged remission rates. For patients who experience relapse, rituximab may be used in combination with a purine analog. Most recently, anti-CD22 immunotoxins and molecular targeted therapy with BRAF inhibitors have been introduced for cases that do not respond to other therapies.
Additional discoveries into the biology of the disease have identified new subtypes of HCL. It is important to distinguish between classic HCL and Hairy Cell leukemia variant (HCLv) because they are treated differently. HCLv may be more aggressive and does not respond well to purine analogs alone. HCLv is often diagnosed at older age than classic HCL In HCLv the WBC is often elevated, with lymphocytosis, and there is a lack of monocytopenia. The hairy cells seen on a peripheral blood smear may be more abundant than in classic HCL. These HCLv cells also often have a distinct nucleolus not seen in HCL cells. As well, these cells can have a morphology that appears to be somewhere between prolymphocytes and hairy cells. Unlike HCL, HCLv cells are negative for CD25 and BRAF-V600E. HCLv represents only about 10% of HCL cases. Because of its rarity, and the gray areas surrounding differential diagnosis between HCL and HCLv, studying these rare cases can help lead to a better understanding and management of both HCL and HCLv patients.
About 1200 new cases of HCL are diagnosed each year in the US. HCL is 4-5 times more common in males, with a median age at diagnosis of 55-60. This is an unusual case because the patient is female, was older at diagnosis, with no cytopenia or splenomegaly noted. This patient is a 79-year-old female who, one year ago, was referred to a Hematology Oncology practice with a several year history of a mildly elevated WBC with increased lymphocytes, without absolute lymphocytosis. She was referred after a peripheral smear exhibited prolymphocytes and the “hairy’ appearing lymphocytes shown below in Image 1.
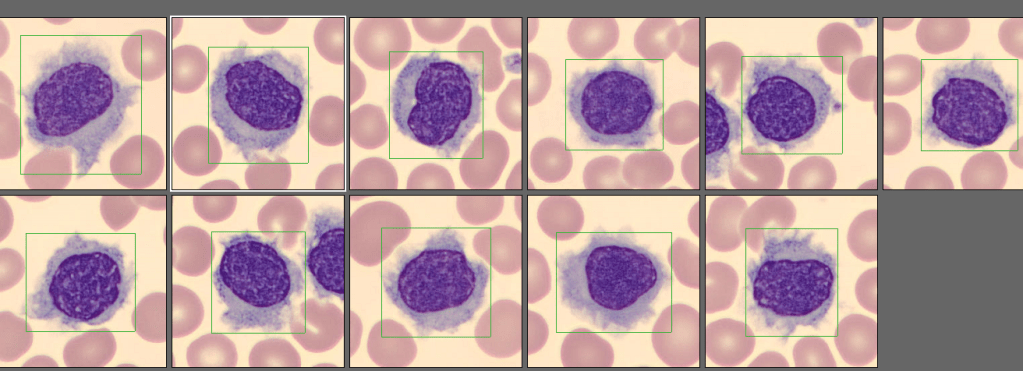
Peripheral blood was sent for myeloid/lymphoid disorders and acute leukemia analysis by flow cytometry. Remarkable in this case were the results of the flow cytometry studies. Flow cytometry performed on the peripheral blood revealed 2 distinct morphological populations of lymphocytes. The majority of lymphs appeared to be small, with scant cytoplasm, round nucleus, clumped chromatin, and inconspicuous nucleoli. These cells were identified as a monoclonal kappa restricted B cell population exhibiting co-expression of CD23 and CD5, consistent with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL). A second population of lymphs were larger, with more abundant granular cytoplasm and hairy projections, large nuclei, condensed chromatin, and inconspicuous nucleoli. This second population displayed CD20 expression and was positive for CD11c, CD103 and FMC-7. CD25, CD5 and CD23 were negative.
The immunophenotyping of this second population of cells suggests a diagnosis HCL; or is it suggestive of HCLv? The patient was older at diagnosis, leukocytosis and lymphocytosis are present, and monocytopenia is absent. Hairy cells were over 8% of the differential, though lacking the distinct nucleoli of HCLv. Prolymphocytes were noted. CD25 was negative in this patient and is usually exhibited in HCL.
An immunological scoring system for HCL has been proposed with one point given to each of markers for CD11c, CD103, CD123 and CD25. One point is given if the marker is expressed and no point when it is not expressed. A score of 3 or 4 is observed in 98% of cases of HCL and is usually 0-1 in other HCL-like disorders. This patient’s cells showed expression of CD11c and CD103, was CD25 negative and CD123 was not evaluated so would score at least a 2, which puts her somewhere in an inconclusive score. Additionally, a bone marrow biopsy has not been done and there therefore results for TRAP or annexin A1 immunostaining, or BRAF-V600E mutations are not available.
