Case History
An 83 year old female with a past medical history of breast cancer, multiple strokes, dysphagia, hypertension and gastroesophageal reflux disease (GERD) presented to an outside hospital with altered mental status, metabolic encephalopathy, decreased appetite, acute kidney injury, and E. coli/Proteus urinary tract infection (UTI). There, she was diagnosed with a perforated gastric ulcer, which was repaired, with a gastrostomy (G) tube in place. The patient later developed a fever and an aspiration pneumonia, which was treated with ampicillin/sulbactam. A follow up imaging revealed a new gastric perforation along the fundus of the stomach with perisplenic fluid collection, along with a pleural effusion with possible communication with the fluid collection in the stomach. Due to her complex medical history, an additional intervention was not pursued and the family agreed to comfort measures, and the patient was discharged home.
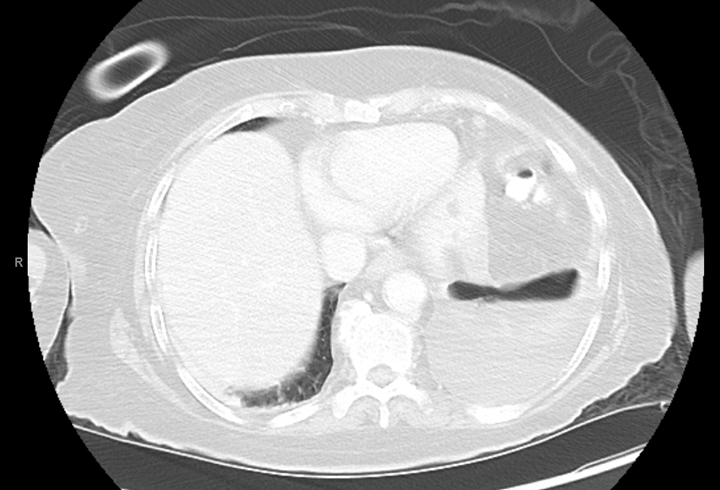
The family presented to our emergency department the same day of discharge, as the patient had not been eating and the family needed assistance in using the G tube. In-house computed tomography (CT) of the abdomen/pelvis again showed an organizing collection near the spleen (Image 1). A medium-sized left pleural effusion with left lower lobe collapse due to the communication with the perforation was observed on CT. The patient received IV fluids and a dose of vancomycin and zosyn in the ED. A vascular and interventional radiology (VIR) consult was recommended for potential drainage of the perisplenic abscess and left pleural effusion.

