An elderly patient with urothelial carcinoma of the bladder was treated with intravesical Bacillus Calmette-Guerin (BCG). The patient presented nearly a year later with back pain and their laboratory tests revealed leukocytosis with neutrophilia. Magnetic Resonance Imaging (MRI) of the back showed findings suspicious for discitis/osteomyelitis of the vertebrae with epidural phegmon/abscess. The abscess fluid was sent for aerobic and anaerobic bacterial, acid-fast bacilli and fungal cultures and empiric intravenous antibiotics was commenced. Gram stain and all cultures were negative.
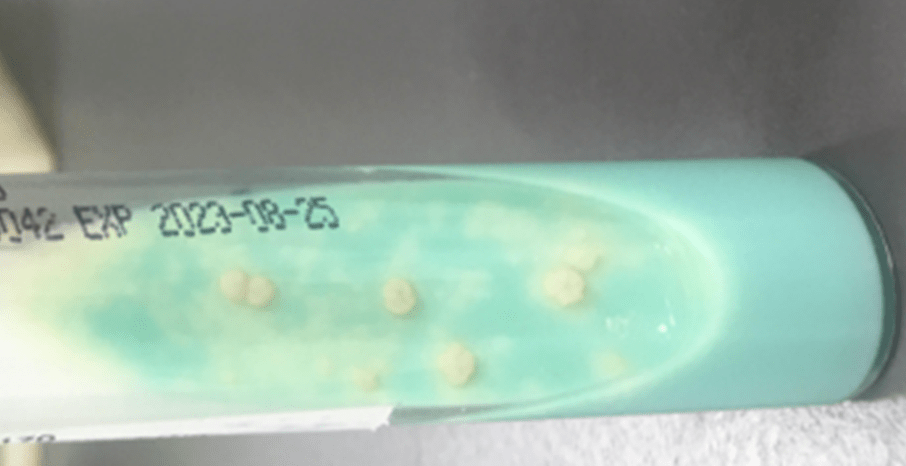
Their symptoms persisted and a repeat MRI 3 months later demonstrated similar findings. Decompression of the vertebrae was repeated and fluid from the disc space was sent for cultures. Again, Gram stain was negative while no growth was seen on aerobic, anaerobic and fungal cultures. However, about eight weeks after incubation, the Lowenstein-Jensen media showed rough and buff colonies (Figure 1). Matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF) performed on an isolate from the media confirmed the presence of Mycobacterium Tuberculosis Complex (MTBC). MALDI-TOF alone cannot distinguish between species within in the complex and thus, the result was reported as MTBC, with a comment indicating that MTBC includes M. tuberculosis and M. bovis.
Upon request, phenotypic antimicrobial susceptibility testing (AST) was performed and it showed susceptibility to all primary anti-mycobacterial drugs including Pyrazinamide (PZA). Also, due to a clinical concern for M. bovis BCG infection, further species-level was required. Therefore, the isolate was sent to the Centers for Disease Prevention and Control (CDC) for species-identification/confirmation and to the State Department of Health for confirmatory AST. Results from the CDC showed M. bovis but the State Department of Health showed PZA susceptibility, inconsistent with M. bovis which is intrinsically resistant to PZA.
Discussion
M. bovis is a part of the MTBC, which includes M. tuberculosis, M. bovis and BCG strain, M. africanum, M. microti, M. orygis, M. canetti, M. caprae, M. pinnipedii, M. suricattae. M. bovis is the main cause of tuberculosis in cattle, deer, and other mammals and compared to M. tuberculosis, is a rare cause of tuberculosis in humans. There were about 59, 273 cases of tuberculosis in the U.S between 2006 and 2013 and 770-948 (1.3-1.6%) of those were due to M. bovis[1]. However, the worldwide burden is thought to be underestimated, especially in regions with considerable consumption of unpasteurized milk.
Risk factors for M. bovis infection include practices. which expose humans to mammals with M. bovis or their products. These practices include livestock farming, veterinary medicine and consumption of unpasteurized milk. Bacillus Calmette-Guérin (BCG) is a live attenuated strain of M. bovis used as tuberculosis vaccine in many areas with relatively high prevalence of tuberculosis. However, it’s also used as adjunctive therapy for non-muscle invasive bladder cancer and unfortunately, this has rarely been complicated by M. bovis BCG infection. There were about 118 cases reported between 2004 and 2015, accounting for approximately 1-5% of patients with intravesical BCG [2]. Some of the risk factors of BCG infection are traumatic catheterization, active cystitis, persistent gross hematuria following transurethral surgery, immunosuppression and age ≥70 years.
M. bovis (and M. bovis BCG) infection is indistinguishable from M. tuberculosis clinically and radiologically. However, there is a higher incidence of extrapulmonary tuberculosis and an increased risk of scrofula -infection of the lymph node(s) in proximity to the mouth and esophagus- and gastrointestinal disease.1 The laboratory workup and findings are also similar. Microscopically, primary specimen smears are screened using auramine-rhodamine stain which is the most sensitive, while carbol-fuchsin (Ziehl-Neelsen or Kinyoun) stain is used to confirm presence of growing acid-fast bacteria. MTBC is slow-growing on culture, requiring at least 7 days to form colonies on solid media. M. bovis colonies appear small and rounded, with irregular edges and a granular surface on egg-based media, and small and flat on agar media.3
MALDI-TOF which is used reliably in the workup of many bacterial infections also can’t differentiate between MTBC species. Where available, biochemical testing can be used to differentiate M. tuberculosis from M. bovis (see table 1). However, this is being replaced by newer modalities especially DNA hybridization or polymerase chain reaction (PCR)-based molecular methods such as the Region of Deletion analysis.
Species level differentiation between M. bovis and M. tuberculosis is extremely important when M. bovis is suspected because the first line drugs for treating M. tuberculosis are Rifampicin, Isoniazide, PZA and Ethambutol, and M. bovis is intrinsically resistant to PZA.1,3 The observation of this mono-resistance pattern on AST of MTBC isolate raises the suspicion for M. bovis and may warrant further workup. Importantly however, M. bovis infection cannot be excluded on the basis of an MTBC AST showing susceptibility to PZA, as this AST is difficult to perform and identifies only about 80% of M. bovis cases and approximately 7% of M. bovis cases are incorrectly reported as PZA susceptible.2 When required, isolates should be sent to a public health laboratory for M. bovis confirmation.

