Platelets are our first line of defense in controlling bleeding. Abnormally low numbers of platelets can lead to easy bruising, tiny leaks from capillaries into the skin and mucous membranes, causing petechiae, and bleeding. The platelet count is a significant parameter in the CBC and it is therefore vital to be able to report accurate and precise platelet counts. Furthermore, physicians must be able to use this information to diagnose the cause of the thrombocytopenia in order to recommend treatment.
What a platelet count alone cannot tell us is the reason for thrombocytopenia. Just as there can be many reasons for a low hemoglobin, and many causes for an increased or decreased WBC, there are numerous causes for a decreased platelet count. After ordering a CBC, the next steps in determining etiology of thrombocytopenia have historically been a thorough physical with attention to any bleeding symptoms and organ enlargements, and a medical history. The medical history should include family history, and notation of recent viruses or drug therapies. After these tests, a bone marrow aspirate and biopsy may also be necessary to clarify etiology. While modern, automated hematology analyzers produce reliable platelet counts, measuring only the circulating platelet count does not give us any information as to the etiology, so there is a need for further testing. With thrombocytopenia, platelet counts can be less reliable than with normal counts.Platelet counts were originally performed by impedance methods, then better accuracy and precision was obtained with optical platelet counts. Physicians rely on precision with very low platelet counts to make informed decisions about when to transfuse patients. The problem with the impedance counts at the low end, is that RBC fragments, schistocytes and microcytic RBCs can be counted as platelets, giving a falsely high count. On the other hand, measuring platelets by size can miss large platelets leading to a falsely low count.
Historically, the mean platelet volume (MPV) has been used along with the platelet count to aid in making a differential diagnosis. The MPV is analogous to the red cell distribution width (RDW) for red cells, and can be used to as an indicator of the maturity of platelets. Young platelets are the largest, and as they age, the size decreases. The normal ranges for MPV are generally about 9-12 femtoliters (fl).The MPV will be higher if more platelets are being released from the bone marrow, and lower if fewer are being newly released and we are counting mature platelets. Thus, the MPV can be used as an indirect marker for platelet production. However, an inherent problem with the MPV is that, similarly to the impedance platelet count, this count can be unreliable because any RBC fragments or particles may interfere with the measurement.
So, what is a physician to do?And how can the lab provide information to help them make the best differential diagnosis and transfusion decisions? In an effort to provide a parameter that could help differentiate causes of thrombocytopenia, the concept of reticulated platelet counts (retPLT) was first introduced in research in the late 1960s. The term is used to describe immature, functional platelets in the peripheral blood.Reticulated platelets are to mature platelets as reticulocytes are to mature red blood cells. These are the youngest platelets, within 24 hours of being released from the bone marrow. Reticulated platelets are large, with increased amounts of RNA, and the number in the circulation can be used to provide an estimate of the rate of thrombopoiesis. Originally, these were stained with new methylene blue and manual counts were done, much like a manual reticulocyte count; tedious,and imprecise. It wasn’t until about 30 years later that a flow cytometry method was described for measuring retPLT. Using traditional flow cytometry, reticulated platelets can be stained with a Thiazole Orange dye and passed through a flow cytometer. This method, however, has been shown to have wide normal ranges from 1-15% because of the lack of analytical standardization. Variations in the concentration of the thiazole dye used, the timing, and the gate settings all make it difficult to compare results obtained from one laboratory to another. In addition, flow cytometry is time consuming, labor intensive and costly.
Newer flow cytometry methods are now available on select hematology analyzers. There are currently 2 companies that have analyzers that can report retPLT using routine CBC reagents and controls. Reticulated platelets can be measured with the same K2 EDTA tube used for the CBC. The test is automated, simple to perform, fast, and gives standardized results with tighter normal ranges. The Abbott CELL-DYN Sapphire measures the retPLT using a fluorescent dye and flow cytometry with 2 dimensional gating. Sysmex XE and XN analyzers offer several Advanced Clinical Parameters including measures of reticulated platelets, expressedas the Immature Platelet Fraction (IPF%) and the Absolute Immature Platelet Fraction Count (IPF#). Sysmex offers a fluorescent platelet count (PLT-F) as an addition to impedance counting (PLT-I) and optical counting (PLT-O). PLT-F is more reliable because it uses a platelet specific dye which eliminates noninterference seen with other methods. The fluorescent dye labels the RNA and forward scatter is used to determine size while fluorescence is used to measure RNA content. With gating set based on cell volume and RNA content, the PLT-Fcan be measured. When there is an abnormal scattergram or a low platelet count,the PLT-F is reflexed and the IPF% and IPF# are also reported.
