There are well over a hundred different cells types in the human body, and all those that have the ability to proliferate physiologically have the potential to succumb to uncontrolled cell division and thus generate a neoplasm. The tumors we most frequently encounter, like epithelial or hematologic disorders, are due to the higher proliferative rate of those cell types and the increased likelihood of an aberration or something going awry, i.e. mutations.2 Though we do come across many sarcomas and other mesenchymal tumors, this case study features a tumor derived from a cell type and structure that is rarely featured in cytology texts (an assumption, as I couldn’t find any information in the available prints within my department).
A 54 year old male patient presented to gastroenterology for severe cholecystitis. After undergoing a laparoscopic cholecystectomy, the pain subsided, but the patient was encouraged to follow up with a colonoscopy and upper endoscopy (EGD) due to his age. On the EGD, the gastroenterologist identified Barrett’s esophagus as well as a small, oval intramural lesion in the greater curvature of the gastric antrum, measuring 1.6 centimeters. The gastroenterologist was sharing the patient’s history with me when he described the endoscopic ultrasound findings of a well-defined hypoechoic and heterogenous lesion appearing to originate from the muscularis propria. His differential diagnoses based on imaging included a GIST, leiomyoma, or glomus tumor. The gastroenterologist did mention that the likelihood of this being a GIST or leiomyoma was very low. Despite lack of vascular structures visualized on Doppler imaging, the needle passes which I received to make air-dried Diff-Quik-stained smears were exceptionally bloody both grossly and microscopically.
I called my pathologist on cytology service for the day, informed him of the history presented to me by the gastroenterologist, and upon seeing the “rare atypical cells” through our telepathology platform, I hear him say, “a glomus tumor? Hmm, yes, it could be a glomus tumor.”
You know that feeling when you’ve asked someone to repeat themselves three times and you still didn’t comprehend them, so out of courtesy, you pretend to know what they said and express understanding? Yet internally you are confused beyond repair?
I repeated back to the gastroenterologist, “he said it could be a glomus tumor!”
Here comes the inner dialogue – what on earth is a glomus tumor? Why am I trying so hard to not look like an idiot right now? It’s okay to not know things, especially rare tumors that you’ve never come across. I have no idea what this tumor is, where it originates, what it consists of, this looks like a cohesive neuroendocrine tumor to me or even a basaloid squamous cell carcinoma. Obviously, it’s representative of the lesion, but I have no idea what a glomus (should I be pronouncing it with a long “o” or a short “o”?) is.
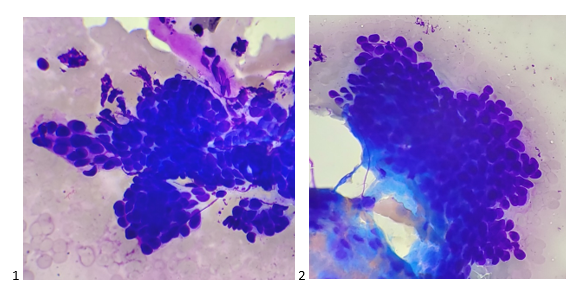
After screening the Pap-stained smears and H&E Cell Block sections the next morning, the cells still had cuboidal cells with features similar to a neuroendocrine tumor to me. The best I could call it was a neoplasm with neuroendocrine features before leaving the case with the pathologist on service to order immunostains.

IHC returned that afternoon, showing the neoplastic cells to be positive for SMA, focal weakly positive for synaptophysin, and negative for PanCK, CD34, desmin, chromogranin, CD45, DOG-1, HMB-45, and S-100 protein. Between the immunostains and the morphology findings, the case was signed out as a glomus tumor. Additional immunostains were performed showing the tumor cells are positive for vimentin and have a Ki-67 proliferation index of only 1%. When the tumor was resected two months after the initial FNA, pathology reported the findings as a 1.0 centimeter glomus tumor that was completely excised.
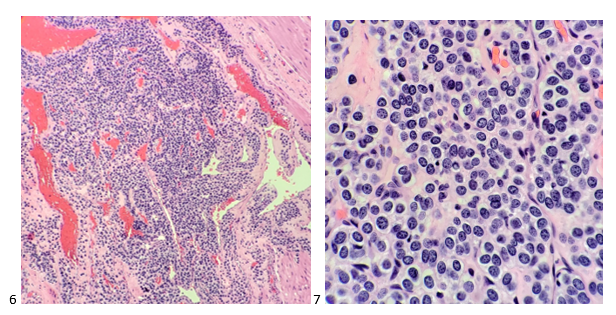
This tumor arises from the glomus body, which is a normal arteriovenous shunt that aids in regulation of temperature and blood flow in the body. Surrounded by smooth muscle tissue, the glomus body contracts and relaxes, closing and opening the shunt between the efferent venules and the afferent arteriole to pull blood flow away from the periphery and back into the body’s core or to allow heat dissipation. Glomus tumors are most often found in the dermis of the fingertips and toes, especially under nail beds due to the pain and cold sensitivity from exposure to cold.1,5 Glomus bodies are also found in the stomach as a thermoregulator (think cold food/liquids entering the digestive system), and account for 1% of mesenchymal gastric tumors.4 Most glomus tumors are benign and rarely undergo malignant transformation, and complete excision of these tumors typically provides immediate relief with little to no chance of recurrence.1,3
References
- Fazwi, R., Chandran, P. A., & Ahmad, T. S. (2011). Glomus Tumour: A Retrospective Review of 15 Years Experience in A Single Institution. Malays Orthop J., 5(3), 8-12. doi:10.5704/MOJ.1111.007
- Holly, J. M. P., Zeng, L., & Perks, C. M. (2013). Epithelial cancers in the post-genomic era: should we reconsider our lifestyle? Cancer and Metastasis Reviews, 32(3–4), 673–705. https://doi.org/10.1007/s10555-013-9445-5
- Nascimento, E. F. R., Fonte, F. P., Mendonça, R. L., Nonose, R., de Souza, C. A. F., & Martinez, C. A. R. (2011). Glomus Tumor of the Stomach: A Rare Cause of Upper Gastrointestinal Bleeding. Case Reports in Surgery, 2011, 1–5. https://doi.org/10.1155/2011/371082
- Papadelis, A., Brooks, C. J., & Albaran, R. G. (2016). Gastric glomus tumor. Journal of Surgical Case Reports, 2016(11), rjw183. https://doi.org/10.1093/jscr/rjw183
- Uddin, M. M., Biswas, S. K., Rahman, M. H., Karmakar, N. C., Rahman, M. M., Alam, S. A. U., & Mondal, A. R. (2017). Sub-ungual Glomus Tumor: Study of 20 Cases. Faridpur Medical College Journal, 12(2), 64–67. https://doi.org/10.3329/fmcj.v12i2.34230

-Taryn Waraksa, MS, SCT(ASCP)CM, CT(IAC), has worked as a cytotechnologist at Fox Chase Cancer Center, in Philadelphia, Pennsylvania, since earning her master’s degree from Thomas Jefferson University in 2014. She is an ASCP board-certified Specialist in Cytotechnology with an additional certification by the International Academy of Cytology (IAC). She is also a 2020 ASCP 40 Under Forty Honoree.
