Case History
A 75-year-old male with a past medical history of hypertension, hyperlipidemia, and benign prostatic hyperplasia underwent an elective right hemicolectomy at an outside hospital after a cecal polypectomy demonstrated an intramucosal adenocarcinoma (in situ) arising in a background of a sessile serrated adenoma. On post-op day 6, he was transferred to our institution for management of an ST-elevation myocardial infarction that was treated with placement of a drug-eluting stent to the right coronary artery. After the cardiac catheterization, he complained of acute-onset abdominal pain and was tachypneic (49/min), hypotensive (72/48 mmHg), and febrile (39.4°C). He was emergently intubated, given vasopressors, and started on vancomycin and piperacillin/tazobactam empirically for septic shock. A chest X-ray showed atelectasis but no pulmonary consolidation. An abdominal X-ray did not show definitive evidence of pneumoperitoneum and abdominal CT showed some free fluid but no acute abdominal pathology. The WBC count was 3,640/cm3 with an absolute neutrophil count (2,880/cm3) within normal limits. The anaerobic bottle in one of two blood culture sets drawn on post-op day 7 became positive at 27 hours and Gram staining (Image 1) demonstrated gram negative bacilli. Subsequently, the bacilli detected in the anaerobic blood culture bottle were identified by MALDI-TOF as Clostridium clostridioforme, requiring a laboratory corrected report. On post-op day 8, two sets of repeat blood cultures were both positive with Clostridium tertium (Images 2 and 3) and Escherichia coli, consistent with bowel flora. Therapy for the patient’s polymicrobial bacteremia, thought to arise from an ileocolic anastomotic leak, was switched to piperacillin/tazobactam and Metronidazole. Blood cultures on post-op days 10 and 14 were negative. Meanwhile, the patient developed diarrhea, secondary to Clostridium difficile colitis, treated with oral vancomycin and oral thrush treated with micafungin. His hospital course was further complicated by formation of intra-abdominal abscesses, containing E. coli, C. tertium, and C. albicans, that required percutaneous drain placement.

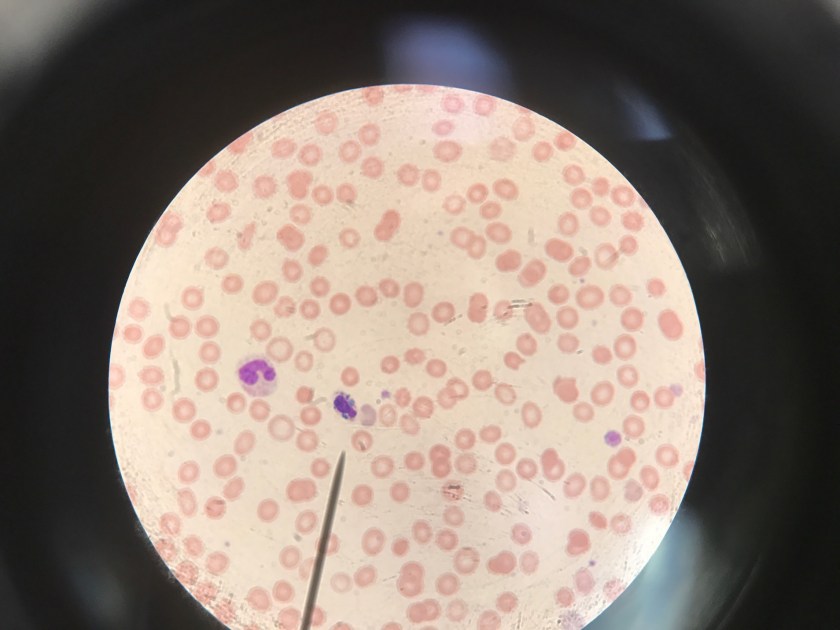
Image 1. Gram stain of Clostridium clostridioforme from a positive anaerobic blood culture bottle demonstrates thin gram negative bacilli with pointed ends arranged in pairs (100x, oil immersion).

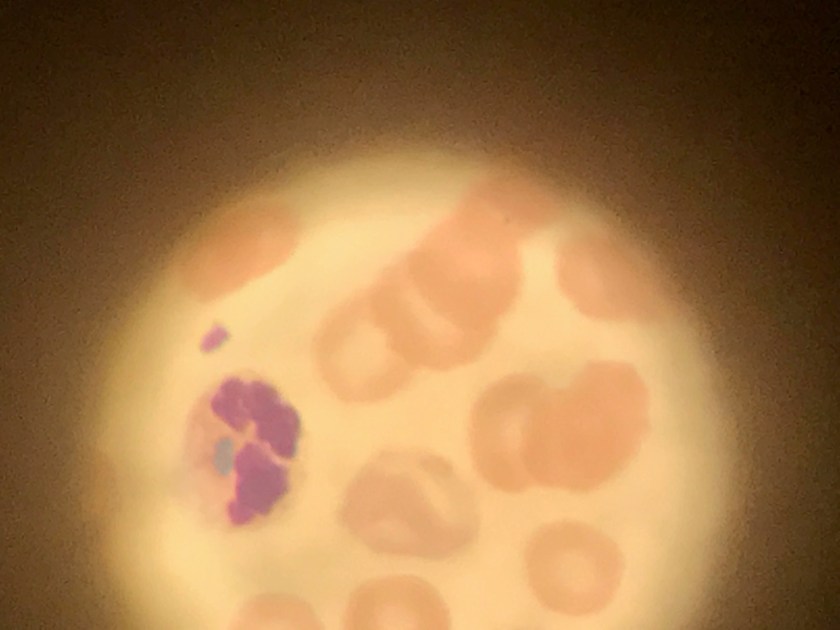
Image 2. Gram stain of Clostridium tertium from a positive anaerobic blood culture bottle demonstrates gram variable bacilli arranged in short chains (100x, oil immersion).