What’s the clinical utility of the IPF? Thrombocytopenia can have many causes.Immature platelets are functioning platelets, and an increased IPF means that we have more newly formed immature platelets circulating. The IPF helps physicians to differentiate thrombocytopenia caused by platelet destruction or consumption versus thrombocytopenia caused by deficient platelet production in bone marrow failure. It is vital to know the pathogenesis of thrombocytopenia in making decisions about treatment. With these advanced parameters, these decisions can often be made without costly, time consuming flow cytometry,without an invasive bone marrow biopsy and without waiting for the results of such biopsy. This can often save a patient an unnecessary platelet transfusion.
The reference range for IPF% in healthy individuals is1.0-7.0%. Together with a low platelet count, an increased IPF indicates an increase in platelet production. This is seen in patients with excessive destruction of platelets. An example of the clinical utility of the IPF can be seen in the diagnosis of immune thrombocytopenic purpura (ITP). ITP is an autoimmune bleeding disorder in which the immune system makes anti-platelet antibodies which destroy platelets. Acute forms occur more often in children while adults can have chronic ITP. ITP can be diagnosed on clinical findings but laboratory confirmation is often necessary. This can be expensive with long turnaround times using traditional flow cytometry and/or bone marrow aspirates.An IPF reported with a CBC is fast, inexpensive, and be extremely beneficial in aiding a timely diagnosis. Patients with ITP have been shown to have the consistently highest IPF values with ranges from 7-28%.1 As their platelet counts recover, the IPF% returns to the normal range, without the need for transfusions. Thus, the IPF can be used not only to help diagnose but also as an indicator of remission.
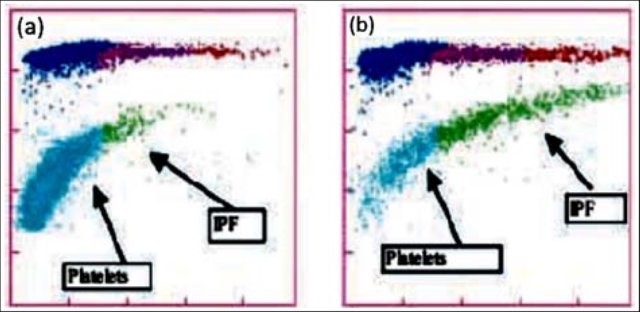
In contrast to what we see with ITP, thrombocytopenia with alow normal or decreased IPF indicates decreased bone marrow production of platelets. Patients with bone marrow failure are more likely to have bleeding episodes with low platelet counts and may need transfusion. Rapid differential diagnosis using the IPF can help physicians help these patients get early treatment.
IPF may also be a reliable indicator of bone marrow recovery. Traditionally, neutrophil counts have been used as an indicator of recovery after a bone marrow transplant. IPF can be used as an indicator of imminent platelet recovery. It has been shown that,post-transplant, the IPF% increases before the platelet count. In a study done with stem cell transplant patients, it was shown that the absolute neutrophil count took an average of 13 days to recover, compared to 9 days for the IPF. The IPF was shown to recover before the Immature reticulocyte count, platelet count and absolute neutrophil count, giving physicians earlier indication that the transplant was successful.2 This is significant because it can eliminate the need for bone marrow biopsies and platelet transfusions.
