Case
A 26 year-old G1P0A0 female at 35 weeks 2 days twin gestation presented with contractions and was found to have oligohydramnios and decelerations of both twins. She reported a subjective fever two days prior and the morning of admission, associated with a mild frontal headache, both of which resolved prior to presentation. At the time of admission she was afebrile, however pancytopenic with WBC 4.17 x 109/L, platelets 105 x 109/L of and hemoglobin 7 g/dL with laboratory evidence of hemolysis; haptoglobin <5 mg/dL, reticulocyte 143 x 109/L (6.5%), lactate dehydrogenase 460 units/L and total bilirubin 2.4 mg/dL. Upon further questioning, the patient reported that she recently emigrated from Central Africa where she was found to be anemic and treated for malaria approximately one month prior to presentation. She reported taking oral medication with no follow-up. A malaria workup, including thick and thin smears (Figure 1), was initiated for the patient and her newborn twins after a successful urgent Caesarian section. The placenta was submitted for standard histopathology (Figure 2) according to protocol due to pre-term birth, multiple gestation and fetal distress.

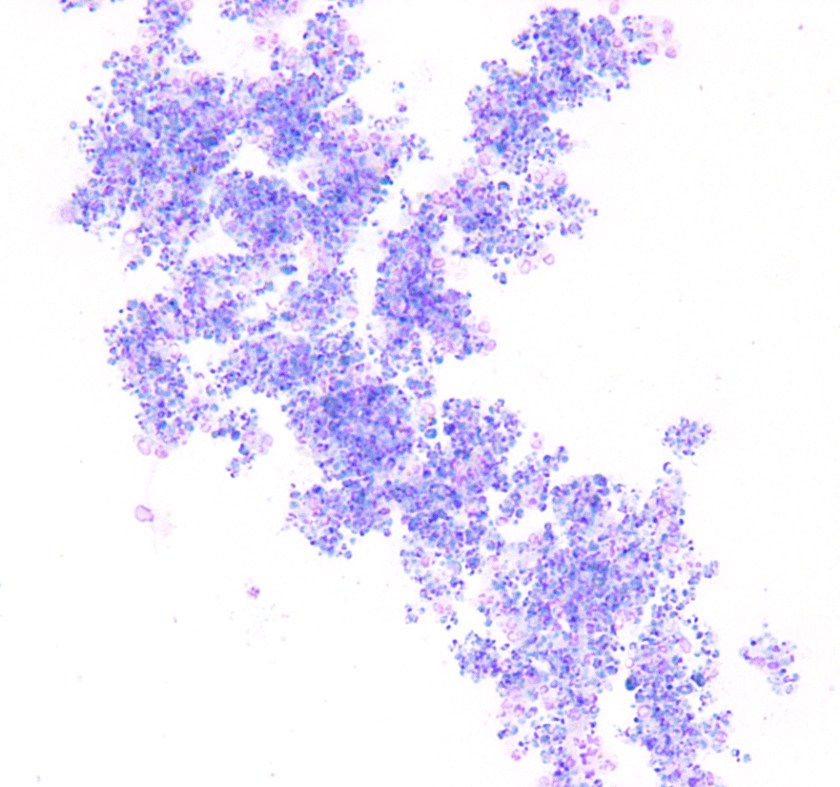

Peripheral blood findings
Geimsa-stained thin smears of the patient’s peripheral blood showed numerous infected RBCs and several individual RBCs infected with multiple trophozoites (ring forms). Occasional extra-cellular ring forms were also identified. The rings were small (<1/3 size of RBC) and delicate with 1 or sometimes 2 chromatin dots that give the appearance of headphones. Some of the ring forms were marginalized at the RBC membrane; the so-called appliqué, accollé or marginal forms. Gametocytes were not identified. Parasitemia was calculated to be 10% prior to initiation of treatment. Thick smears showed numerous aggregated ring forms. These features are consistent with Plasmosdium falciparum.
Placenta findings
H&E stained sections of FFPE placental tissue, showed focally increased intervillous macrophages associated with mild perivillous fibrin deposits. Malaria pigment (hemazoin) was scattered throughout the specimen; both intracellular pigment within macrophages and extracellular pigment entrapped in fibrin deposits and in the cytoplasm of trophoblasts was identified. Infected red blood cells, containing ring forms, were only identified in the intervillous (maternal) spaces, sometimes appearing as black, refractile dots (inset).
P. falciparum is a blood parasite transmitted by female Anopheles mosquitos found in the tropics and subtropics worldwide with the highest transmission rates in Sub-Saharan Africa and parts of Oceania. Sporozoites are injected into the bloodstream during a blood meal and travel to the liver to proliferate during the exo-erythrocytic cycle. Schizonts mature in hepatocytes and then rupture releasing merozoites into the blood. Unlike P. ovale and P. vivax, P. falciparum does not have a dormant liver stage (hypnozoites). The erythrocytic cycle begins when merozoites infect RBCs at any stage and undergo asexual multiplication (erythrocyte schizogony) producing ring forms (trophozoites) that mature into schizonts which eventually rupture and release more merozoites into the blood. Some trophozoites mature into gametocytes that can undergo sexual (sporogonic) lifecyles within the mosquito vector when consumed by a female Anopheles mosquito. Banana shaped gametocytes can be found in the peripheral blood in late P. falciparum infection. Stages other than trophozoites and gametocytes are rarely found in peripheral blood during P. falciparum infection due to sequestration in the capillaries and venules of organs.
The diagnosis of malaria is typically made by morphologic examination of 2-3 thick and thin smears over a 24 hour period. Exclusion of malaria can only be made after examination of at least 100 oil immersion fields on thick smears or 300 fields on thin smears to achieve a sensitivity of 5 parasites/μL. Flow cytometric, rapid diagnostic antigen, serologic and molecular (PCR) methods have also been developed, however are not widely available, particularly in endemic areas. The degree of parasitemia (percent infected red cells) is an important prognostic factor in P. falciparum infection, with >2% parasitemia considered severe.
