A 60 year old patient with history of breast cancer presented with pancytopenia and bone marrow study was done to evaluate for metastatic tumor.
CBC revealed pancytopenia with a WBC count of 1.9 K/uL, macrocytic anemia (Hb 7.6 g/dl, MCV 106.2 fl) and decreased platelets at 15 K/uL.
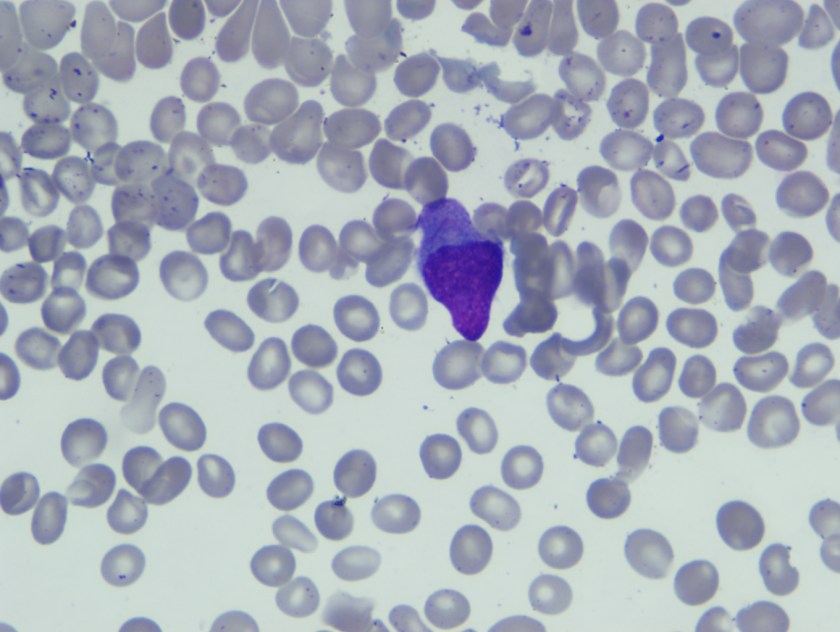
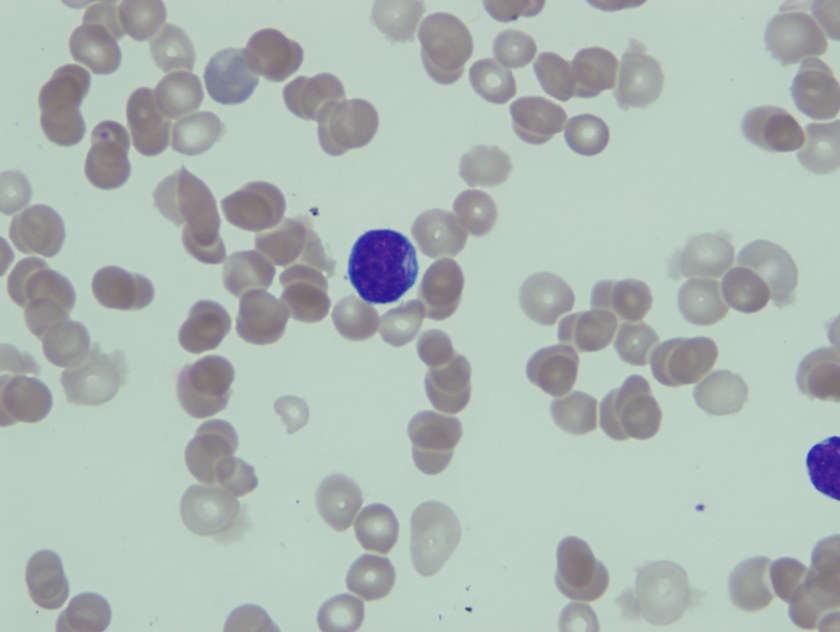
Review of peripheral smear revealed circulating blasts.
