Case Description
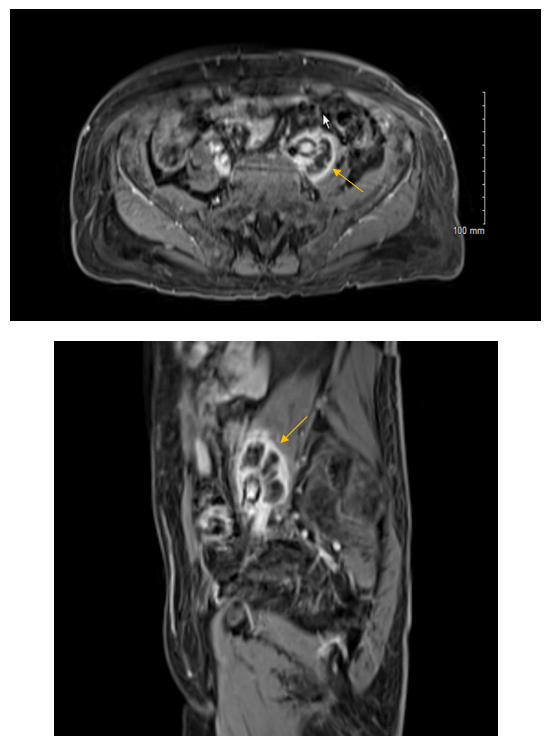
A 70 year old male with a past medical history of hypertension and non-invasive multifocal high grade papillary urothelial carcinoma was being followed closely for recurrence and underwent magnetic resonance imaging (MRI) of the abdomen and pelvis. The report described a 2.6 x 3.9 x 5.2 cm lobulated cystic mass involving the left psoas muscle. Furthermore, there was encasement of the left common iliac artery and less involvement of the left common iliac vein (Image 1). Further evaluation of the lesion was pursued to determine the etiology. An important aspect of this case to consider is the patient’s prior cancer treatment regimen, which included intravesical Bacille Calmette-Guerin (BCG) for 5/6 cycles. The final BCG treatment was held because the patient developed “BCG-osis” comprised of chest pain, rigors, chills and hypotension. Given the only pathology to date on the patient was non-invasive papillary carcinoma (even though it is high grade), the oncology group did not think the psoas muscle lesion was a metastasis. Fine needle aspiration (FNA) was pursued and the CT-guided aspiration demonstrated “abundant histiocytes and acute inflammation with necrotic debris…Acid fast organisms identified on AFB (acid fast bacilli) stain…Negative for malignant cells” (Image 2). Microbiology cultures were obtained at the time of FNA. The AFB smear showed 1+ AFB. AFB grew in culture and reacted with the Mycobacterium tuberculosis complex probe. The patient’s interferon gamma response assay (IGRA) was negative the year prior. Antimicrobial susceptibilities (AST) using the Mycobacterial Growth Indicator Tube system with single drug concentrations revealed susceptibility to isoniazid, rifampin, and ethambutol with resistance to pyrazinamide. Per protocol, the isolate was sent to a reference laboratory for identification which returned as BCG. During the interim, before AST was available, the patient was referred to our Infectious Diseases outpatient clinic where he was started on R(ifampin)-I(soniazid)-P(yrazinamide)-E(thambutol) therapy and then followed up his care with the county health department.
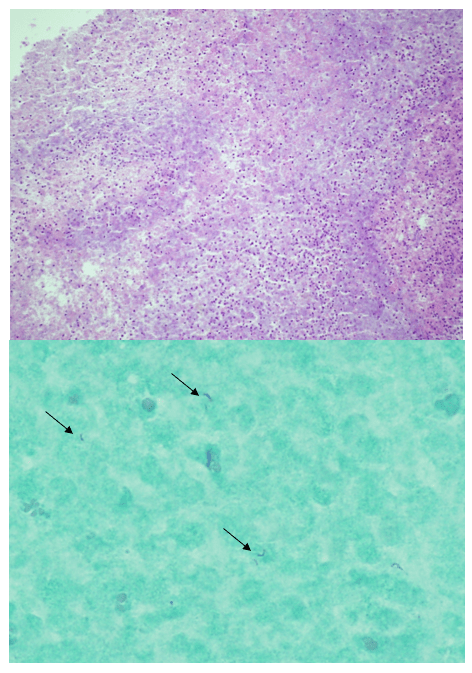
Top) hematoxylin and eosin photomicrograph demonstrating abundant neutrophils and necrotic debris (10x objective).