References
- Talbot, E. (n.d.). Mycobacterium bovis. UpToDate. Retrieved December 11, 2022, from https://www.uptodate.com/contents/mycobacterium-bovis?search=m%20bovis&source=search_result&selectedTitle=1~38&usage_type=default&display_rank=1
- O’Donnell, M., & Orr, P. (n.d.). Infectious complications of intravesical BCG immunotherapy. UpToDate. Retrieved December 11, 2022, from https://www.uptodate.com/contents/infectious-complications-of-intravesical-bcg-immunotherapy?search=bcg%20bladder%20cancer&source=search_result&selectedTitle=2~150&usage_type=default&display_rank=2
- Pfyffer, G. “Mycobacterium: General Characteristics, Laboratory Detection, and Staining Procedures.” In Manual of Clinical Microbiology, Eleventh Edition, pp. 536-569. American Society of Microbiology, 2015.

-Adesola Akinyemi, M.D., MPH, is a fourth year anatomic and clinical pathology resident and Chief resident at University of Chicago (NorthShore Program). He will be undergoing fellowship trainings in cytopathology (Northwell Health, NY) and oncologic surgical pathology (Memorial Sloan Kettering Cancer Center, NY). He is also passionate about health outcomes improvement through systems thinking and design, and other aspects of healthcare management.
Twitter: @AkinyemiDesola

-Paige M.K. Larkin, PhD, D(ABMM), M(ASCP)CM is the Director of Molecular Microbiology and Associate Director of Clinical Microbiology at NorthShore University HealthSystem in Evanston, IL. Her interests include mycology, mycobacteriology, point-of-care testing, and molecular diagnostics, especially next generation sequencing.
