Case History
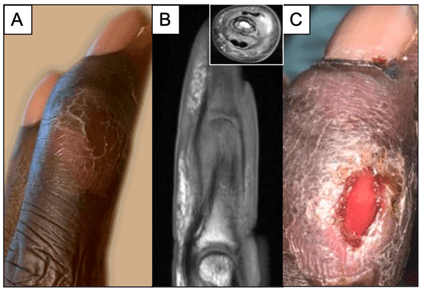
A thirty year old male on dialysis with past medical history of nephrotic syndrome and end-stage renal disease presented to the emergency department due to complaints of severe abdominal pain. Culture of paracentesis fluid confirmed a diagnosis of bacterial peritonitis, and he was admitted for intra-peritoneal antibiotic therapy. During his admission however, he also complained of progressive worsening right middle finger pain and swelling after smashing his finger in a door one month prior. Upon exam the finger appeared visibly swollen with slight discoloration, but exhibited full range of motion (Image 1A). Imaging revealed osteomyelitis of the intermediate and distal phalanx, including involvement of the distal interphalangeal joint (Image 1B). Additionally, a small abscess was detected (Image 1B, inset) requiring incision and drainage (I&D). Following the I&D, the patient was discharged home on broad spectrum antimicrobial therapy pending results of intra-operative cultures. The patient was readmitted the following week for severe electrolyte imbalance due to a missed dialysis session. His right middle finger continued to cause him significant pain, and the I&D site appeared poorly healing with excess fluid drainage (Image 1C).
Laboratory Identification
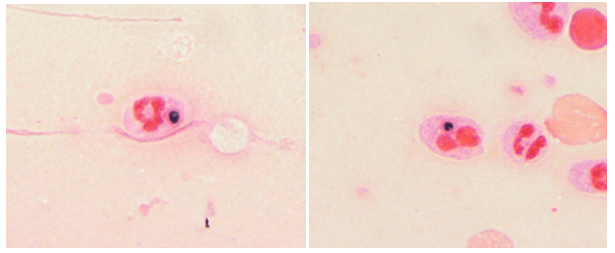
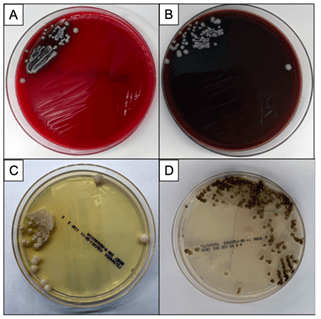
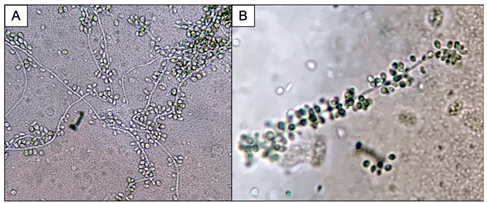
Bacterial, fungal and mycobacterial cultures of purulent material from the I&D were obtained. Following four days of incubation, creamy white yeast-like colonies appeared on blood and chocolate agar plates at 35°C (Images 2A, B). Fungal medias grew colonies which were initially white (Image 2C), but became more wrinkled with increased central dark coloration and white periphery (Image 2D) within two weeks at 30°C. Cellophane tape preparations with lactophenol cotton blue were performed for microscopic evaluation of the mature colonies from potato dextrose agar. Narrow, septate hyphae with slender, tapering conidiophores were observed (Image 3A). The conidiophores had many small, tear-shaped hyaline conidia forming “daisy-like” clusters (Image 3B). Based upon these morphologic characteristics, the organism was identified as member of the Sporothrix schenckii complex.
Discussion
Sporotrichosis (also known as rose gardener’s disease) is caused by dimorphic molds belonging to the Sporothrix schenckii complex. These fungi inhabit soil, plant matter and decaying vegetation, thus explaining their traditional association with gardening. As thermally dimorphic fungi, members of the Sporothrix schneckii complex exhibit both yeast and filamentous mold forms. The yeast colonies are smooth with cream or tan coloration when grown close to body temperature (35℃). Microscopically, the yeasts appear as round, oval and fusiform budding cells3,4 best visualized with GMS or PAS stains due to their relative infrequency when observed in tissue. These are commonly referred to as “cigar bodies”, and are typically seen on histopathology, sometimes with accompanying pyogranulomatous inflammation or suppurative granulomas.2 By contrast, the mold colonies begin small, white to pale-orange and become moist, wrinkled, leathery, or velvety and often darken to a salt-and-pepper brown-black as the mold ages. Microscopically, narrow, septate hyphae with slender, tapering conidiophores can be visualized. The conidiophore apex is often slightly swollen and bears many small, tear-shaped or almost round hyaline conidia (2-3 X 2-6 µm) on delicate hair-like denticles, forming the characteristic “rosette” or “daisy-like” clusters in young cultures.1,3,4 This also illustrates the etymology of the organism: from latin, spor (spore) + thrix (hair).5
The most common presentation of sporotrichosis is a lymphocutaneous form originating from a puncture or trauma that inoculates contaminated material into the wound. A pustule forms in subcutaneous tissue followed by ulceration and development of secondary lesions via lymphatic spread in a distal to proximal fashion.2 This form often occurs in immunocompetent individuals. More uncommon forms include osteoarticular, pulmonary, and disseminated infections, usually associated with immunocompromised hosts and alcoholics. In our case of osteoarticular sporotrichosis, the patient suffered from nephrotic syndrome which is often associated with immunodeficiency. The key inciting event to the development of his infection was likely the traumatic injury to his finger which was followed by wound inoculation of soil/material containing the organism. Following his diagnosis, the patient revealed that he was an avid gardener.
In conclusion, the patient was started on oral itraconazole initially for medical management. However, due to continued pain after a few weeks of therapy, the patient opted for amputation of the involved portion of the finger. Following successful amputation, the patient is to remain on itraconazole for one year due to concern for possible surgical margin involvement. He has healed well to date, with no continued pain in the remaining portion of the finger.
References
- Love GL, Ribes JA. 2018. Color Atlas of Mycology, An Illustrated Field Guide Based on Proficiency Testing. College of American Pathologists (CAP), p. 214-218.
- Pritt BS. 2018. Atlas of Fundamental Infectious Diseases Histopathology. College of American Pathologists (CAP), p. 120-122.
- Walsh TJ, Hayden RT, Larone DH. 2018. Larone’s Medically Important Fungi, A Guide to Identification. 6th Ed. ASM Press, p. 53, 178-180.
- St-Germain G, Summerbell R. 2011. Identifying Fungi: A Clinical Laboratory Handbook, 2nd Edition, Star Publishing Company Inc., pg. 238-239.
- Sellera, FP and Larsson, CE. 2019. Sporothrix schneckii. Etymologica. Emerg. Infect. Dis. 25(9):1631.
–John Markantonis, DO is the current Medical Microbiology fellow at UT Southwestern and will be completing his Clinical Pathology residency in 2022. He is also interested in Transfusion Medicine and parasitic diseases.
–Samira Peyrovi, M(ASCP)CM is a medical technologist at UT Southwestern Medical Center. She graduated with a degree in biology from Texas Women’s University and has worked in the microbiology section at Clements University Hospital for the past 5 years.

-Andrew Clark, PhD, D(ABMM) is an Assistant Professor at UT Southwestern Medical Center in the Department of Pathology, and Associate Director of the Clements University Hospital microbiology laboratory. He completed a CPEP-accredited postdoctoral fellowship in Medical and Public Health Microbiology at National Institutes of Health, and is interested in antimicrobial susceptibility and anaerobe pathophysiology.