Every FNA ROSE attended where the patient is conscious and attentive can be tricky to navigate. You have to remain cognizant of your word choice, your demeanor, and the delivery of your adequacy statement to the clinician. The patient is already in a heightened state of awareness because he or she is about to be probed with a needle (or six!) for a test that is likely to rule out a benign or malignant process. I prefer to go into my biopsies with some sort of clinical picture and as many details as I can retain – is there a previous history of cancer? Where is the lesion located? Is it a single mass or are there multiple lesions? What does the radiologic imaging suggest? Are there any elevated serum tumor markers? I need to be able to walk the walk and talk the talk. However, there are rare instances when cytotechs are asked to rush down to an unscheduled add-on biopsy where we have yet to research the impression documented in the patient’s medical record. In those situations, I ask the clinician (typically an interventional radiologist) all the questions I can think of while still emulating some form of confidence to the patient.
I entered the procedure room and greeted the radiologist, radiology fellow, tech, nurse, and the patient, a 57-year-old male who was prone and alert on the table. I jotted down notes during the timeout and pulled the radiologist aside to ask, “does the patient have a history of cancer?” In this case, the answer was “they have a soft tissue tumor in the left gluteus, which is what we’re biopsying.” “Alright, let’s get those differentials rolling – sarcoma; after my hibernoma experience – a lipomatous tumor; or could it be a carcinoma (because yes, I’ve seen a lung adenocarcinoma metastasize to the gluteal muscle before)? Hmm… what else? What other mesenchymal tumors could originate here… or metastasize here?” My brainstorming balloon was popped by the radiologist asking if I was ready for the first needle pass. I replied, “Yes, of course!” I glance over at the patient and smile, trying to assure him AND myself that I’ll be able to give him a definitive answer to his puzzle.
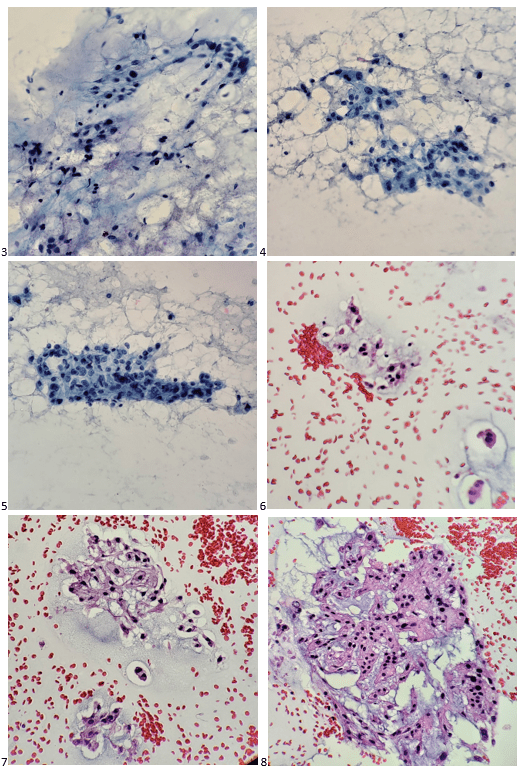
Here’s what I visualized under the microscope after I stained the first air-dried smear in our Diff-Quik solutions.

My inner monologue became: “Well, it’s not a sarcoma or a carcinoma. It doesn’t look malignant. Not quite a hibernoma. What is with that myxoid matrix? It’s not mucinous or serous, so… what is it…? It’s granular! Wait. Those nuclei. They’re so… what’s the word? It’s definitely representative of the lesion. Regardless, it’s adequate!” I turned away from my microscope to face the team – “The sample is adequate. May I have a few more passes for my cell block, and will you collect core biopsies, too? “Yes and yes,” the radiologist replied. I smiled again at the patient, and he mouthed, “thank you.” “Phew, mission accomplished,” I thought. “Now what the heck are those hallmark cells called mixed in with a majority of epithelioid cells arranged in chords?”
I climb the stairs up to the lab and do a quick Google search. “DUH! Physaliphorous cells!” These cells have a distinct feature where the nucleus is centrally located but is also scalloped by cytoplasmic vacuoles. There weren’t as many physaliphorous (physaliferous) cells as I had hoped to appreciate. Some of the cells looked lipoblastic in nature with larger vacuoles displacing the nuclei to the periphery, almost signet ring in nature, many were cuboidal. But that was it… those cells! Now, imagine the scene in Finding Nemo where Nemo struggles to tell his classmates he lives in an anemone. That was my garbled attempt at pronouncing “physaliphorous” to the attending pathologist when sharing my interpretation. She looked at me like I was saying anything other than the word I was trying to reproduce. I cannot blame her; I still turn beet-red at the memory. But I was convinced that a chordoma was the tumor I presented to her.
After I processed my FNA, I examined the patient’s electronic health record to see if he had any previously biopsied neoplasms on file, and much to my surprise, the patient was diagnosed with a primary chordoma of the sacrum and treated with en bloc resection and radiation in 2013. Mutation analysis was performed on the resection of this chordoma, which exhibited a homozygous loss of CDK2NA (p16). The patient had one recurrence at an outside facility in 2015 and transferred his care to our institution for follow-up. Now, the patient presented with this gluteal metastasis and soon thereafter, a paraspinal metastasis. As the patient’s chordoma did not completely respond to radiation, the clinical care team turned to the tyrosine kinase inhibitor, Gleevec, which was discontinued due to disease progression. The patient’s regimen then went on to include sunitinib, which was also discontinued due to disease progression, palbociclib, then nivolumab, followed by radiation to the thoracic spine, sorafenib, and now is on a clinical trial for patients with advanced refractory cancers.
When I turned in my Diff-Quik & Pap-stained slides and the cell block H&E sections with a diagnosis of chordoma the next day, the attending cytopathologist paged through one of our cytology texts to a tidbit on chordomas before signing out the case. She reviewed the section with me. Other than the unique physaliphorous cells, it turns out a diagnosis of chordoma is fairly rare, as it is the only malignancy derived from the notochord, typically occurring at either end of the axial skeleton.1 Metastasis of these tumors is also rare, so this case of widespread metastatic disease was even more intriguing to me.
References
- Cibas, E. S., & Ducatman, B. S. (2009). Cytology: Diagnostic Principles and Clinical Correlates, Expert Consult – Online and Print (3rd ed.). Saunders.

-Taryn Waraksa, MS, SCT(ASCP)CM, CT(IAC), has worked as a cytotechnologist at Fox Chase Cancer Center, in Philadelphia, Pennsylvania, since earning her master’s degree from Thomas Jefferson University in 2014. She is an ASCP board-certified Specialist in Cytotechnology with an additional certification by the International Academy of Cytology (IAC). She is also a 2020 ASCP 40 Under Forty Honoree.
