Hello again – let’s continue our discussion of Next Generation, or Massively Parallel, Sequencing and how it is performed. Over the last two blogs we have seen why NGS is being used in a Molecular Diagnostics Lab and how library preparation is executed. Specifically, we reviewed how Ion Torrent and MiSeq libraries may be prepared for DNA amplicon sequencing. The final product of this work is a collection of amplicons that have been amplified, barcoded, tagged with the appropriate platform adapters and purified. These are what compose a specimen’s “library.”
The next step in NGS preparation is template preparation. The main goal of this step is to create multiple copies of the same amplicon in close proximity so that when it is sequenced, it creates a strong enough signal to be detected. This occurs for each amplicon in the specimen’s library. Again, this technique is platform specific, so each has a different way to achieve this goal.
Ion Torrent “Template Preparation by Emulsion PCR” or “Clonal Amplification”
In the Ion Torrent method of template preparation, the multiple copies are created on an Ion Sphere Particle or ISP. This looks like a bead with primers all over the surface of it. Eventually this ISP will be deposited in a well on a chip and be sequenced. In order for this ISP to create enough of a signal to be detected by the instrument, it must have many copies of the fragment all over the surface of the ISP.
At the beginning of the clonal amplification step, a specific concentration of combined libraries is added to the instrument, along with all the components of a standard PCR (buffer, dNTPs, polymerase) with the addition of the Ion Sphere Particles, which provide the primer, and oil. The primers on the ISP are complementary to one of the adapters added during library preparation so that only the universal primer is necessary on the ISPs, instead of each individual gene-specific primer. Through a series of steps, ideally, what is produced is a droplet of oil containing one ISP, one sample’s amplicon, and the components of the master mix. This, along with millions of other ISPs in droplets of oil, will undergo cycles of PCR, with the primers on the ISP priming the specimen’s amplicon. These amplicons will replicate all over the ISP, and as a final step, NaOH will be added to separate the strands. The strands that are not anchored to the ISP by the universal primer will be lost, leaving each ISP single stranded and ready for priming in the sequencing step.

One thing to consider is the concentration of the combined libraries that are added at the beginning of the template preparation. If the concentration is too low, obviously not enough amplicons will be amplified on the ISPs, and the end result will be not enough data. Conversely, if the concentration is too high, there is a possibility of more than one sample amplicon ending up in the droplet of oil. In the end, more than one fragment gets amplified on the ISP. This ISP is called “polyclonal” and the data from it will get thrown out. Optimizing the concentration takes a few runs and the concentration can be different for each instrument in the lab.
Illumina MiSeq “Cluster Generation by Bridge Amplification”
Illumina’s method of template preparation is termed cluster generation by bridge amplification and actually takes place on the MiSeq a step before the sequencing step. The multiple copies are created in close proximity to each other, just as with clonal amplification, but instead of using a separate ISP for each specimen, a separate location on the flow cell is used. A flow cell is essentially a glass slide that has universal primer anchored all over it. This universal primers are, again, complimentary to the adapters added during the library preparation. The combined libraries are flowed over the slide at the beginning of the run and they anneal to the universal primer. The fragment then folds over and anneals to the second universal primer. This strand is then replicated. After replication, the strands are denatured creating two single strands. These then replicate again, thus producing a cluster of the same fragment in a localized area on the slide. This occurs for each specimen’s amplicons all over the slide. At the end of the cluster generation step, the reverses are all cleaved off leaving only the single stranded forwards ready for sequencing.
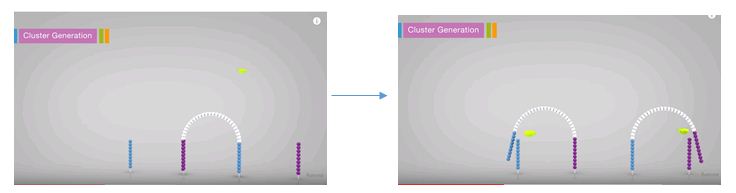

Concentration is just as important in this setup as in the Ion Torrent setup. If the concentration is too high with this assay, the clusters generated will be too close together on the flow cell, thus the sequencing signal from each cluster will overlap. The data generated from these areas will not be able to be discerned so it will get thrown out.
Join me next quarter for the next installment – sequencing!

-Sharleen Rapp, BS, MB (ASCP)CM is a Molecular Diagnostics Coordinator in the Molecular Diagnostics Laboratory at Nebraska Medicine.
