Clinical History
A 55 year old patient with past medical history of stage IV non-Hodgkin’s lymphoma on rituximab and Campylobacter jejuni bacteremia 1 year prior presented to the Emergency Department on the orders of their primary care provider, after outpatient blood cultures grew gram negative bacilli resembling Campylobacter species. Their symptoms included a 1-2 month history of fatigue and weakness and a 3 week history of intermittent fevers and chills with developing productive cough, sinus pressure, sore throat, progressive dyspnea on exertion, nausea, and decreased appetite.
Laboratory Findings
Initial (outpatient) blood culture was positive in the aerobic bottle at 60.1 hours, with the initial gram stain showing no organisms. The bottle was placed back in the analyzer and flagged positive again, at which point a second gram stain was performed, which again showed no organisms. An acridine orange stain was performed (Image 1), revealing multiple spiral/”gull shaped” rods. A third gram stain (Image 2) with more time allowed for safranin staining revealed faint gram negative rods. MALDI-TOF MS was attempted with no identification. The culture growth was sent to a reference laboratory and was identified via sequencing as Helicobacter species. The organism was not viable for susceptibility testing.
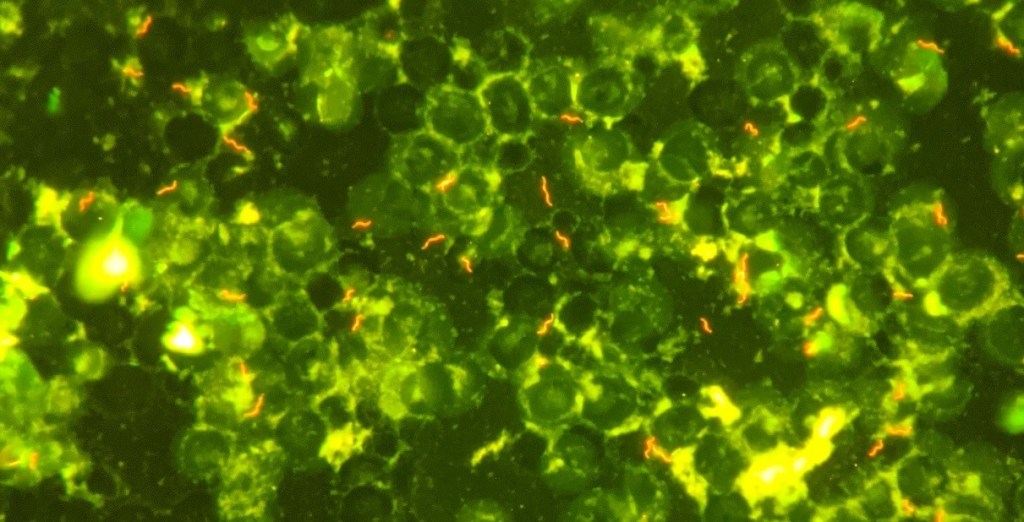
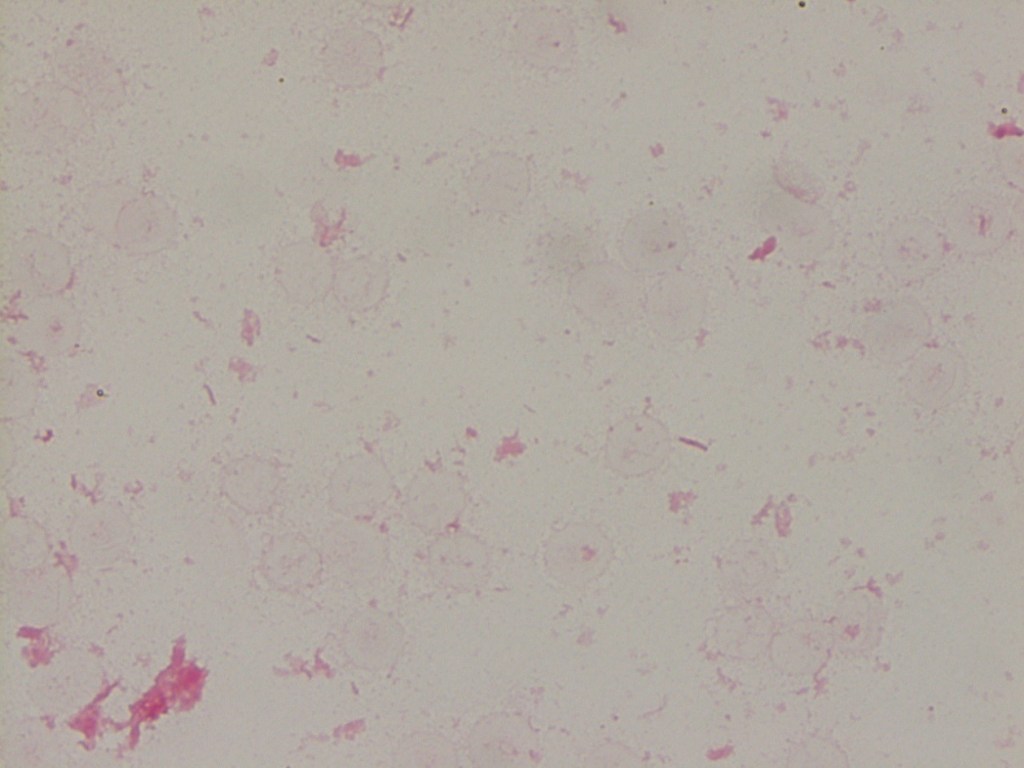
Two sets of subsequent blood cultures also grew gram negative bacilli at 65 and 67 hours. The blood culture broth from one of these cultures was also sent to the reference lab, but again did not have viable growth for susceptibility testing.