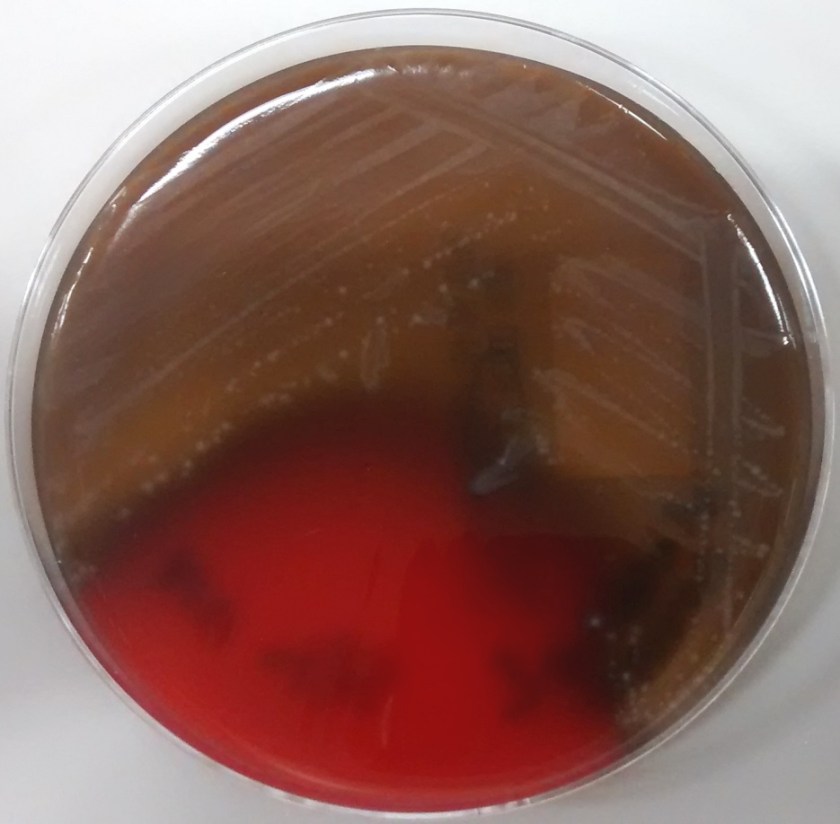
Image 3. Clostridium tertium colonies are β-hemolytic on an anaerobic (Schaedler) blood agar plate and appear circular with slightly irregular margins, matte, and grey-white.
Discussion
The genus Clostridium contains approximately 200 species, of which approximately 32 have been associated with human pathologies (1). These organisms are normal members of the human gastrointestinal and cervical-vaginal microflora. Clostridia are also ubiquitously present in nature within soil. Thus, human infection may occur via endogenous or exogenous means. They are classified as gram positive rods and, as such, they do not grow on media that inhibit the growth of gram positive organisms (ie. MacConkey agar). However, upon gram staining, Clostridia may appear gram positive, gram variable, or gram negative. Due to the gram stain variability, inconsistent presence of spores, and atypical colony morphologies, laboratory identification of Clostridum species is problematic.
Clostridium clostridioforme was initially detected in the anaerobic blood culture bottle at 27 hours. Gram staining (Image 1) demonstrates gram negative long, thin bacilli with pointed ends, described as “elongated football shaped” that are arranged in pairs but may also lie singly or in short chains. Oval spores may not be seen but they can be central or subterminal. As obligate anaerobes, C. clostridioforme may be cultured on anaerobic blood agar plates where the gamma-hemolytic colonies appear small, convex to slightly peaked, translucent to opaque, and grey-white. They possess peritrichous flagella that confer motility. It is believed that C. clostridioforme may represent three different species that are frequently isolated anaerobically from blood cultures, particularly in association with mixed cultures, typical of colonic flora (2).
Subsequent blood cultures one day later were positive for both Escherichia coli (detected at 18 hours) and Clostridium tertium (detected at 21 hours). The anaerobic blood culture bottle gram stain (Image 2) demonstrates C. tertium staining as gram variable bacilli arranged in short chains. Terminal spores, only produced under anaerobic conditions, are not seen in Figure 2. C. tertium is one of the aerotolerant clostridia and was cultured on an anaerobic blood agar plate (Figure 3). Colonies appear circular with slightly irregular margins, low convex, matte, and grey-white. Hemolysis can be beta, alpha, or gamma. It was likely overgrown by the E. coli on the aerobic plates. This species is generally considered a weak human pathogen but it has been implicated as a cause of bacteremia in immunocompromised patients. In non-neutropenic patients, C. tertium bacteremia can occur in the setting of gastrointestinal mucosal injury due to gastrointestinal tract pathology or surgery (3).
References
- Tille PM. Bailey & Scott’s Diagnostic Microbiology, 13th ed. Elsevier Health Sciences; 2014. pp458-479.
- Finegold SM, Song Y, Liu C, et al. Clostridium clostridioforme: a mixture of three clinically important species. Eur J Clin Microbiol Infect Dis. 2005;24(5):319-24.
- Miller DL, Brazer S, Murdoch D, Reller LB, Corey GR. Significance of Clostridium tertium bacteremia in neutropenic and nonneutropenic patients: review of 32 cases. Clin Infect Dis. 2001;32(6):975-8.
-Adina Bodolan, MD is a 1st year anatomic and clinical pathology resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Associate Professor at the University of Vermont.