Clinical Case
A 46 year old male with history of anal HPV with AIN II and anal fissure status-post sphincterectomy and fissurectomy, tobacco and cannabis use, and recent shingles outbreak presented with 1 year of diarrhea, fevers, chills and weight loss and a 2 week history of congestion and productive cough. He was found to have diffuse ground glass opacities with large cysts of the lungs on CT scan, and after admission was found to be positive for HIV with concern for AIDS. He received a bronchoscopy on that showed Pneumocystis jiroveci pneumonia. He was treated and clinically improved over several days when HAART therapy was initiated. Shortly afterwards he became neutropenic with his ANC as low as 150. The initial BAL fluid and stool became positive for acid fast bacilli.
Laboratory Identification
On Löwenstein-Jensen media, the organisms show small, flat, translucent, smooth colonies. They are slow-growers readily detected by acid fast and Kinyoun staining. In broth, the organisms do not show clustering or “cording.”
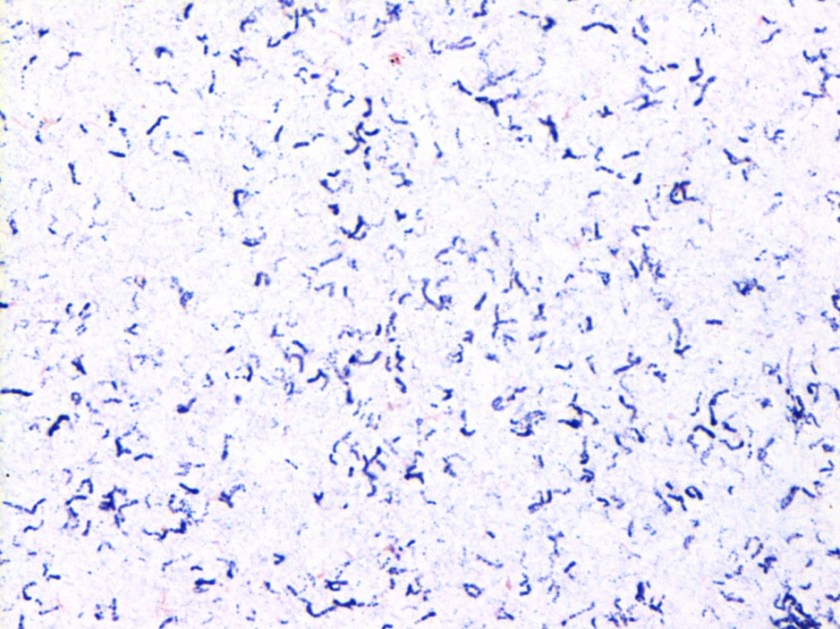
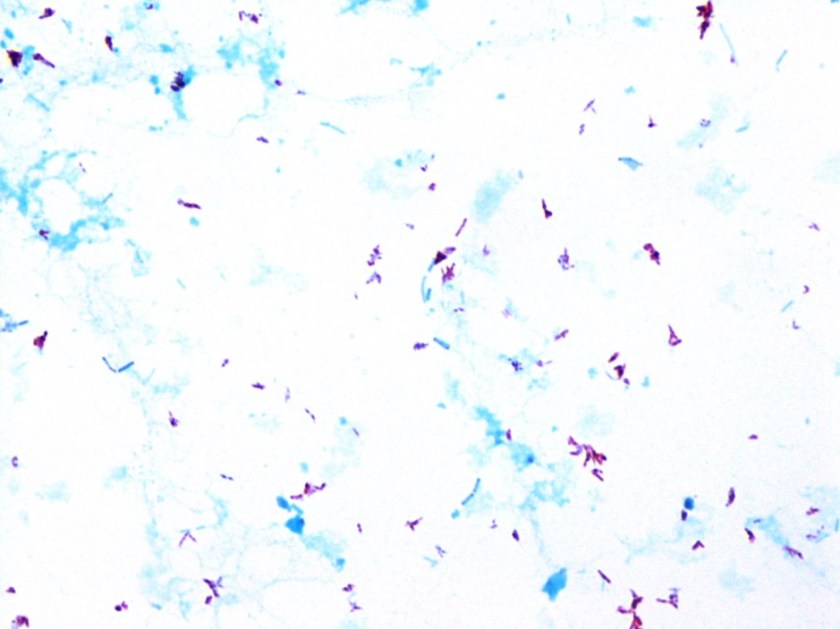
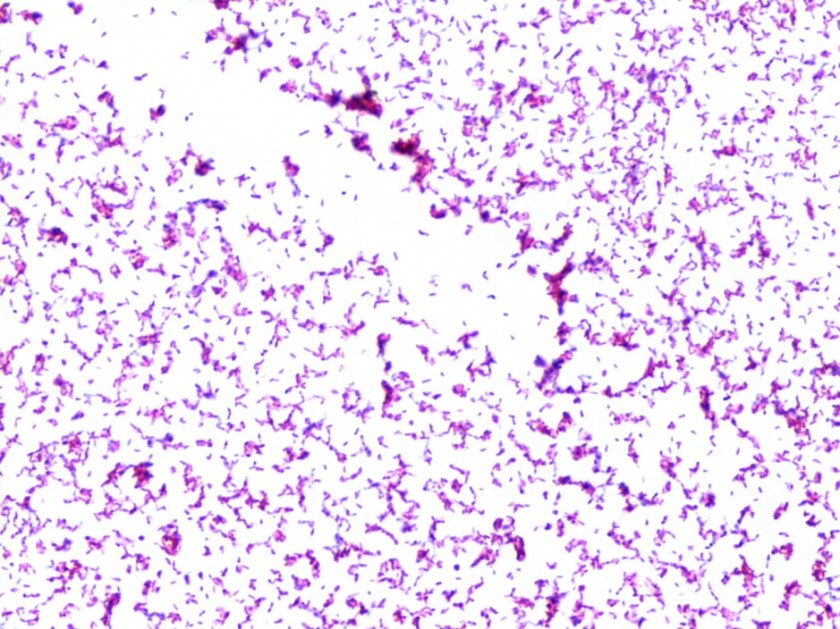

Discussion
Mycobacterium avium complex (MAC) is the most common nontuberculous mycobacterium (NTM) species causing human disease in the United States and is ubiquitous in the environment. MAC refers to infection caused by one of two slowly-growing NTM species, M. avium and M. intracellulare.
The pathogenesis of MAC lung disease is poorly understood. Infection is most likely acquired via ingestion or inhalation of aerosols from the environment inoculating a mucosal surface. Soon after inhalation or ingestion of MAC organisms, the infection disseminates lymphohematogenously. The bacteria are taken up by mononuclear phagocytic cells throughout the body, seeding other organs and tissues. Unlike M. tuberculosis, there is no convincing evidence demonstrating human-to-human transmission of MAC.
