Case History
A 67 year old male presented with type II diabetes, hypertension, hyperlipidemia, obstructive sleep apnea, recurrent GI bleeds, and atrial fibrillation (status post ablation and on rivaroxaban). Given the history of recurrent GI bleeds, he was taken off rivaroxaban and underwent a left atrial appendage occluder device implant procedure. Several hours later, he developed severe leg pain and loss of lower extremity pulses. CT angiogram confirmed Watchman device embolization to the abdominal aorta. The patient received emergent surgical removal of the device. In the ICU, the patient developed worsening rhabdomyolysis, anuria, hypotension, ischemic bowel disease, and died within hours. An autopsy was requested by the next of kin, which revealed an unexpected finding of a 6 cm hilar-based lung mass.
Microbiology
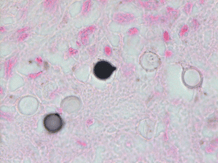
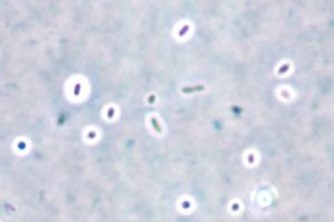
Premortem and postmortem cultures were not collected. Fontana-Masson stain of a section from the hilar lung tissue reveals yeast of varying size with a lighter shade in the center and a thick capsule, though the capsule does not stain and can instead be appreciated as a “halo” (Image 1). Fontana Masson stain also reveals narrow based budding (Image 2). Both findings indicate Cryptococcus neoformans.

Discussion
Cryptococcus neoformans is a saprophytic yeast (5-10 µm) identified best by its thick polysaccharide, antiphagocytic capsule. It can be infectious when inhaled, often from soil or avian droppings.
While most immunocompetent individuals clear the pathogen, in the immunocompromised, it can form a primary focus in the lungs and then disseminate. It is often asymptomatic when localized to the lungs but can present as a cough or dyspnea. Dissemination to the brain presents as meningitis. Cryptococcal neoformans is the most common cause of fungal meningitis.
While C. neoformans most often presents as meningitis in the immunocompromised, a retrospective case analysis found diabetes mellitus II as a newly defined independent factor contributing to morbidity and mortality. This study analyzed cryptococcal infections in patients with DMII from 1997-2015. 57% of the DMII patients did not have any other underlying disease and 69% of patients who presented with pulmonary Cryptococcus neoformans experienced a misdiagnosis and treatment delays.1
Qualities that aid in the diagnosis include urease positivity, positive latex agglutination test due to its thick polysaccharide capsule, and characteristic features on mucicarmine red, methenamine silver, India ink, and Fontana-Masson stains.2
The Fontana-Masson silver (FMS) stain is a histochemical technique that oxidizes melanin and melanin-like pigments as it reduces silver. FMS can be used to highlight the melanin-like pigment in Cryptococcus spp., including capsule-deficient variants because this pigment is cell-wall (and not capsule) associated. FMS is a very sensitive, but not completely specific stain, for Cryptococcus spp. as other yeasts and fungi can also produce melanin and melanin-like pigments.3 Though two C. neoformans yeast close together can resemble broad-based budding, Image 2 demonstrates the narrow-based budding.
While the India ink stain is often discussed as a popular stain for C. neoformans, it can only be performed on liquid samples (CSF, fluid samples) and cannot be performed on paraffin-embedded tissue samples. Of note, the India ink stain is a “negative stain”, resulting in the classic “halo” effect (image 3) because it is not picked up by the capsule of Cryptococcus spp. Because of this, it will miss capsule-deficient infections.4
Prognosis varies by the mechanism of immunosuppression. Acute mortality in in cryptococcal meningitis for HIV patients has improved dramatically with antifungals and ART, ranging from 6-16%. Poor prognostic indicators include abnormal mental status, a high yeast burden defined as CSF antigen titer > 1:1024 by latex agglutination or > 1:4000 by lateral flow assay, or a poor host response defined as CSF WBC count < 20/microL.5
Treatment of cryptococcal infections includes initial therapy with amphotericin B and flucytosine followed by long term fluconazole.6
References
- Boulware DR, Rolfes MA, Rajasingham R, von Hohenberg M, Qin Z, Taseera K, Schutz C, Kwizera R, Butler EK, Meintjes G, Muzoora C, Bischof JC, Meya DB. Multisite validation of cryptococcal antigen lateral flow assay and quantification by laser thermal contr. (n.d.).
- Li Y, Fang W, Jiang W, Hagen F, Liu J, Zhang L, Hong N, Zhu Y, Xu X, Lei X, Deng D, Xu J, Liao W, Boekhout T, Chen M, Pan W. Cryptococcosis in patients with diabetes mellitus II in mainland China: 1993-2015. Mycoses. 2017 Nov;60(11):706-713. doi: 10.1111/. (n.d.).
- McFadden, D., & Casadevall, A. (2001). Capsule and Melanin Synthesis in Cryptococcus neoformans. Medical Mycology, 39, 19-30.
- Perfect JR, Dismukes WE, Dromer F, Goldman DL, Graybill JR, Hamill RJ, Harrison TS, Larsen RA, Lortholary O, Nguyen MH, Pappas PG, Powderly WG, Singh N, Sobel JD, Sorrell TC. Clinical practice guidelines for the management of cryptococcal disease: 2010 up. (n.d.).
- Saag MS, Powderly WG, Cloud GA, Robinson P, Grieco MH, Sharkey PK, Thompson SE, Sugar AM, Tuazon CU, Fisher JF, et al. Comparison of amphotericin B with fluconazole in the treatment of acute AIDS-associated cryptococcal meningitis. The NIAID Mycoses Study. (n.d.).
- Winn, W. C., & Koneman, E. W. (2006). Koneman’s color atlas and textbook of diagnostic microbiology. Philadelphia: Lippincott Williams & Wilkins. (n.d.).
-Joe Teague is a Pathology Student Fellow and Brianna Waller, MD is a 3rd year Anatomic and Clinical Pathology Resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Associate Professor at the University of Vermont.