Case History
An adult woman presented to the emergency department five days after undergoing gynecological surgery. The patient presented with fever and severe right lower quadrant abdominal pain. Computed tomography (CT) scan with contrast showed a ring enhanced loculated fluid collection within the cervix, which was concerning for an abscess. The patient was admitted to the hospital and empirically started on piperacillin-tazobactam, but continued to have fevers despite the antibiotics. Blood and urine samples were sent to the microbiology lab for bacterial culture but no organisms were isolated from either source. Two days later, the patient underwent a diagnostic laparoscopy, abdominal wash-out, and drainage of the abscess. The abscess fluid was sent for aerobic and anaerobic bacterial culture. Gram stain of the specimen showed 3+ white blood cells with no organism seen. The anaerobic culture grew 4+ pinpoint white colonies on blood agar after 5 days of incubation. Further identification of these colonies by MALDI-TOF MS revealed Mycoplasma hominis.
Discussion
Mycoplasma hominis is often a commensal of the urogenital tract, but it can be associated with urogenital infections including pelvic inflammatory disease (PID), pregnancy-related infections, and urethritis in males. There are multiple risk factors for Mycoplasma hominis genital infection including young adult age, multiple sexual partners, and pregnancy. Immunocompromised patients have a higher risk for Mycoplasma hominis extragenital infections as nearly 50% of reported extragenital infections isolated from immunocompromised patients.2 Mycoplasma hominis can cause extragenital infections including septic arthritis,4 septicemia, osteitis, retroperitoneal abscesses3, mediastinitis,1 and pneumonia.
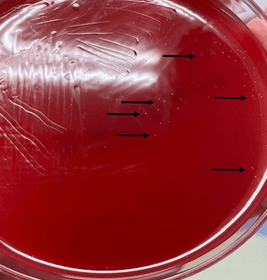
Laboratory diagnosis of Mycoplasma hominis is challenging due to the fastidious nature of the organism and its lack of the cell wall makes it undetectable by gram staining. The more specific tests including molecular tests for Mycoplasma hominis are not routinely ordered unless there is a strong clinical suspicion, which makes diagnosis more challenging. Mycoplasma hominis can grow on 5% sheep blood and chocolate agars; however, such growth is very slow and may take from 2 to 7 days of incubation.1 The usual growth of Mycoplasma hominis reveals tiny-sized pinpoint colonies that may be overlooked (Image 1). Once growth is observed, MALDI-TOF MS can be used for identification.6
There are multiple types of selective media for the isolation of Mycoplasma hominis including SP4 agar supplemented with arginine, Hayflick agar, A7, and A8 agars.9 Both A7 and A8 agars contain arginine to enrich Mycoplasma growth but differ in the antibiotic content used to inhibit the growth of other commensals. Agar plates should be put for incubation under 5 to 10% CO2 or under anaerobic conditions at 35°C for at least 5 days.9 On these selective agars Mycoplasma hominis has a characteristic fried egg appearance and can be seen by the aid of a stereomicroscope. However, use of specific agar is not widespread.
Molecular testing of Mycoplasma hominis using nucleic acid amplification (NAAT) assays such as polymerase chain reaction (PCR) is a more sensitive and faster method of detecting Mycoplasma hominis compared with culture. However, PCR is neither widely available nor standardized. PCR assays for Mycoplasma hominis generally use 16S rRNA as a gene target, but other targets, including gap, fstY, and yidC, have been developed.7 Clinical picture should be taken into account when evaluating the significance of a positive PCR test as Mycoplasma hominis can be a commensal organism and PCR does not distinguish between live and dead organisms.
Mycoplasma spp. lack a peptidoglycan cell wall. This makes Mycoplasma spp. intrinsically resistant to β-lactams and to all antibiotics, which target the cell wall, including glycopeptide antibiotics. Mycoplasma hominis is also resistant to rifampin, sulfonamides and trimethoprim. Tetracyclines, macrolides, and fluoroquinolones are often used. Antimicrobial susceptibility testing is rarely performed, with only a few specialized laboratories offering the testing. Clinical and laboratory standards institute guidelines (CLSI M43) is followed using microbroth dilution. Agar disc diffusion testing is not used for Mycoplasma hominis as there is no correlation between inhibitory zones and minimal inhibitory concentrations.8 Mycoplasma hominis can be evaluated for susceptibility to clindamycin, tetracycline, and levofloxacin.10
After isolation of Mycoplasma hominis was reported, doxycycline was added to the patient’s antibiotic regimen. The patient responded well with subsiding of the fever and stabilization of her vital signs.
References
- Xiang, L., & Lu, B. 2019. Infection due to Mycoplasma hominis after left hip replacement: case report and literature review. BMC infectious diseases, 19(1), 50. https://doi.org/10.1186/s12879-019-3686-z
- Meyer RD, Clough W. 1993. Extragenital Mycoplasma hominis infections in adults: emphasis on immunosuppression. Clin Infect Dis. Suppl 1:S243-9. doi: 10.1093/clinids/17.supplement_1.s243. PMID: 8399923.
- Adams M, Bouzigard R, Al-Obaidi M, Zangeneh TT. 2020. Perinephric abscess in a renal transplant recipient due to Mycoplasma hominis: Case report and review of the literature. Transpl Infect Dis.(5):e13308. doi: 10.1111/tid.13308. Epub 2020 Jul 7. PMID: 32378787.
- Luttrell LM, Kanj SS, Corey GR, Lins RE, Spinner RJ, Mallon WJ, Sexton DJ. 1994. Mycoplasma hominis septic arthritis: two case reports and review. Clin Infect Dis.19(6):1067-70. doi: 10.1093/clinids/19.6.1067. PMID: 7888535.
- Wylam ME, Kennedy CC, Hernandez NM, Peters SG, Maleszewski JJ, Cassivi SD, Scott JP. 2013. Fatal hyperammonemia caused by Mycoplasma hominis. Lancet 382:1956.
- Pereyre S, Tardy F, Renaudin H, Cauvin E, Del Pra Netto Machado L, Tricot A, Benoit F, Treilles M, Bebear C. 2013. Identification and subtyping of clinically relevant human and ruminant mycoplasmas by use of matrix-assisted laser desorption ionization–time of flight mass spectrometry. J Clin Microbiol 51:3314–3323.
- Ferandon C, Peuchant O, Janis C, Benard A, Renaudin H, Pereyre S, Bebear C. 2011. Development of a real-time PCR targeting the yidC gene for the detection of Mycoplasma hominis and comparison with quantitative culture. Clin Microbiol Infect 17:155–159.
- Clinical and Laboratory Standards Institute. 2011. Methods for antimicrobial susceptibility testing for human mycoplasmas; approved guideline M43-A. Clinical and Laboratory Standards Institute, Wayne, PA.
- Stabler S, Faure E, Duployez C, Wallet F, Dessein R, Le Guren R. 2021. Mycoplasma hominis extragenital abscess. J Clin Microbiol, 59(4). https://doi.org/10.1128/JCM.02343-20
- https://sites.uab.edu/dml/tests/
–Omar Abdelsadek, MD is a PGY-1 (AP/CP) Pathology Resident at University of Chicago (NorthShore) Pritzker School of Medicine.

-Paige M.K. Larkin, PhD, D(ABMM), M(ASCP)CM is the Director of Molecular Microbiology and Associate Director of Clinical Microbiology at NorthShore University HealthSystem in Evanston, IL. Her interests include mycology, mycobacteriology, point-of-care testing, and molecular diagnostics, especially next generation sequencing.