Bottom) AFB stain demonstrating multiple AFB (see arrows) within the necrotic cellular debris (50x oil immersion objective).
Discussion
BCG is an attenuated strain of Mycobacterium bovis that has, historically, been used as a vaccine to Mycobacterium tuberculosis (MTB) most often used in areas of endemicity. M. bovis is a member of the MTB complex so hybridization probes used in clinical laboratories can distinguish MTB complex from Mycobacterium kansasii or Mycobacterium avium complex but cannot distinguish the MTB complex members from one another. Reference laboratories have molecular techniques including PCR or sequencing that can separate the differing members of the MTB complex. Another traditional distinguishing characteristic between M. bovis (including BCG) and MTB is susceptibility to pyrazinamide. MTB carries a pyrazinamidase which is required to activate the antibiotic; M. bovis does not, so it is intrinsically resistant to pyrazinamide. A laboratorian or clinician should be cognizant of this when a culture result returns as MTB complex that is susceptible to rifampin, ethambutol and isoniazid but is resistant to pyrazinamide alone. Furthermore, IGRAs were designed, in part, to distinguish those who have been vaccinated with BCG versus those exposed to MTB or wild type M. bovis who are latently infected. IGRAs will be negative in those exposed to BCG but would be reactive with either MTB exposure or M. bovis (non-BCG strains).
This case describes an uncommon complication of BCG immunomodulator therapy as a treatment of superficial papillary urothelial carcinoma. BCG’s use as a therapeutic intervention for malignancy is unique. It is postulated that the instillation of the organism stimulates the host’s immune response which can cause inflammation and sloughing of the bladder lining (urothelial cells), which effectively removes foci of superficial pre-cancerous in situ lesions or other intact foci of superficial urothelial carcinoma. The typical course of treatment is 6 cycles of BCG. Local inflammation resulting in cystitis is the most common complication experienced in 27-95% of patients.1 However, approximately 19% of patients undergoing BCG therapy experience severe enough complications to prematurely terminate BCG therapy, according to one study by the European Organization for Research and Treatment of Cancer.2 The patient described developed systemic symptoms during the course of his BCG therapy which prompted his physicians to terminate it. Although far less common than local genitourinary symptoms, mycotic aneurysms can occur in an estimated 0.7-1.4% of cases.1 Psoas abscesses are thought to arise from mycotic aneurysm leak.3
References
- Liu Y, Lu J, Huang Y, Ma L. Clinical spectrum of complications induced by intravesical immunotherapy of bacillus Calmette-Guerin for bladder cancer. Journal of Oncology. 2019. DOI: 10.1155/2019/6230409
- Sylvester, R, Brausi M, Kirkels W, et al. Long-term efficacy results of EORTC genito-urinary group randomized phase 3 study 30911 comparing intravesical instillations of epirubicin, bacillus Calmette-Guerin, and bacillus Calmette-Guerin plus isoniazid in patients with intermediate- and high-risk stage Ta T1 urothelial carcinoma of the bladder. European Urology. 2010. 57:5; 766-773.
- Leo E, Molinari A, Rossi G, et al. Mycotic abdominal aneurysms due to Mycobacterium bovis after intravesical bacillus Calmette-Guerin therapy. Annals of Vascular Surgery. 2015. 29:6;1381.e1-1318.e6.
-Dominick Cavuoti is a Professor at UT Southwestern Medical Center who specializes in Cytopathology, Infectious Disease pathology and is a medical director of the Microbiology laboratory at Parkland Health and Hospital System.
-Kelley Carrick is a Professor at UT Southwestern Medical Center who specializes in Cytopathology and Gynecologic pathology. She is the Chief of Cytopathology at Parkland Health and Hospital System.

-Clare McCormick-Baw, MD, PhD is an Assistant Professor of Clinical Microbiology at UT Southwestern in Dallas, Texas. She has a passion for teaching about laboratory medicine in general and the best uses of the microbiology lab in particular.