With a diagnosis of a B-lymphocytosis consistent with CLL/SLL and a simultaneous HCL, or HCLv, this patient is an interesting case. Several articles and reviews in literature of other patients with CLL and HCL give further insight into the biology of HCL. Literature suggests that concurrent HCL and CLL may indicate a common origin. Patients with HCL may subsequently develop CLL, which can mimic a relapse of HCL. Therapy requires treating each case individually and watchful waiting in asymptomatic cases. Rituximab with or without purine analogs have been useful to treat both disorders simultaneously.
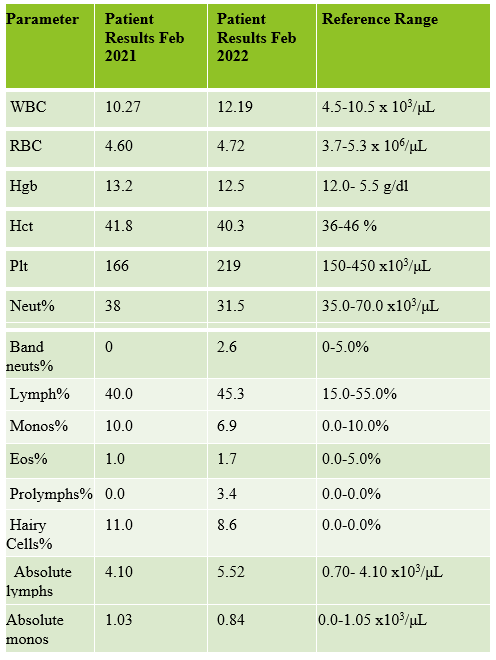
This patient at 1 year following diagnoses has developed a mildly increasing lymphocytosis and is being monitored. Both her CLL/SLL and HCL still appear to be in the indolent, “wait and see” stage. The patient has declined further workups at this time.
References
- Bain, Barbara J. Blood Cells: A Practical Guide. 5th ed. Wiley Blackwell, 2015. Print.
- Chang-Hun Park, Hyun-Young Kim, M.D.et al. Efficacy of Annexin A1 Immunostaining in Bone Marrow for the Diagnosis of Hairy Cell Leukemia. Laboratory Medicine Online 2019; 9(4): 236-241
- Falini B, Tiacci E. New treatment options in hairy cell leukemia with focus on BRAF inhibitors. Hematol Oncol. 2019; 37(Suppl. 1): 30– 7..Maitre, E.; Cornet, E.; Troussard, X. Hairy cell leukemia: 2020 update on diagnosis, risk stratification, and treatment. Am. J. Hematol. 2019, 94, 1413–1422.
- Obiorah IE, Francischetti IMB, Wang HW, Ahn IE, Wang W, Raffeld M, Kreitman RJ, Wiestner A, Calvo KR. Concurrent chronic lymphocytic leukemia/small lymphocytic lymphoma and hairy cell leukemia: clinical, pathologic and molecular features. Leuk Lymphoma. 2020 Dec;61(13):3177-3187.
- Scheinberg M, Brenner AI, Sullivan AL, Cathcart ES, Katayama I. The heterogeneity of leukemic reticuloendotheliosis, “hairy cell leukemia”. Evidence for its monocytic origin. Cancer. 1976 Mar;37(3):1302-7
- Shao, Haipeng et al. “Distinguishing hairy cell leukemia variant from hairy cell leukemia: development and validation of diagnostic criteria.” Leukemia research vol. 37,4 (2013)
- Verma V, Giri S, Bhatt VR, Amador-Ortiz C, Armitage JO. Synchronous or Metachronous Hairy Cell Leukemia and Chronic Lymphocytic Leukemia: A Case Series and Literature Review. Front Oncol. 2017 Jan 9;6:270.
- X. Troussard, M.R. Grever. The revised guidelines for the diagnosis and management of hairy cell leukaemia and the hairy cell leukaemia variant. r J Haematol, 193 (1) (2021), pp. 11-14

-Becky Socha, MS, MLS(ASCP)CMBBCM graduated from Merrimack College in N. Andover, Massachusetts with a BS in Medical Technology and completed her MS in Clinical Laboratory Sciences at the University of Massachusetts, Lowell. She has worked as a Medical Technologist for over 40 years and has taught as an adjunct faculty member at Merrimack College, UMass Lowell and Stevenson University for over 20 years. She has worked in all areas of the clinical laboratory, but has a special interest in Hematology and Blood Banking. She currently works at Mercy Medical Center in Baltimore, Md. When she’s not busy being a mad scientist, she can be found outside riding her bicycle.