Differential Count (100 cells):
- 13% Blasts
- 18% Neutrophils
- 69% Lymphocytes
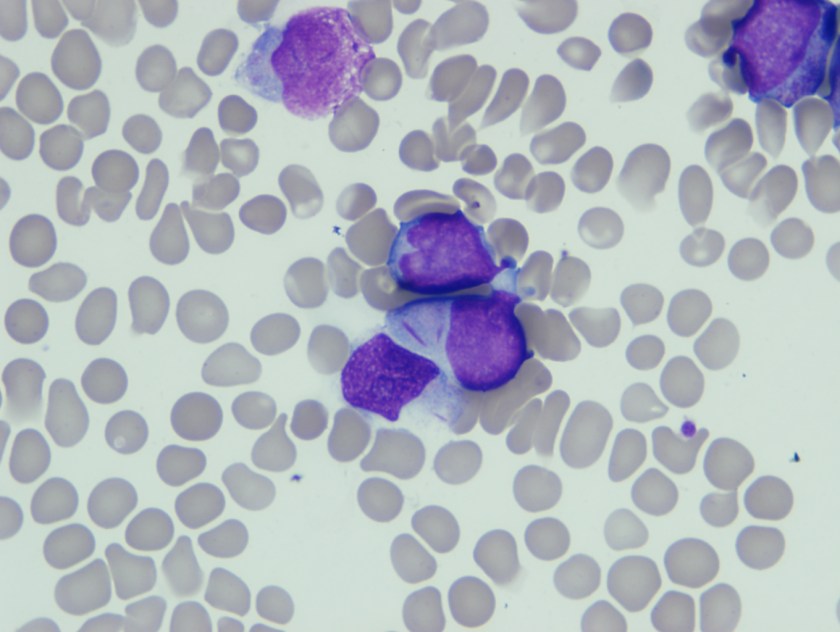
Blasts were intermediate in size with high N/C ratio, fine nuclear chromatin, 0-1 nucleoli, and scant basophilic cytoplasm. Few blasts have convoluted nuclei and in an occasional blasts slender Auer rod was seen.
Based on this morphological finding FISH for t8;21) was ordered along with karytoype analysis.
Translocation (8;21) was detected by both karyotype and FISH analysis (50.5% cells).
Cytogenetic results
Karyotype
46,X,?inv(X)(p22.1q22),der(8)t(8;21)(q21.3;q22),der(21)t(8;21)(q21.3;q22)add(8)(q24.3)[19]
46,XX[1]
FISH Results:
nuc ish (RUNX1T1,RUNX1x3)(RUNX1T1 con RUNX1x2)[101/200]
nuc ish (MYC,IgH)x2[97/200]
Based on presence of complex karyotype and t(8;21) together with prior history of chemotherapy for breast carcinoma diagnosis of Therapy related AML with t(8;21) was made.
Discussion
Acute myeloid leukemia (AML) associated with the t(8;21)(q22;q22) represents approximately 5% to 12% of de novo AMLs. At the molecular genetic level, this neoplasm is defined by the presence of the t(8;21)(q22;q22) involving the AML1 (RUNX1) gene on chromosome 21q22.3 and the ETO (RUNX1T1) gene on chromosome 8q22. TheAML1–ETO (RUNX1–RUNX1T1) fusion product disrupts the core binding factor transcription complex, which affects cell differentiation, proliferation, apoptosis, and self-renewal and, thus, initiates leukemogenesis. Patients with de novo AML-t(8;21) have a high complete remission rate and relatively long disease-free survival, especially adults treated with high-dose cytarabine in the consolidation phase.
Therapy-related AML (t-AML) is a known complication of cytotoxic chemotherapy and radiation therapy, which are known to be mutagenic. Causative agents include alkylating agents and topoisomerase-II inhibitors and platinum drugs. The outcomes for patients with t-AML have been historically poor compared with those for patients with de novo AML. t-AML-t(8;21) is uncommon.
Blasts in t-AML and de novo AML associated with t(8;21)(q22;q22) share characteristic morphologic and immunophenotypic features, and affected patients have a comparable initial response to induction chemotherapy. Patients with t-AML-t(8;21), however, have been reported to have a shorter survival.
Therapy related AML with t(8;21) shares morphologic and immunophenotypic features with de novo AML-t(8;21).
Common morphological features include the presence of large blasts with abundant cytoplasm, often containing Auer rods which appear long and slender with tapered ends as was seen in our patient. Additional common morphological findings seen in AML with t(8;21) include presence of numerous azurophilic granules and perinuclear clearing or very large granules (pseudo-Chediak-Higashi), suggesting abnormal fusion.
Both de novo AML with t(8;21) and therapy related AML with t(8;21) are characterized by the AML1–ETO (RUNX1–RUNX1T1) fusion. However, AML1–ETO fusion in t-AML does not predict a favorable outcome in the therapy-related setting. Patients with t-AML-t(8;21) usually have a higher frequency of KIT mutations, which is an adverse prognostic indicator.

-Neerja Vajpayee, MD, is an Associate Professor of Pathology at the SUNY Upstate Medical University, Syracuse, NY. She enjoys teaching hematology to residents, fellows and laboratory technologists.

very helpful