Thrombocytopenia is not an uncommon finding in neonates, particularly in the neonatal intensive care unit (NICU). There are various causes for this, including sepsis, placental insufficiency and immune thrombocytopenia. The IPF% and IPF# can be used to diagnose and distinguish the cause of thrombocytopenia in neonates, and direct the treatment. When platelet count platelet count drops below 50 x 103/Lin an otherwise healthy appearing infant in the first 72 hours of life, neonatal alloimmune thrombocytopenia (NAIT) can be suspected. This condition is similar in pathogenesis to hemolytic disease of the fetus and newborn (HDFN), and is caused by an incompatibility in human platelet antigens between mother and baby. This occurs most often when the mother is HPA-1b and the father and baby are HPA-1a. The mother forms anti-HPA-1a which crosses the placenta and destroys the fetus’ platelets.This is a thrombocytopenia caused by platelet destruction, and the IPF% is high. The condition is self-limiting and resolves in 1-4 weeks. Neonatal sepsis can also present with a high IPF, but typically is found in very sick or premature babies and the degree of thrombocytopenia is not as severe as with NAIT. In contrast, neonatal thrombocytopenia due to placental insufficiency would exhibit a decreased IPF due to a deficiency in platelet production. Using the IPF% and IPF# to help differentiate the causes of neonatal thrombocytopenia can help steer the treatment and save infants from unnecessary invasive procedures and transfusions.
TheIPF has proved to be very valuable in the clinical setting. It has been used in the investigation of etiology in secondary thrombocytopenias due to chronichepatitis C, liver disease and HIV. It has been used to guide treatment in thrombocytopenias such as thrombotic thrombocytopenic purpura (TTP). IPF can also be useful in evaluation of hereditary platelet thrombocytopenias. The IPF% and IPF# can be compared after transfusion to support the theory that, after platelet transfusion, theIPF% will decrease due to the newly increased platelet count, but the IPF#remains the same. This validates that the IPF is a reflection of continual platelet production by the bone marrow.4
IPF%and IPF# are expanded CBC parameters that physicians can use to aid in differentiation of various thrombocytopenic states. Treatment for the different classes of thrombocytopenia can differ drastically, and knowing the class of thrombocytopenia helps direct the management. The IPF parameters are automated,easy to perform at the same time as the CBC, and provide standardized results that are inexpensive and available 24 hours a day in the hospital setting. Using the IPF can also reduce diagnostic costs for the patient. Many studies have been conducted on the varied applications of the IPF and research continues investigating possible further uses of this advanced clinical parameter. This is the new hematology, constantly providing the clinician with better tools for making diagnoses and treating patients. Platelet counts alone and MPVs are out. Make room for the new kid on the block; the IPF is in.
References
- Arshi Naz et al. Importance of Immatureplatelet Fraction as a predictor of immune thrombocytopenic purpura. Pak J MedSci 2016 Vol 32 No 3:575-579
- Zucker ML et al. Immature Platelet fraction asa predictor of platelet recovery following hematopoietic progenitor celltransplanatation. Lab Hematol 2006 12(3):125-30
- Briggs,C. Assessment of an immature plateletfraction (IPF) in peripheral thrombocytopenia. Br J Haematol 2004Jul;126(1):93-9
- Sysmex White Paper. The role of the ImmaturePlatelet Fraction(IPF) in the differential diagnosis of thrombocytopenia. www.sysmex.com/us
- Fujii,T et al.. A new approach to detectreticulated platelets stained with thiazole orange in thrombocytopenicpatients. Thromb Res. 2000 Mar 15;97(6):431-40
- Cremer Malte The immature platelet fraction(IPF) in neonates. Diagnostic Perspectives 2011 Vol1:36-42
- Cremer M. et al. Immature platelet values indicateimpaired megakaryopoietic activity in neonatal early-onset thrombocytopenia.Thrombosis and Haemostasis 2010; May;103(5):1016-21

-Becky Socha, MS, MLS(ASCP)CM BB CM graduated from Merrimack College in N. Andover, Massachusetts with a BS in Medical Technology and completed her MS in Clinical Laboratory Sciences at the University of Massachusetts, Lowell. She has worked as a Medical Technologist for over 30 years. She’s worked in all areas of the clinical laboratory, but has a special interest in Hematology and Blood Banking. When she’s not busy being a mad scientist, she can be found outside riding her bicycle.