Discussion
The genus Helicobacter includes 35 species, consisting of gram negative spiral bacilli, previously considered to be part of the Campylobacter genus. Pathogenic species are classically associated with the gastrointestinal tract as they are able to survive in the harsh acidic conditions of the human stomach. The most common clinically relevant species is H. pylori, which is associated with gastric ulcers as well as other inflammatory processes in the stomach and duodenum. In prior reports, bacteremia caused by Helicobacter species is typically associated with some other underlying disease process, such as malignancy, immunocompromised state, or disruption of the GI mucosal barrier (1, 2, 3, 4, 5, 6).
Helicobacter spp. are similar in morphology to Campylobacter spp. on a gram stain; given the patient’s prior history of C. jejuni bacteremia, it was not unreasonable for the gram smear to initially be called consistent with Campylobacter spp. However, the clinical course and antibiotic susceptibility profiles of Helicobacter and Campylobacter bacteremia cases can differ in important ways. Further, susceptibilities can differ between different species of Helicobacter. There are no established guidelines for the treatment of Helicobacter spp. bacteremia and breakpoints for antibiotic susceptibility testing for some Helicobacter species have not been established. (7)
The patient in this case was discharged on a course of azithromycin with clinical improvement: at that time, the sequencing result revealing Helicobacter had not yet been received, and the clinical team was acting on the belief that the organism in the patient’s blood was a recurrence of the previous Campylobacter infection. On a follow up outpatient appointment with Infectious Disease, wherein sequencing results were available, tetracycline was prescribed due to concern about the possibility of resistance or relapsing infection.
References
- Abidi, Maheen Z., et al. “Helicobacter Canis Bacteremia in a Patient with Fever of Unknown Origin.” Journal of Clinical Microbiology, vol. 51, no. 3, 2013, pp. 1046–1048.
- Araoka, Hideki, et al. “Clinical Characteristics of Bacteremia Caused by Helicobacter Cinaedi and Time Required for Blood Cultures To Become Positive.” Journal of Clinical Microbiology, vol. 52, no. 7, 2014, pp. 2745–2745.
- De Luca, et al. “Helicobacter Pylori Bacteremia: An Unusual Finding.” Infectious Disease Reports, vol. 8, no. 3, 2016, pp. 74–75.
- Han, Xiang Y., et al. “Helicobacter Pylori Bacteremia with Sepsis Syndrome.” Journal of Clinical Microbiology, vol. 48, no. 12, 2010, pp. 4661–4663.
- Imataki, Osamu, et al. “Enteral Malakoplakia Prior to Helicobacter Cinaedi Bacteremia.” American Journal of Gastroenterology, vol. 112, no. 1, 2017, pp. 187–188.
- Saito, Sho, et al. “Helicobacter Fennelliae Bacteremia: Three Case Reports and Literature Review.” Medicine, vol. 95, no. 18, 2016, p. e3556.
- Yamamoto, Kei, et al. “Comparison of the Clinical and Microbiological Characteristics of Campylobacter and Helicobacter Bacteremia: the Importance of Time to Blood Culture Positivity Using the BACTEC Blood Culture Systems.” BMC Research Notes, vol. 10, no. 1, 2017, pp. 1–6.
-Tom Koster, DO is a 1st year Anatomic and Clinical Pathology Resident at the University of Vermont Medical Center.

-Christi Wojewoda, MD, is the Director of Clinical Microbiology at the University of Vermont Medical Center and an Associate Professor at the University of Vermont.