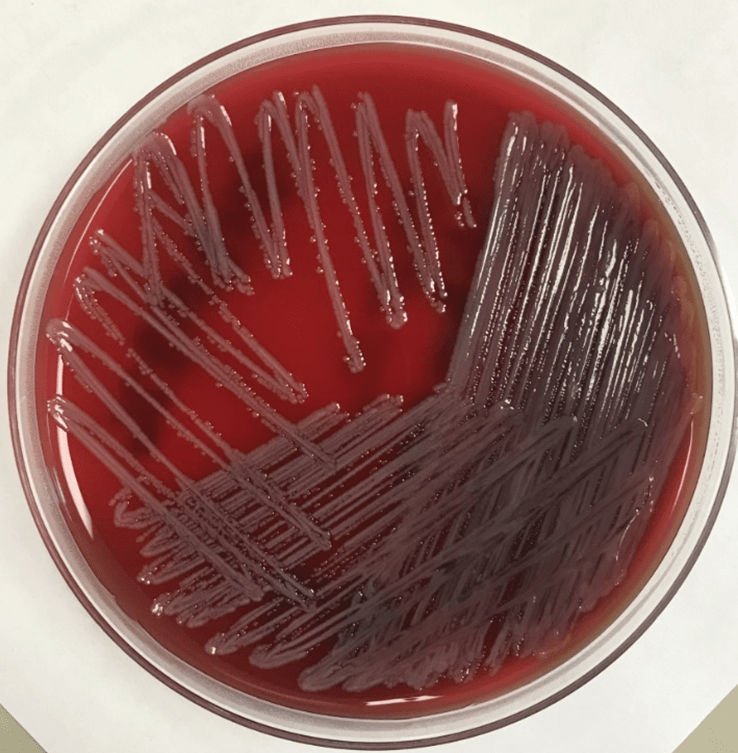
VIR drained the perisplenic abscess, which was sent to the microbiology lab for aerobic & anaerobic cultures. The Gram stain revealed many white blood cells (WBC’s) and rare yeast. The culture grew 3+ Burkholderia cepacia complex (Burkholderia cenocepacia) and 3+ Candida glabrata. The Gram stain and colony of the subculture of B. cepacia on a blood agar plate are shown in Images 2 and 3. C. glabrata was also isolated from the urine culture. Susceptibility testing of B. cepacia showed that it was sensitive to both meropenem and trimethoprim-sulfamethoxazole. Vancomycin and zosyn were stopped and the patient was switched to IV trimethoprim sulfamethoxazole for B. cepacia and IV micafungin for C. glabrata.
Due to the recurrent perforation, the G tube could not be used; a jejunostomy (J) tube was placed instead. Feeds were successfully started with the J tube. Trimethoprim-sulfamethoxazole was also given via the J-tube. A follow-up endoscopy showed a normal esophagus, the known perforation in the gastric fundus, and erythematous duodenopathy at the level of the duodenal bulb, the remainder of the duodenum was normal. The patient’s clinical status improved and she was discharged home with the support of home health services.
Discussion
We present an uncommon presentation of Burkholderia cenocepacia, a member of B. cepacia complex, in perisplenic abscess fluid. B. cepacia complex consists of at least 17 closely related species. They are rod-shaped, aerobic, motile Gram-negative bacteria. B. cepacia complex has been well characterized as opportunistic pathogens, particularly in patients with cystic fibrosis and chronic granulomatous disease (CGD). However, they can also infect immunocompetent patients and have been reported to cause endocarditis (specifically in IV drug abusers), pneumonitis, UTIs, osteomyelitis, dermatitis, and other wound infections. In the United States, B. multivorans and B. cenocepacia together account for approximately 80% of B. cepacia complex infections. Burkholderia have been isolated on contaminated hospital equipment and even disinfectants. They present a large problem in nosocomial infections due largely to their ability to survive in aqueous environments.1 They are soil-dwelling bacteria commonly found on plant roots. They are of environmental interest secondary to their antifungal and antinematodal properties as well as the ability to degrade many toxic compounds in agriculture (pesticides, herbicides, preservatives).2 Clinically important Burkholderia species outside of the B. cepacia complex include B. pseudomallei, the causative agent of melioidosis, and B. mallei, the causative agent of glanders.1
Rare case reports have previously documented B. cepacia isolated from splenic abscesses/infections. Most B. cepacia splenic infections occurred secondary to pneumonia or multi-organ involvements in CGD patients. 3, 4, 5 However, one report indicated the B. cepacia-mediated multiple splenic abscesses, in the setting of malignancy and diabetes. 6 While the splenic abscesses in the context of meliodosis, either due to B. pesudomallei or B. mallei infection, have been reported, 7 B. cepacia-mediated splenic infections are rarely encountered.
B. cepcacia complex has intrinsic resistance toseveral antibiotics including penicillins, amoxicillin-clavulanate, ertapenem, polymixin B, Colistin, and fosfomycin.8 B. cepacia complex possesses an inducible β-lactamase, encoded by the gene penA, which can hydrolyze penicillin and use it as a source of carbon. In one study involving 40 bloodstream isolates of B. cepacia in patients without cystic fibrosis, 93% of the isolates were susceptible to ceftazadime and 95% of isolates were susceptible to trimethoprim-sulfamethoxazole.9 Following discussion with our infectious disease colleagues, we believe that the B. cepacia isolate from our patient was likely a nosocomial infection from possible contamination of her G-tube in combination with the gastric perforation.
References
- Jorgensen, J. H., Pfaller, M. A., & Carroll, K. C. (2015). Manual of clinical microbiology. Washington, DC, DC: ASM Press.
- Kenyon College Department of Biology. (2011, April 22). Burkholderia cepacia. Retrieved September 21, 2020, from https://microbewiki.kenyon.edu/index.php/MicrobeWiki
- Clegg HW, Ephros M, Newburger PE. Pseudomonas cepacia pneumonia in chronic granulomatous disease. Pediatr Infect Dis. 1986 Jan-Feb;5(1):111. PMID: 3945563.
- Sirinavin, Sayomporn MD*; Techasaensiri, Chonnamet MD*; Pakakasama, Samart MD*; Vorachit, Malai DSc†; Pornkul, Rattanaporn MD‡; Wacharasin, Rames MD‡ Hemophagocytic Syndrome and Burkholderia cepacia Splenic Microabscesses in a Child With Chronic Granulomatous Disease, The Pediatric Infectious Disease Journal: September 2004 – Volume 23 – Issue 9 – p 882-884 doi: 10.1097/01.inf.0000137565.23501.03
- Bottone EJ, Douglas SD, Rausen AR, Keusch GT. Association of Pseudomonas cepacia with chronic granulomatous disease. J Clin Microbiol. 1975 May;1(5):425-8. doi: 10.1128/JCM.1.5.425-428.1975. PMID: 1176612; PMCID: PMC275137.
- Jayawardena, M. N., Chandrasiri, N. S., Wijekoon, S., Madanayake, P., Corea, E., Ranasinghe, D. D., & Lamahewage, N. D. (2017). Burkholderia cepacia; an unusual cause of multiple splenic abscesses : A case report. Sri Lankan Journal of Infectious Diseases, 7(2), 123. doi:10.4038/sljid.v7i2.8146
- Chen, H., Hu, Z., Fang, Y., Lu, X., Li, L., Li, Y, Mao, X, Qian, L. (2018). Splenic abscess caused by Burkholderia pseudomallei. Medicine, 97(26). doi:10.1097/md.0000000000011208
- Patel, J. B., Weinstein, M. P., Eliopoulos, G.M., Jenkins, S.G., Lewis, J.S., Limbago, B., Mathers, A., Mazzulli, T., Patel, R., Richter, S.S., Satlin, M., Swenson, J.M., Traczewski, M.M., Turnidge, J.D. & Zimmer, B.L. (2017). Performance standards for antimicrobial susceptibility testing. Wayne, PA: Clinical and Laboratory Standards Institute.
- Bressler A.M., Kaye K.S., LiPuma, J.J., Alexander, B.D., Moore, C.M., Reller, L.B. & Woods, C.W. Risk factors for Burkholderia cepacia complex bacteremia among intensive care unit patients without cystic fibrosis: A case-control study. Infect Control Hosp Epidemiol 2007; 28(8):951-8 doi : http://dx.doi.org/10.1086/519177

-J. Stephen Stalls, MD is a PGY-II pathology resident at the East Carolina University Department of Pathology and Laboratory Medicine. He plans to pursue hematopathology and molecular pathology fellowships, but also greatly enjoys his time in the microbiology lab. Outside of work, he enjoys playing the drums and going to concerts.

-Phyu Thwe, PhD, MLS(ASCP)CM is Technical Consultant/Technical Director of Clinical Microbiology Laboratory at Vidant Medical Center, Greenville, NC.