Clinical symptoms are associated with asexual erythrocytic schizgony and include “malaria attacks” consisting of a cold stage (shivering), a hot stage (fevers, headaches, vomiting and seizures in children) and a sweating stage (with malaise and body aches following). These attacks classically occur every 2nd day (48 hours) in P.falciparum infection, which is a “tertian” parasite. Severe malaria and mortality is associated with complications such as cerebral malaria, severe hemolytic anemia, adult respiratory distress syndrome, coagulopathy and organ failure. Adults living in areas of stable transmission that have been infected with malaria multiple times may acquire partially immunity.
Pregnant women and children are particularly susceptible to malaria due to altered and immature immunity respectively. Pregnancy can also reduce previously acquired anti-malarial immunity. The placenta sequesters parasites contributing to severe maternal anemia, intrauterine growth retardation, intra-uterine fetal demise, premature delivery and neonatal death. The severity of complications of malaria during pregnancy increases in HIV infected women and primi-gravidae, however increased severity can extend to second and third gravidae in low transmission areas. In endemic areas where women have acquired anti-malarial immunity, severe maternal anemia may be the only symptom of P.falciparum infection during pregnancy. In some cases, parasitemia may not be detected despite placental infection. Some of the histologic features of placental malaria are described above. Several classification systems have been proposed and generally categorize placental infection (i.e. active, active-chronic, past-chronic) based on the degree of placental changes; namely presence of perivillous fibrin deposits, intervillous macrophages and fibrinoid necrosis of chorionic villi, location of hemazoin pigment, cytotrophoblast proliferation, thickening of trophoblastic basement membranes and identification of infected RBCs. Although placental malaria is a prerequisite for congenital malaria, the frequency of vertical transmission is relatively low, with a ranges reported from 0-25%. Lower transmission risk has been attributed to passive immunization of maternal antibodies and conversely higher transmission risk is associated with non-immunity of mothers.
Discussion
It is suspected, due to high parasitemia with seemingly mild symptoms, that the patient presented above had acquired partial immunity to malaria. In addition, she likely experienced Malarone (Atovaquone-proguanil) failure after initial treatment prior to emigration. She responded well to oral Coartem (artemether-lumefantrine) with non-detectable parasitemia after the third day of treatment. Both infants’ thick and thin smears were negative for parasites.
References
- Bulmer JN, Rasheed FN, Morrison L, Francis N, Greenwood BM. Placental malaria. I. Pathological classification. Histopathology. 1993;22:211–218.
- Bulmer JN, Rasheed FN, Morrison L, Francis N, Greenwood BM. Placental malaria. II. A semi-quantitative investigation of the pathological features. Histopathology. 1993;22:219–226.
- http://www.cdc.gov/malaria
- Newman RD, Robalo M, Quakyi I. Malaria during pregnancy: epidemiology, current prevention strategies, and future directions. Emerg Infect Dis [serial on the Internet]. 2004 November [March 7, 2016]. http://dx.doi.org/10.3201/eid1011.040624_09
- Uneke CJ. Impact of Placental Plasmodium falciparum Malaria on Pregnancy and Perinatal Outcome in Sub-Saharan Africa: I: Introduction to Placental Malaria. The Yale Journal of Biology and Medicine. 2007;80(2):39-50.
- Uneke CJ. Impact of Placental Plasmodium falciparum Malaria on Pregnancy and Perinatal Outcome in Sub-Saharan Africa: II: Effects of Placental Malaria on Perinatal Outcome; Malaria and HIV. The Yale Journal of Biology and Medicine. 2007;80(3):95-103.
- Uneke CJ. Impact of Placental Plasmodium falciparum Malaria on Pregnancy and Perinatal Outcome in Sub-Saharan Africa: Part III: Placental Malaria, Maternal Health, and Public Health. The Yale Journal of Biology and Medicine. 2008;81(1):1-7.
- http://www.who.int/malaria/en
-Petra Rahaman, M.D., 4th year Anatomic and Clinical Pathology resident, UT Southwestern Medical Center

-Erin McElvania TeKippe, Ph.D., D(ABMM), is the Director of Clinical Microbiology at Children’s Medical Center in Dallas Texas and an Assistant Professor of Pathology and Pediatrics at University of Texas Southwestern Medical Center.