Disseminated NTM disease occurred in 5.5% of AIDS cases reported to the Centers for Disease Control and Prevention (CDC) from 1981 to 1987. This dropped to 4% after 1996, and is now at a rate of less than 1% per year. The dramatic decline in the disseminated disease is attributed to the use of effective prophylaxis with clarithromycin and azithromycin, as well as the advent of potent antiretroviral therapy. However, as in our case, patients with low CD4 cell counts remain at risk. Blood cultures for MAC isolation should be obtained before prophylaxis is initiated if there is any suspicion of clinical disease; the treatment regimen is different if blood cultures are positive (ie, the patient has active disease).
Traditional methods of speciating mycobacterial isolates were based upon growth characteristics on solid media and subsequent biochemical tests, requiring additional weeks for subcultures. Now we have commercially available highly-accurate nucleic acid probes that can identify MAC isolates within one day of growth. Other techniques such as 16S ribosomal DNA sequencing, Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry, PCR-restriction length polymorphism analysis (PRA), and high-performance liquid chromatography (HPLC) are also available.
Susceptibility testing of MAC is difficult and controversial compared with M. tuberculosis. Exceptions to this are macrolides and amikacin, for which the MICs have been shown to correlate clinically with in vivo response. Additionally, clarithromycin resistance can be detected by a mutation in the 23S ribosomal macrolide binding site.
-Thomas Rogers, DO is a 3rd year anatomic and clinical pathology resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Assistant Professor at the University of Vermont.

Great presentation, thank you Thomas and Christi